UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
For the fiscal year ended
or
For the transition period from _________ to _________
Commission file number:
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including
area code:
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Securities registered pursuant to Section 12(g) of the Act
Warrants, each exercisable for one-half of one share of common stock, $0.0001 par value, at an exercise price of $11.50 per share.
Indicate by check mark if the registrant is a well-known seasoned
issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not required to file
reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ☐
Indicate by check mark whether the registrant (1) has filed all
reports required by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such
shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the
past 90 days.
Indicate by check mark whether the registrant has submitted electronically
every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the
preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| ☒ | Smaller reporting company | ||
| Emerging Growth Company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on
and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section
404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section
12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction
of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as
defined in Rule 12b-2 of the Exchange Act). Yes ☐
No
On June 30, 2023, the last day of the Registrant’s most recently
completed second fiscal quarter, the aggregate market value of the Registrant’s shares of Common Stock held by non-affiliates of
the Registrant was $
The number of shares outstanding of the Registrant’s shares of
Common Stock as of March 28, 2024 was
BIOMX INC.
Annual Report on Form 10-K for the Year Ended December 31, 2023
i
On March 15, 2024, BiomX Inc. acquired Adaptive Phage Therapeutics, Inc., a Delaware corporation, or APT, and such acquisition, the Acquisition, pursuant to an agreement and plan of merger, or the Merger Agreement, by and among BiomX Inc., APT, BTX Merger Sub I, Inc., a Delaware corporation, and BTX Merger Sub II, LLC, a Delaware limited liability company. References in this Annual Report on Form 10-K , or the Annual Report to the “Company,” “BiomX,” “we,” “us” or “our” mean BiomX Inc. and its consolidated subsidiaries, including APT, unless otherwise expressly stated or the context indicates otherwise, provided, however, that all financial information included in this Annual Report, including financial information as of and for the years ended December 31, 2023 and December 31, 2022 and other information as of a date before March 15, 2024, unless noted specifically, does not include APT. References in this Annual Report to BiomX Ltd. mean BiomX Ltd., our wholly owned Israeli subsidiary. The description of the Company herein describes the post Acquisition Company and reflects the integration of APT’s business. As further described elsewhere in this Annual Report, on October 28, 2019, Chardan Healthcare Acquisition Corp., a special purpose acquisition company, combined with BiomX Ltd. in the Business Combination (as defined below) and changed its name to BiomX Inc.
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended or the Exchange Act. The statements contained in this Annual Report that are not purely historical are forward-looking statements. Forward-looking statements include statements about our expectations, beliefs, plans, objectives, intentions, assumptions and other statements that are not historical facts. Words or phrases such as “anticipate,” “believe,” “continue,” “estimate,” “expect,” “intend,” “may,” “ongoing,” “plan,” “potential,” “predict,” “project,” “will” or similar words or phrases, or the negatives of those words or phrases, may identify forward-looking statements, but the absence of these words does not necessarily mean that a statement is not forward-looking. Examples of forward-looking statements in this Annual Report include, but are not limited to, statements regarding our disclosure concerning our operations, cash flows, financial position and also regarding our preclinical and clinical development plans, the safety, tolerability and efficacy of our phage therapy and the conducting, design, aims and timing of its preclinical and clinical studies and announcing results thereof.
Forward-looking statements appear in a number of places in this Annual Report including, without limitation, in the sections entitled “Management’s Discussion and Analysis of Financial Conditions and Results of Operations,” and "Business.” The risks and uncertainties include, but are not limited to:
| ● | the ability to generate revenues, and raise sufficient financing to meet working capital requirements; |
| ● | the integration of the operations of APT into the Company; |
| ● | the receipt of our stockholders’ approval to certain proposals relating to the Acquisition and related private investment transaction; |
| ● | the unpredictable timing and cost associated with our approach to developing product candidates using phage technology; |
| ● | political and economic instability, including, without limitation, due to natural disasters or other catastrophic events, such as the Russian invasion of Ukraine and world sanctions on Russia, Belarus, and related parties, terrorist attacks, hurricanes, fire, floods, pollution and earthquakes; |
| ● | obtaining U.S. Food and Drug Administration, or FDA, acceptance of any non-U.S. clinical trials of product candidates; |
| ● | our ability to enroll patients in clinical trials and achieve anticipated development milestones when expected; |
| ● | the ability to pursue and effectively develop new product opportunities and acquisitions and to obtain value from such product opportunities and acquisitions; |
| ● | penalties and market withdrawal associated with any unanticipated problems with product candidates and failure to comply with labeling and other restrictions; |
| ● | general economic conditions, our current low stock price and other factors on our operations, the continuity of our business, including our preclinical and clinical trials, and our ability to raise additional capital; | |
| ● | expenses associated with compliance with ongoing regulatory obligations and successful continuing regulatory review; |
| ● | market acceptance of our product candidates and ability to identify or discover additional product candidates; |
ii
| ● | our ability to obtain high titers for specific phage cocktails necessary for preclinical and clinical testing; |
| ● | the availability of specialty raw materials and global supply chain challenges; |
| ● | the ability of our product candidates to demonstrate requisite, safety and efficacy for drug products, or safety, purity and potency for biologics without causing adverse effects; |
| ● | the success of expected future advanced clinical trials of our product candidates; |
| ● | our ability to obtain required regulatory approvals; |
| ● | delays in developing manufacturing processes for our product candidates; |
| ● | competition from similar technologies, products that are more effective, safer or more affordable than our product candidates or products that obtain marketing approval before our product candidates; |
| ● | the impact of unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives on our ability to sell product candidates or therapies profitably; |
| ● | protection of our intellectual property rights and compliance with the terms and conditions of current and future licenses with third parties; |
| ● | infringement on the intellectual property rights of third parties and claims for remuneration or royalties for assigned service invention rights; |
| ● | our ability to acquire, in-license or use proprietary rights held by third parties necessary to our product candidates or future development candidates; |
| ● | ethical, legal and social concerns about synthetic biology and genetic engineering that may adversely affect market acceptance of our product candidates; |
| ● | reliance on third-party collaborators; |
| ● | political, economic and military instability in the State of Israel, and in particular, the war in Gaza following the October 7 attack, additional potential conflicts with other middle eastern countries and the continuation of the proposed judicial and other legislation reform by the Israeli government; |
| ● | our ability to attract and retain key employees or to enforce the terms of noncompetition agreements with employees; |
| ● | the failure to comply with applicable laws and regulations other than drug manufacturing compliance; |
| ● | potential security breaches, including cybersecurity incidents; and |
| ● | other factors discussed in the section of this report entitled “Risk Factors” beginning on page 29. |
Forward-looking statements are subject to known and unknown risks and uncertainties and are based on our management’s potentially inaccurate assumptions that could cause actual results to differ materially from those expected or implied by the forward-looking statements. While these statements are based upon information available to us as of the filing date of this Annual Report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. Actual results could differ materially from those anticipated in forward-looking statements for many reasons, including the factors discussed in the section of this Annual Report entitled “Risk Factors”. Except as may be required by applicable law, we undertake no obligation to publicly revise any forward-looking statement to reflect circumstances or events after the date of this Annual Report or to reflect the occurrence of unanticipated events. You should, however, review the factors and risks we describe in the reports we will file from time to time with the U.S. Securities and Exchange Commission or the SEC, after the date of this Annual Report.
iii
RISK FACTORS SUMMARY
The summary below provides an overview of many of the risks the Company faces, and a more detailed discussion of risks can be found in Item 1A. “Risk Factors” below. You should carefully consider these risks and uncertainties when investing in our securities. The principal risks and uncertainties affecting our business include, but are not limited to, the following:
| ● | We are a clinical-stage company with limited operating history and have incurred losses since our inception. We anticipate that we will continue to incur significant expenses, and we will continue to incur significant losses for the foreseeable future. |
| ● | We will need to raise additional capital in the future to support our operations which may not be available at terms that are favorable to us and might cause significant dilution to our stockholders or increase our debt towards third parties. |
| ● | Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern, which could prevent us from obtaining new financing on reasonable terms or at all. |
| ● | There is no guarantee that our acquisition of APT will increase stockholder value. | |
| ● | We are seeking to develop product candidates using phage technology, an approach for which it is difficult to predict the potential success and time and cost of development. To our knowledge, no bacteriophage has thus far been approved as a drug in the United States or in the European Union. |
| ● |
We are required to use reasonable best efforts to solicit stockholder approval for the conversion of shares of Convertible Preferred Stock (as defined below) and the exercise of the Warrants (as defined below) issued in the Acquisition (as defined below) and the March 2024 PIPE (as defined below). If we do not obtain such approval within 150 days of the initial issuance of the Convertible Preferred Stock, we could be required to cash settle the Convertible Preferred Stock. |
| ● | Our product candidates must undergo clinical testing which may fail to demonstrate the requisite safety and efficacy for drug products, or safety, purity, and potency for biologics, and any of our product candidates could cause adverse effects, which would substantially delay or prevent regulatory approval and/or commercialization. | |
| ● |
We have not completed composition development of our product candidates. |
| ● |
We may not be successful in our efforts to identify or discover additional product candidates. |
| ● |
We intend to continue to rely on our BOLT proprietary product platform to develop our phage therapies. Our competitive position could be materially harmed if our competitors develop similar platforms and develop rival product candidates. | |
| ● |
Our limited operating history may make it difficult to evaluate the success of our business to date and to assess our future viability. |
| ● | We have never generated any revenue from product sales and may never be profitable or, if achieved, may not sustain profitability. | |
| ● |
Results from preclinical studies of our product candidates may not be predictive of the results of clinical trials or later stage clinical development. |
| ● | Our product candidates are subject to significant regulatory approval requirements, which could delay, prevent or limit our ability to market or develop our product candidates. |
| ● | Our relationships with healthcare providers, physicians and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and other consequences. |
iv
| ● | Even if we receive regulatory approval of any product candidates for therapeutic indications, we will be subject to ongoing regulatory compliance obligations and continued regulatory review which may result in significant additional expense. Additionally, any of our product candidates, if approved, could be subject to labeling and other restrictions and market withdrawal, and we may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our product candidates. | |
| ● |
Any products that we may develop may become subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, which could make it difficult for us to sell any product candidates or therapies profitably. | |
| ● |
Ongoing health care legislative and regulatory reform measures may have a material adverse effect on our business and results of operations. |
| ● | The license agreements we maintain, including the Yeda 2015 License Agreement (as defined below), are important to our business. If we or the other parties to our license agreements fail to adequately perform under the license agreements, or if we or they terminate the license agreements, the development, testing, manufacture, production and sale of our phage-based therapeutic product candidates would be delayed or terminated, and our business would be adversely affected. |
| ● | We are highly dependent on intellectual property licensed from third parties, and termination or limitation of any of these licenses could result in the loss of significant rights and materially harm our business. |
| ● | We are dependent on patents and proprietary technology. If we fail to adequately protect this intellectual property or if we otherwise do not have exclusivity for the marketing of our products, our ability to commercialize products could suffer. | |
| ● |
If we infringe the rights of third parties, we could be prevented from selling products, forced to pay damages and/or royalties, and forced to defend against litigation. | |
| ● |
We rely on our proprietary product platform to identify phage-based therapies. Our competitive position could be materially harmed if our competitors develop a similar platform and develop rival product candidates. | |
| ● |
We rely on trade secrets and other forms of non-patent intellectual property protection. If we are unable to protect our trade secrets, other companies may be able to compete more effectively against us. | |
| ● |
If we are sued for infringing intellectual property rights of third parties or if we are forced to engage in an interference proceeding, it will be costly and time-consuming, and an unfavorable outcome in that litigation or interference would have a material adverse effect on our business. | |
| ● |
Third-party relationships are important to our business. If we are unable to maintain our collaborations or enter into new relationships, or if these relationships are not successful, our business could be adversely affected. |
| ● | Our headquarters, research and development and other significant operations are located in Israel, and, therefore, our results may be adversely affected by political, economic and military instability in Israel, including the recent war with Hamas and other terrorist organizations from the Gaza Strip |
v
| ● |
The Israeli government grants we have received for research and development expenditures restrict our ability to manufacture products and transfer technology outside of Israel and require us to satisfy specified conditions. If we fail to satisfy these conditions, we may be required to refund grants previously received, together with interest and penalties. |
| ● | Exchange rate fluctuations between the U.S. Dollar, the New Israeli Shekel, the Euro and other foreign currencies, may negatively affect our future revenues and expenses. | |
| ● |
It may be difficult to enforce a U.S. judgment against us or our officers and directors in Israel or the United States or to assert U.S. securities laws claims in Israel or serve process on our officers and directors. | |
| ● |
Our product candidates rely on the availability of specialty raw materials, which may not be available to us on acceptable terms or at all. |
| ● | A significant number of shares of our Common Stock are subject to issuance upon exercise of outstanding warrants and options or conversion of our Convertible Preferred Stock, which upon exercise or conversion may result in dilution to our security holders. | |
| ● |
We have never paid dividends on our Common Stock, and we do not anticipate paying any cash dividends on our Common Stock in the foreseeable future. | |
| ● |
Our Public Warrants (as defined below) have been delisted, and we may be unable to maintain the listing of our securities in the future. | |
| ● |
The market price of our Common Stock and other securities may be volatile and fluctuate substantially, which could result in substantial losses for purchasers of our Common Stock. | |
| ● |
As a “smaller reporting company” we are permitted to provide less disclosure than larger public companies, which may make our Common Stock less attractive to investors. | |
| ● |
Our success depends, in part, on our ability to retain key executives and to attract, retain and motivate qualified personnel. | |
| ● |
Expectations relating to environmental, social and governance (ESG) programs may impose additional costs and expose us to new risks. | |
| ● |
Our business and operations would suffer in the event of computer system failures, cyber-attacks or deficiencies in our cyber-security. |
vi
PART I
ITEM 1. BUSINESS
Overview
We are a clinical stage product discovery company developing products using both natural and engineered phage technologies designed to target and kill specific harmful bacteria associated with chronic diseases, such as cystic fibrosis, or CF and diabetic foot osteomyelitis, or DFO. Bacteriophage or phage are bacterial, species-specific, strain-limited viruses that infect, amplify and kill the target bacteria and are considered inert to mammalian cells. By utilizing proprietary combinations of naturally occurring phage and by creating novel phage using synthetic biology, we develop phage-based therapies intended to address both large-market and orphan diseases.
Based on the urgency of treating the infection (whether acute or chronic), the susceptibility of the target bacteria to phage (e.g. the ability to identify a phage cocktail that would target a broad range of bacterial strains) and other considerations, we offer two phage-based product types:
| (1) | Fixed cocktail therapy – in this approach a single product containing a fixed number of selected phages is developed to cover a wide range of bacterial strains, thus allowing treatment of broad patient populations with the same product. Fixed cocktails are developed using our proprietary BOLT platform, in which high throughput screening, directed evolution, and bioinformatic approaches are leveraged to produce an optimal phage cocktail. | |
| (2) | Personalized therapy – in this approach a large library of phages is developed, of which single optimal phages are personally matched to treat specific patients. Matching optimal phages with patients is carried out using a proprietary phage susceptibility testing, or PST, where multiple considerations are analyzed simultaneously – allowing for an efficient screen of the phage library while maintaining short turnaround times. |
In our therapeutic programs, we focus on using phage therapy to target specific strains of pathogenic bacteria that are associated with diseases. Our phage-based product candidates are developed utilizing our BOLT proprietary research and development platform. The BOLT platform is unique, employing cutting edge methodologies and capabilities across disciplines including computational biology, microbiology, synthetic engineering of phage and their production bacterial hosts, bioanalytical assay development, manufacturing and formulation, to allow agile and efficient development of natural or engineered phage combinations, or cocktails. The cocktail contains phage with complementary features and is optimized for multiple characteristics such as broad target host range, ability to prevent resistance, biofilm penetration, stability and ease of manufacturing.
Our goal is to develop multiple products based on the ability of phage to precisely target harmful bacteria and on our ability to screen, identify and combine different phage, both naturally occurring and created using synthetic engineering, to develop these treatments.
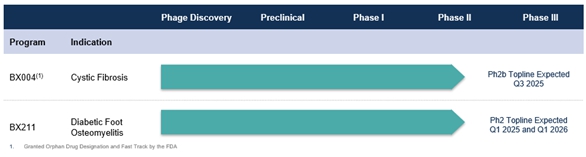
Our Product Pipeline
The chart below identifies our product candidates’ pipeline, their current status and expected timing for upcoming milestones. We do not have any products approved or available for sale, our product candidates are still in the preclinical and clinical development stages, and we have not generated any revenue from product sales.
Ongoing Programs
BX004 – Treatment of Cystic Fibrosis
BX004 is our therapeutic phage product candidate under development for chronic pulmonary infections caused by Pseudomonas aeruginosa, or P. aeruginosa, a main contributor to morbidity and mortality in patients with CF. Enhanced resistance to antibiotics develops, particularly in CF patients, due to extensive drug use consisting of prolonged and repeated broad-spectrum antibiotic courses often beginning in childhood, and leading to the appearance of multidrug-resistant strains. In preclinical in vitro studies, BX004 was shown to be active against antibiotic resistant strains of P. aeruginosa and demonstrated the ability to penetrate biofilm, an assemblage of surface-associated microbial cells enclosed in an extracellular polymeric substance and one of the leading causes for antibiotic resistance.
1
The Phase 1b/2a trial in CF patients with chronic respiratory infections caused by P. aeruginosa. was comprised of two parts. The study design was based on recommendations from the Cystic Fibrosis Therapeutic Development Network.
In February 2023, we announced positive results from Part 1 of the Phase 1b/2a trial evaluating BX004. Part 1 evaluated the safety, tolerability, pharmacokinetics, or PK, and microbiologic activity of BX004 over a 7-day ascending treatment period in nine CF patients (7 on BX004, 2 on placebo) with chronic P. aeruginosa pulmonary infection in a single ascending dose and multiple dose design.
Results from Part 1 of the Phase 1b/2a trial included the following findings: No safety events related to treatment with BX004 occurred; Mean P. aeruginosa colony forming units, or CFU, at Day 15 (compared to baseline): -1.42 log (BX004) vs. -0.28 log (placebo). This reduction was seen on top of standard of care inhaled antibiotics; Phage were detected in all patients treated with BX004 during the dosing period, including in several patients up to Day 15 (one week after end of therapy); no phage were detected in patients receiving placebo; there was no evidence of treatment-related resistance to BX004 during or after treatment , compared to placebo; and as expected due to the short duration of treatment, there was no detectable effect on % predicted forced expiratory volume in 1 second, or FEV1.
In November 2023, we announced positive topline results from Part 2 of the Phase 1b/2a trial evaluating BX004. The objectives of Part 2 of the Phase 1b/2a trial were to evaluate the safety and tolerability of BX004 in a larger number of CF patients dosed for a longer treatment duration than Part 1 of the study, with the anticipation that the longer treatment might result in greater effects than in the Part 1. In Part 2, 34 CF patients were randomized in a 2:1 ratio with 23 CF patients receiving BX004 and 11 patients receiving placebo via nebulization twice daily for 10 days.
Key results from Part 2 of the Phase 1b/2a trial included the following findings:
| ● | Study drug was safe and well-tolerated, with no related SAEs (serious adverse events) or related APEs (acute pulmonary exacerbations) to study drug. |
| ● | In the BX004 arm, 3 out of 21 (14.3%) patients with quantitative CFU at baseline converted to sputum culture negative for P. aeruginosa after 10 days of treatment (including 2 patients after 4 days) compared to 0 out of 10 (0%) in the placebo arm. |
| ● | BX004 vs. placebo showed a positive clinical effect in a predefined subgroup of patients with reduced baseline lung function (FEV1<70%). Difference between groups at Day 17: relative FEV1 improvement of 5.67% (change from baseline +1.46 vs. -4.21) and +8.87 points in Cystic Fibrosis Questionnaire-Revised (CFQR) respiratory symptom scale (change from baseline +2.52 vs. -6.35). |
| ● | In full population, BX004 vs. placebo P. aeruginosa levels were more variable in sputum, potentially driven by aligning initiation of study drug administration with the initiation of standard of care antibiotic treatment regimen. In a prespecified subgroup of patients on standard of care inhaled antibiotics on continuous regimen, BX004 vs. placebo reduced sputum P. aeruginosa levels at Day 10: difference in change from baseline between groups of -2.8 log10 CFU/g sputum (change from baseline -2.91 vs -0.11), exceeding Part 1 results. |
| ● | Alternating/cycling background antibiotic regimen likely associated with fluctuations in P. aeruginosa levels potentially confounding the ability to observe a P. aeruginosa reduction in this subgroup. |
| ● | During the study period, based on current available data, no evidence of treatment-related phage resistance was observed in patients treated with BX004 compared to placebo. |
In August 2023, the FDA granted BX004 Fast Track designation for the treatment of chronic respiratory infections caused by P. aeruginosa bacterial strains in patients with CF. In addition, in December 2023, BX004 received orphan drug designation from the FDA.
BiomX expects to initiate a randomized, double blind, placebo-controlled, multi-center Phase 2b study in CF patients with chronic P. aeruginosa pulmonary infections in the fourth quarter of 2024. The study is designed to enroll approximately 60 patients randomized at a 2:1 ratio to BX004 or placebo. Treatment is expected to be administered via inhalation twice daily for a duration of 8 weeks. The study is designed to monitor the safety and tolerability of BX004 and is designed to demonstrate improvement in microbiological reduction of P. aeruginosa burden and evaluation of effects on clinical parameters such as lung function measured by FEV1 and patient reported outcomes. Study results are expected in the third quarter 2025.
2
BX211 – Treatment of Diabetic Foot Osteomyelitis (DFO)
BX211 is a personalized phage therapy for the treatment of DFO associated with Staphylococcus aureus, or S. aureus. The personalized phage treatment tailors a specific phage selected from a proprietary phage-bank according to the specific strain of S. aureus biopsied and isolated from each patient. DFO is a bacterial infection of the bone that usually develops from an infected foot ulcer and is a leading cause of amputation in patients with diabetes. We believe that scientific literature demonstrating the potential benefit in treating osteomyelitis using phage in animal models as well as numerous successful compassionate cases using phage therapy to treat DFO patient support our approach of using phage therapy to treat DFO.
The ongoing randomized, double-blind, placebo-controlled, multi-center phase 2 study investigating the safety, tolerability, and efficacy of BX211 for subjects with DFO associated with S. aureus is expected to enroll approximately 45 subjects randomized at a 2:1 ratio to BX211 or placebo. BX211 or placebo is designed to be administered weekly, by topical and intravenous, or IV route at week 1 and by the topical route only at each of weeks 2-12. Over the 12-week treatment period, all subjects are expected to continue to be treated in accordance with standard of care which will include antibiotic treatment as appropriate. A first readout of study topline results is expected at week 13 evaluating healing of the wound associated with osteomyelitis, followed by a second readout at week 52 evaluating amputation rates and resolution of osteomyelitis based on X-ray, clinical assessments, and established biomarkers (Erythrocyte Sedimentation Rate, or ESR, and C-Reactive Protein, or CRP). These readouts are expected in the first quarter of 2025 and the first quarter of 2026, respectively.
National Institutes of Health, or NIH study in Cystic Fibrosis
We are supporting a study conducted by the NIH and The Antibacterial Resistance Leadership Group targeting P. Aeruginosa infections in CF patients under FDA emergency Investigational New Drug, or eIND allowance. The Phase 1b/2, multi-centered, randomized, double-blind, placebo-controlled trial is assessing the safety and microbiological activity of a single IV dose of bacteriophage therapy in cystic fibrosis subjects colonized with P. aeruginosa.
Programs on hold
BX005 – Treatment of Atopic Dermatitis
BX005 is our topical phage product candidate targeting Staphylococcus aureus, or S. aureus, a bacterium associated with the development and exacerbation of inflammation in atopic dermatitis. S. aureus is more abundant on the skin of atopic dermatitis patients than on the skin of healthy individuals and on lesional skin than non-lesional skin. It also increases in abundance, becoming the dominant bacteria, when patients experience flares. By reducing the load of S. aureus, BX005 is designed to shift the skin microbiome composition to its ‘pre-flare’ state and potentially provide a clinical benefit. In preclinical in vitro studies, BX005 was shown to eradicate over 90% of strains, including antibiotic resistant strains, from a panel of S. aureus strains (120 strains isolated from skin of subjects from the U.S. and Europe). On April 8, 2022, the FDA approved the Company’s IND application for BX005.
As of the date of this Annual Report, we have paused development efforts for BX005 due to prioritizing resources towards our CF and DFO programs, and we cannot provide guidance on resuming its development.
Prosthetic Joint Infections, or PJI
Our personalized phage therapy for treating PJI targets multiple bacterial organisms such as Staphylococcus aureus, Staphylococcus epidermidis and Enterococcus faecium. This treatment was granted Orphan-drug designation by the FDA in July 2020. As of the date of this Annual Report, we have paused development efforts of this program due to prioritizing resources towards our CF and DFO programs, and we cannot provide guidance on resuming its development.
3
Our Strategy
Our goal is to develop multiple products based on the ability of phage to precisely target harmful bacteria and on our ability to screen, identify and optimally combine different phage, both naturally occurring and generated using synthetic engineering, to develop these treatments. We intend to continue to:
| ● | Investigate clinical safety and efficacy of our lead phage-based product candidates to treat CF and DFO; |
| ● | Identify new pathogenic bacteria to be targeted by phage therapy for our existing indications and possible new indications; and |
| ● | Develop and partner microbiome-based biomarker tests, based on our proprietary XMarker platform, that can be used for disease diagnosis or as companion diagnostics. |
Our phage discovery platform
Our approach is driven by the convergence of several factors: a rapidly increasing understanding of phage, including the links between phage behaviors and their genomes; growing evidence that the presence of specific harmful bacteria may impact chronic diseases, such as CF, making them in principle, amenable to treatment with phage; and by a growing number of anecdotal reports from different academic centers of successful compassionate use of phage to treat seriously ill patients who were unresponsive to other therapies. We believe our phage therapeutic product candidates have the potential to treat conditions and diseases by precisely targeting pathogenic bacteria without disrupting elements of the healthy microbiota.
Our phage-based product candidates, either fixed phage cocktails or personalized phage treatments, are developed utilizing our proprietary research and development platforms, named BOLT and PST. The BOLT, platform is unique, employing cutting edge methodologies and capabilities across disciplines including computational biology, microbiology, synthetic engineering of phage and their production bacterial hosts, bioanalytical assay development, manufacturing and formulation, to allow agile and efficient development of natural or engineered phage combinations, or cocktails.
The PST platform utilizes proprietary assays to allow us to screen extensive phage libraries in search of optimal phage for treatment of the specific target bacteria isolated from a given patient.
BOLT is designed to allow the rapid development of optimized phage cocktails. These cocktails may be comprised of naturally-occurring or synthetically engineered phage. The cocktail contains phage with complementary features and is optimized for multiple characteristics such as broad target host range, ability to prevent resistance, biofilm penetration, stability and ease of manufacturing. Pre-clinical development of the optimized phage cocktail is anticipated to require 1-2 years.
We combine multiple technologies that originate from the laboratories of our scientific founders and that were developed internally. Technologies that were developed by our scientific founders are described in leading scientific journals. One of our scientific founders, Professor Rotem Sorek, a Professor in the Department of Molecular Genetics at the Weizmann Institute of Science, or WIS, is a world leader in phage genomics and bacterial defense mechanisms. Another scientific founder, Professor Eran Elinav, a Professor in the Department of Immunology at the WIS, is an expert in investigating the link between the microbiome and human health and disease. Our third scientific founder, Professor Timothy K. Lu, is a world leader in synthetic biology approaches to engineering gene circuits and phage, leading the Synthetic Biology Group in the Department of Electrical Engineering and Computer Science and the Department of Biological Engineering at the Massachusetts Institute of Technology. In addition, through the acquisition of the privately held Israel-based company, RondinX Ltd. in 2017, we gained access to high throughput genomic analyses techniques developed by Professor Eran Segal, a leading computational biologist from the Department of Computer Science and Applied Mathematics at the WIS. The combination of the technologies and expertise from these leaders in each of their respective fields is critical in enabling us to focus on treating complex human diseases and conditions by precise manipulation of the microbiome.
4
Additionally, we developed proprietary assays and screening technology for robust and high throughput testing PST. The PST platform combines state of the art automation with advanced microbiology assays. The output is a reproducible conclusive decision for optimal phage matching, based on multiple factors, including success of phage infection, suppression of resistant mutants, and antibiofilm activity.
Manufacturing
We have developed manufacturing processes that utilize state of the art industrial methods for the manufacturing of our product candidates. These processes are designed to comply with current Good Manufacturing Practice, or cGMP, with the appropriate scale to meet our clinical study needs, and to fulfill the requirements of regulators for human studies.
In February 2021, we consolidated our U.S. Good Manufacturing Practice, or GMP, manufacturing, testing and development into a 6,100 square feet space in our Gaithersburg facility and in March 2021, we moved into a new 6,500 square feet manufacturing facility in our headquarters, in Ness Ziona, Israel. Both facilities are designed to produce clinical quantities of our product candidates required for early-stage clinical development with compliance suitable for this stage of development and to support eIND.
The Ness Ziona facility consists of two suites for drug substance phage production/development as well as formulation and final drug product production rooms to support topical, oral, inhaled and injectable phage-based products in a liquid, cream, semi-solid or dry form.
The Gaithersburg facility consists of three manufacturing suites, one for upstream seed banking, one for drug substance phage production, and one for formulation and fill of the final drug product. This facility is also equipped with in-house quality control testing laboratories to support the release of injectable phage-based products in a liquid form. Additional laboratory space is allocated for process development and there are laboratory and office spaces available that can be repurposed for future GMP expansion.
We currently operate a manufacturing model that combines in-house process development, manufacturing and testing with the flexibility to outsource to third-party development, manufacturing, testing, and logistics organizations, when needed. We maintain service agreements with multiple manufacturers, testing laboratories and a third-party logistics warehouse for product candidate distribution. These service agreements are generally short-term in nature and can be extended or renewed. As such, for BX004, we have engaged a third-party to supplement our in-house process development activities. We selected this organization based on its experience, capability, capacity and regulatory status. Manufacturing and development projects are managed by a team of internal staff who assure compliance with the technical aspects and regulatory requirements of the manufacturing process.
Additional phage bank product candidates collectively known as BX211 are manufactured at our in-house GMP facility in Gaithersburg. Such product candidates are produced and released by internal staff in compliance with cGMPs. We perform release testing in house for most release assays and also outsource testing to qualified laboratories. In addition, we utilize a third-party logistics warehouse for product storage and distribution to clinical sites.
5
We are considering consolidation of the two GMP sites into one based on future needs. While we do not have a current need for a commercial scale manufacturing capacity, at the appropriate time we intend to evaluate building large scale cGMP internal manufacturing capabilities, which may include expansion of our operations.
Intellectual Property
We strive to protect the proprietary technology that we believe is important to our business, including seeking and maintaining patent protection in the United States and internationally for our product candidates and discovery platform. We also rely on trademarks, trade secrets, know-how, copyrights, continuing technological innovation and in-licensing opportunities to develop and maintain our proprietary position. For more information regarding the risks related to our intellectual property, see “Risk Factors — Risks Related to our Licensed and Co-Owned Intellectual Property.”
We plan to continue to expand our intellectual property estate by filing patent applications directed to formulations, related methods of treatment, methods of manufacture or identification from our ongoing development of our product candidates, as well as discovery based on our proprietary product platform. Our success will depend on our ability to obtain and maintain patent and other proprietary protection for commercially important technology, inventions and know-how related to our business, defend, and enforce any patents that we may obtain, preserve the confidentiality of our trade secrets and know-how and operate without infringing the valid and enforceable patents and proprietary rights of third parties.
Because patent applications in the United States and certain other jurisdictions are maintained in secrecy for 18 months or potentially even longer, and because publication of discoveries in the scientific or patent literature often lags behind actual discoveries and patent application filings, we cannot be certain of the priority of inventions covered by pending patent applications. Accordingly, we may not have been the first to invent the subject matter disclosed in some of its patent applications or the first to file patent applications covering such subject matter, and we may have to participate in interference proceedings or derivation proceedings declared by the United States Patent and Trademark Office, or USPTO, to determine priority of invention.
Patent portfolio
Our patent portfolio consists of owned patent applications, as well as both licensed and co-owned patent applications (that are also licensed). See “Risk Factors — Risks Related to our Licensed and Co-Owned Intellectual Property.” For some of these applications, prosecution has not started, and others are in the early stages of prosecution in the United States and in selected jurisdictions outside of the United States. We solely own four patent families. We co-own one US patent family with Keio University in Tokyo, Japan, or Keio, one international patent family (United States, Australia, Canada, European Patent Office national filings) with Yeda Research and Development Company Limited, the technology transfer office of the WIS, or Yeda, and one international patent family (United States, Europe) with both Keio and Yeda. We have an exclusive license from Yeda and Keio for these co-owned patent applications. We have exclusive licenses from Yeda or Keio for the rest of the patents and patent applications in its portfolio.
A significant portion of our portfolio is directed to our product candidates, specifically: CF and atopic dermatitis as well as product candidates relevant to programs which we have stopped their development such as: inflammatory bowel disease, or IBD, primary sclerosing cholangitis and colorectal cancer, or CRC, as well as to our bacterial target discovery and bacteriophage discovery technology platforms. Prosecution has yet to commence for most of the pending patent applications covering our product candidates. Prosecution is a lengthy process, during which the scope of the claims initially submitted for examination by the USPTO are often significantly narrowed by the time they issue, if they issue at all. We expect this to be the case with respect to our licensed and co-owned patent applications, described briefly below.
6
In connection with the Acquisition, we further enhanced our intellectual property portfolio with the addition of APT’s portfolio comprising of 7 issued or allowed patents, 19 patent families (including applications in United States, Europe, Australia, Canada, China, India, Japan, Korea, Israel, Brazil, and South Africa). APT’s patents and patent applications consist of patents and patent applications with respect to pharmaceutical compositions and methods of treatment, methods of manufacture of such compositions and expire between June 2037 and October 2043.
CF
We solely own one patent family (United States, Australia, Canada, European Patent Office, Japan and China) containing claims directed to pharmaceutical compositions comprising combinations of bacteriophage to treat chronic Pseudomonas lung infections, especially common in CF patients, methods of use for these bacteriophage combinations, and methods of identifying patients who will respond to these bacteriophage combinations. Any United States patents issuing from the pending application covering our lead bacteriophage combination in this program, if issued, are expected to expire in 2042. Patent term adjustments or patent term extensions could result in later expiration dates.
Atopic Dermatitis
We solely own one patent family (United States, Australia, Canada, European Patent Office and Japan) containing claims directed to pharmaceutical compositions comprising combinations of bacteriophage to treat skin infections, especially common in atopic dermatitis patients, methods of use for these bacteriophage combinations, and methods of identifying patients who will respond to these bacteriophage combinations. Any United States patents issuing from the pending application covering our lead bacteriophage combination in this program, if issued, are expected to expire in 2042. Patent term adjustments or patent term extensions could result in later expiration dates.
Patent term
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries in which we file patent applications, including the United States, the base term is 20 years from the filing date of the earliest-filed non-provisional patent application from which the patent claims priority. The term of a United States patent can be lengthened by patent term adjustment, which compensates the owner of the patent for administrative delays at the USPTO. In some cases, the term of a United States patent is shortened by a terminal disclaimer that reduces its term to that of an earlier-expiring patent. The term of a United States patent may be eligible for patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Act, to account for at least some of the time the drug is under development and regulatory review after the patent is granted. With regard to a drug for which FDA approval is the first permitted marketing of the active ingredient, the Hatch-Waxman Act allows for extension of the term of one United States patent that includes at least one claim covering the composition of matter of such an FDA-approved drug, an FDA-approved method of treatment using the drug and/or a method of manufacturing the FDA-approved drug. The extended patent term cannot exceed the shorter of five years beyond the non-extended expiration of the patent or fourteen years from the date of the FDA approval of the drug, and a patent cannot be extended more than once or for more than a single product. During the period of extension, if granted, the scope of exclusivity is limited to the approved product for approved uses. Some foreign jurisdictions, including Europe and Japan, have analogous patent term extension provisions, which allow for extension of the term of a patent that covers a drug approved by the applicable foreign regulatory agency.
In the future, if and when our product candidates receive FDA approval, we expect to apply, if appropriate, for patent term extension on patents directed to those product candidates, their methods of use and/or methods of manufacture. However, there is no guarantee that the applicable authorities, including the FDA in the United States, will agree with our assessment of whether such extensions should be granted, and if granted, the length of such extensions.
7
Trade Secrets and Know-How
In addition to patents, we rely on trade secrets and know-how to develop and maintain our competitive position. We typically rely on trade secrets to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection. We protect trade secrets and know-how by establishing confidentiality agreements and invention assignment agreements with our employees, consultants, scientific advisors, contractors and collaborators. These agreements provide that all confidential information developed or made known during the course of an individual’s or entities’ relationship with us must be kept confidential during and after the relationship. These agreements also provide that all inventions resulting from work performed for us or relating to our business and conceived or completed during the period of employment or assignment, as applicable, shall be our exclusive property. In addition, we take other appropriate precautions, such as physical and technological security measures, to guard against misappropriation of its proprietary information by third parties.
Although we take steps to protect our proprietary information and trade secrets, including through contractual means with our employees and consultants, third parties may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose our technology. Thus, we may not be able to meaningfully protect our trade secrets and benefit from the exclusive use thereof. For more information regarding the risks related to our intellectual property, see “Risk Factors — Risks Related to Our Licensed and Co-Owned Intellectual Property.”
Competition
The biotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, strong competition and an emphasis on proprietary products. While we believe that our technology, knowledge and experience provide us with competitive advantages, we face substantial competition from many different sources, including larger pharmaceutical companies with more resources. Specialty biotechnology companies, academic research institutions, governmental agencies, as well as public and private institutions are also potential sources of competitive products and technologies. We believe that the key competitive factors affecting the success of any of our product candidates will include efficacy, safety profile, time to market, cost, level of promotional activity and intellectual property protection.
We are aware of a number of biotechnology companies developing bacteriophage products to treat diseases. To our knowledge, several biotechnology companies, such as Locus Biosciences, Inc., Armata Pharmaceuticals, Inc. and SNIPR Biome, as well as academic institutions, have discovery stage or clinical programs utilizing naturally occurring phage or synthetic biology approaches. In addition, we are aware of several investigational and marketed products to treat the indications that we are targeting with our product candidates, including, but not limited to:
| ● | CF: Trikafta, Symdeco, Pulmozyme, Tobramycin, Aztreonam |
| ● | DFO: TP-102 being developed by Technophage, a phage-based product being developed by Phaxiam |
Many of our competitors, either alone or with their strategic partners, have substantially greater financial, technical and human resources than ours and significantly greater experience in the discovery and development of product candidates, obtaining FDA and other regulatory approvals of products and the commercialization of those products. Accordingly, our competitors may be more successful than us in discovering product candidates, obtaining approval for such product candidates and achieving widespread market acceptance. Our competitors’ products may be more effective, or more effectively marketed and sold, than any product we may commercialize and may render our product candidates obsolete or non-competitive before we can recover the expenses of developing and commercializing any of our product candidates. We anticipate that we will face intense and increasing competition as new drugs enter the market and advanced technologies become available.
These third parties compete with us in recruiting and retaining qualified scientific, clinical, manufacturing, sales and marketing and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our program.
8
Sales and Marketing
We intend to pursue the commercialization of our drug product candidates either by building internal sales and marketing capabilities or through collaborations with others.
In October 2021, we entered into a stock purchase agreement with a subsidiary of Maruho, a leading dermatology-focused pharmaceutical company in Japan, pursuant to which we issued to Maruho 375,000 shares of Common Stock, at a price of $8.00 per share for gross proceeds of $3 million. We also granted Maruho a right of first offer to license our BX005 product candidate for atopic dermatitis in Japan. The right of first offer will commence following the availability of results from a the Phase 1/2 study which is currently on hold.
Government Regulation
Government authorities in the United States and other countries regulate, among other things, the research, development, testing, manufacture, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, post-approval monitoring and reporting, marketing and export and import of drug and biological products. Generally, before a new drug or biologic can be studied in human clinical trials or marketed, considerable data demonstrating its quality, safety, efficacy, purity, and/or potency must be obtained, organized into a format specific for each regulatory authority, submitted for review and approved by the regulatory authority where the product is intended to be studied or marketed.
U.S. Biological Product Development Process
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or the FDCA, and its implementing regulations under the FDCA, the Public Health Service Act, or the PHSA, and their implementing regulations. Both drugs and biologics are also subject to other federal, state and local statutes and regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state and local statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with applicable U.S. requirements at any time during the product development, approval, or post-marketing process may subject an applicant to administrative or judicial sanctions. These sanctions could include, among other actions, the FDA’s refusal to approve pending applications, withdrawal of an approval or license revocation, a clinical hold, untitled or warning letters, product recalls or market withdrawals, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement and civil or criminal penalties. Any agency or judicial enforcement action could have a material adverse effect on us.
Certain of our current product candidates and future product candidates must be approved by the FDA through a Biologics License Application, or BLA, process before they may be legally marketed in the United States. The process generally involves the following:
| ● | Completion of extensive preclinical studies in accordance with applicable regulations, including studies conducted in accordance with GLP requirements, if needed; |
| ● | Submission to the FDA of an IND, which must become effective before human clinical trials may begin; |
| ● | Approval by an institutional review board, or IRB, at each clinical trial site before each trial may be initiated; |
| ● | Performance of adequate and well-controlled human clinical trials in accordance with applicable IND regulations, good clinical practice, or GCP, requirements and other clinical trial-related regulations to establish the safety, purity, potency and efficacy of the investigational product for each proposed indication; |
| ● | Submission to the FDA of a BLA; |
| ● | A determination by the FDA within 60 days of its receipt of a BLA to accept the application for review; |
| ● | Satisfactory completion of an FDA pre-approval inspection of the manufacturing facility or facilities where the biologic will be produced to assess compliance with cGMP requirements to assure that the facilities, methods and controls are adequate to preserve the biologic’s identity, strength, quality and purity; |
9
| ● | Potential FDA audit of the clinical trial sites that generated the data in support of the BLA; |
| ● | Payment of user fees for FDA review of the BLA (unless a fee waiver applies); and |
| ● | FDA review and approval of the BLA, including consideration of the views of any FDA advisory committee, prior to any commercial marketing or sale of the biologic in the United States. |
Preclinical Studies and IND
Preclinical studies include laboratory evaluation of product chemistry and formulation, as well as in vitro and animal studies to establish a rationale for therapeutic use and in some cases to assess the potential for adverse events. The conduct of preclinical studies is subject to federal regulations and requirements, including in some cases GLP regulations for safety/toxicology studies. An IND sponsor must submit the results of the preclinical tests, together with manufacturing information, analytical data, any available clinical data or literature and plans for clinical trials, among other things, to the FDA as part of an IND. An IND is a request for authorization from the FDA to administer an investigational product to humans, and, must become effective before human clinical trials may begin. Some long-term preclinical testing may continue after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time, the FDA raises concerns or questions related to one or more proposed clinical trials and places the trial on clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to commence.
Clinical Trials
Clinical trials involve the administration of the drug or biological product candidate to healthy volunteers or disease-affected patients under the supervision of qualified investigators, generally physicians not employed by, or under, the trial sponsor’s control. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical trial, dosing procedures, subject selection and exclusion criteria, and the parameters to be used to monitor subject safety and efficacy, including stopping rules that assure a clinical trial will be stopped if certain adverse events should occur. Each protocol and any amendments to the protocol must be submitted to the FDA as part of the IND. Clinical trials must be conducted and monitored in accordance with the FDA’s regulations comprising the GCP requirements, including the requirement that all research subjects provide informed consent. Further, each clinical trial must be reviewed and approved by an IRB at or servicing each institution at which the clinical trial will be conducted. An IRB is charged with protecting the welfare and rights of study participants and considers such items as whether the risks to individuals participating in the clinical trials are minimized and are reasonable in relation to anticipated benefits. The IRB also approves the form and content of the informed consent that must be signed by each clinical trial subject or his or her legal representative and must monitor the clinical trial until completed. There are also requirements governing the reporting of ongoing clinical trials and completed clinical trial results to public registries. Information about certain clinical trials, including clinical trial results, must be submitted within specific timeframes for publication on the www.clinicaltrials.gov website.
Clinical trials generally are conducted in three sequential phases, known as Phase 1, Phase 2 and Phase 3, and may overlap.
| ● | Phase 1 clinical trials generally involve a small number of healthy volunteers or disease-affected patients who are initially exposed to a single dose and then multiple doses of the product candidate. The primary purpose of these clinical trials is to assess the metabolism, pharmacologic action, side effect tolerability and safety of the product candidate. |
10
| ● | Phase 2 clinical trials generally involve studies in disease-affected patients to evaluate proof of concept and/or determine the dosing regimen(s) for subsequent investigations. At the same time, safety and sometimes further pharmacokinetic and pharmacodynamic information is collected, possible adverse effects and safety risks are identified and a preliminary evaluation of efficacy is conducted. |
| ● | Phase 3 clinical trials generally involve a large number of patients at multiple sites and are designed to provide the data necessary to demonstrate the effectiveness of the product for its intended use, its safety in use and to establish the overall benefit/risk relationship of the product and provide an adequate basis for labeling for new drugs. |
Post-approval trials, sometimes referred to as Phase 4 clinical trials, may be conducted after initial marketing approval. These trials are conducted to gain additional experience from the treatment of patients in the intended therapeutic indication. In certain instances, the FDA may mandate the performance of Phase 4 clinical trials as a condition of approval of a BLA.
Progress reports detailing the results of the clinical trials, among other information, must be submitted at least annually to the FDA and written IND safety reports must be submitted to the FDA and the investigators for serious and unexpected suspected adverse events, findings from other studies or animal or in vitro testing that suggest a significant risk for human subjects and any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator brochure.
It is possible for Phase 1, Phase 2, Phase 3 and other types of clinical trials not to be completed successfully within a specified period, if at all. The FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the biologic has been associated with unexpected serious harm to patients. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, or the Data Safety Monitoring Board. This group provides authorization for whether a trial may move forward at designated check points based on access to certain data from the trial.
Concurrent with clinical trials, companies may complete additional animal studies and also must develop additional information about the chemistry and physical characteristics of the biologic as well as finalize a process for manufacturing the product in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the product and, among other things, companies must develop methods for testing the identity, strength, quality and purity of the final product. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate that the product candidates do not undergo unacceptable deterioration over their shelf life.
11
FDA Review Process
Following completion of the clinical trials, data are analyzed to assess whether the investigational product is safe and effective for the proposed indicated use or uses, and also meets the regulatory requirements for potency and purity. The results of preclinical studies and clinical trials are then submitted to the FDA as part of a BLA, along with proposed labeling, chemistry and manufacturing information to ensure product quality and other relevant data. The BLA is a request for approval to market the biologic for one or more specified indications and must contain proof of safety, purity and potency. The application may include both negative and ambiguous results of preclinical studies and clinical trials, as well as positive findings. Data may come from company-sponsored clinical trials intended to test the safety and efficacy of a product’s use or from a number of alternative sources, including studies initiated by investigators. To support marketing approval, the data submitted must be sufficient in quality and quantity to establish the safety and efficacy in the intended indication, purity and potency of the investigational product to the satisfaction of the FDA. FDA approval of a BLA must be obtained before a biologic may be marketed in the United States. Under the Prescription Drug User Fee Act, or PDUFA, as amended, each BLA must be accompanied by a user fee. The FDA adjusts the PDUFA user fees on an annual basis. Fee waivers or reductions are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business. Additionally, no user fees are assessed on BLAs for products designated as orphan drugs, unless the product also includes a non-orphan indication.
The FDA reviews all submitted BLAs before it accepts them for filing and may request additional information rather than accept the BLA for filing. The FDA must make a decision on accepting a BLA for filing within 60 days of receipt, and such a decision could include a refusal to file by the FDA. Once the submission is accepted for filing, the FDA begins an in-depth review of the BLA. Under the goals and policies agreed to by the FDA under PDUFA, the FDA has 10 months, from the filing date, in which to complete its initial review of an original BLA and respond to the applicant, and six months from the filing date of an original BLA designated for priority review. The FDA does not always meet its PDUFA goal dates for standard and priority BLAs, and the review process is often extended by FDA requests for additional information or clarification.
Before approving a BLA, the FDA will conduct a pre-approval inspection of the manufacturing facilities for the new product to determine whether they comply with cGMP requirements. The FDA will not approve the product unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. The FDA also may audit data from clinical trials to ensure compliance with GCP requirements. Additionally, the FDA may refer applications for novel products or products which present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved and under what conditions, if any. The FDA is not bound by recommendations of an advisory committee, but it considers such recommendations when making decisions on approval. The FDA likely will reanalyze the clinical trial data, which could result in extensive discussions between the FDA and the applicant during the review process.
After the FDA evaluates a BLA, it will issue an approval letter, or a Complete Response Letter. An approval letter authorizes commercial marketing of the biologic with specific prescribing information for specific indications. A Complete Response Letter indicates that the review cycle of the application is complete and the application will not be approved in its present form. A Complete Response Letter usually describes all the specific deficiencies in the BLA identified by the FDA. The Complete Response Letter may require additional clinical data and/or other significant and time-consuming requirements related to clinical trials, preclinical studies or manufacturing. If a Complete Response Letter is issued, the applicant may either resubmit the BLA, addressing all the deficiencies identified in the letter, or withdraw the application. Even if such data and information are submitted, the FDA may decide that the BLA does not satisfy the criteria for approval. Data obtained from clinical trials are not always conclusive and the FDA may interpret data differently than the sponsor’s interpretation of the same data.
Orphan Drug Designation
Under the Orphan Drug Act of 1983, or the Orphan Drug Act, the FDA may grant orphan designation to a drug or biological product intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making the product available in the United States for this type of disease or condition will be recovered from sales of the product. Orphan drug designation for a biologic must be requested before submitting a BLA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process.
12
Orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages and user-fee waivers. If a product that has orphan designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications to market the same drug for the same indication for seven years from the date of such approval, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity by means of greater effectiveness, greater safety or providing a major contribution to patient care, or in instances of drug supply issues. Competitors, however, may receive approval of either a different product for the same indication or the same product for a different indication but that could be used off-label in the orphan indication. Orphan drug exclusivity also could block the approval of one of our products for seven years if a competitor obtains approval before we do for the same product, as defined by the FDA, for the same indication we are seeking approval, or if our product is determined to be contained within the scope of the competitor’s product for the same indication or disease. If one of our products designated as an orphan drug receives marketing approval for an indication broader than that which is designated, it may not be entitled to orphan drug exclusivity. In December 2023, BX004, received orphan drug designation from the FDA.
Expedited Development and Review Programs
The FDA has a fast-track program that is intended to expedite or facilitate the process for reviewing new drugs and biologics that meet certain criteria. Specifically, new drugs and biologics are eligible for fast-track designation if they are intended to treat a serious or life-threatening condition and preclinical or clinical data demonstrate the potential to address unmet medical needs for the condition. Fast track designation applies to the combination of the product and the specific indication for which it is being studied. Any product submitted to the FDA for marketing, including under a fast-track program, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. A product is eligible for priority review if it treats a serious or life-threatening condition and, if approved, would provide a significant improvement in safety and effectiveness compared to available therapies. The FDA will attempt to direct additional resources to the evaluation of an application for a new drug or biologic designated for priority review in an effort to facilitate the review.
A product may also be eligible for accelerated approval if it treats a serious or life-threatening condition and demonstrates an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, or IMM, that is reasonably likely to predict an effect on IMM or other clinical benefit. As a condition of approval, the FDA generally requires that a sponsor of a drug or biologic receiving accelerated approval perform adequate and well-controlled post-marketing clinical trials. Products receiving accelerated approval may be subject to expedited withdrawal procedures if such clinical trials fail to verify the predicted clinical benefit or if the sponsor fails to conduct such trials in a timely manner.
Additionally, a drug or biologic may be eligible for designation as a breakthrough therapy if the product is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over currently approved therapies on one or more clinically significant endpoints. The benefits of breakthrough therapy designation include the same benefits as fast-track designation, plus intensive guidance from the FDA to ensure an efficient drug development program.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or the time period for FDA review or approval may not be shortened. Furthermore, fast track designation, priority review, accelerated approval and breakthrough therapy designation do not change the standards for approval, but may expedite the development or approval process.
13
Pediatric Information
Under the Pediatric Research Equity Act of 2003, or PREA, a BLA or supplement to a BLA must contain data to assess the safety and efficacy of the biologic for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may grant deferrals for submission of pediatric data or full or partial waivers. A sponsor who is planning to submit a marketing application for a drug that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration must submit an initial Pediatric Study Plan, or PSP, within 60 days of an end-of-Phase 2 meeting or, if there is no such meeting, as early as practicable before the initiation of the Phase 3 or Phase 2/3 study. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including study objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. The FDA and the sponsor must reach an agreement on the PSP. A sponsor can submit amendments to an agreed-upon initial PSP at any time if changes to the pediatric plan need to be considered based on data collected from preclinical studies, early phase clinical trials and/or other clinical development programs.
Post-marketing Requirements
Following approval of a new product, the manufacturer and the approved product are subject to continuing regulation by the FDA, including, among other things, monitoring and record-keeping activities, reporting of adverse experiences, complying with promotion and advertising requirements, which include restrictions on promoting products for unapproved uses or patient populations (known as “off-label use”) and limitations on industry-sponsored scientific and educational activities. Although physicians may prescribe legally available products for off-label uses, manufacturers may not market or promote such uses. Prescription drug and biologic promotional materials must be submitted to the FDA in conjunction with their first use. Further, if there are any modifications to the biologic, including changes in indications, labeling or manufacturing processes or facilities, the applicant may be required to submit and obtain FDA approval of a new BLA or BLA supplement, which may require the development of additional data or preclinical studies and clinical trials.
The FDA may also place other conditions on approvals including the requirement for a Risk Evaluation and Mitigation Strategy, or REMS, to assure the safe use of the product. If the FDA concludes a REMS is needed, the sponsor of the BLA must submit a proposed REMS. The FDA will not approve the BLA without an approved REMS, if required. A REMS could include medication guides, physician communication plans or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of products. Newly discovered or developed safety or effectiveness data may require changes to a product’s approved labeling, including the addition of new warnings and contraindications, and also may require the implementation of other risk management measures, including a REMS or the conduct of post-marketing studies to assess a newly discovered safety issue. Product approvals may be withdrawn for non-compliance with regulatory standards or if problems occur following initial marketing.
FDA regulations require that products be manufactured in specific approved facilities and in accordance with cGMP regulations, which require, among other things, quality control and quality assurance, the maintenance of records and documentation and the obligation to investigate and correct any deviations from cGMP. Manufacturers and other entities involved in the manufacture and distribution of approved drugs or biologics are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP requirements and other laws. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain cGMP compliance. The discovery of violative conditions, including failure to conform to cGMP regulations, could result in enforcement actions, and the discovery of problems with a product after approval may result in restrictions on a product, manufacturer or holder of an approved BLA, including recall.
14
Biosimilars and Exclusivity
An abbreviated approval pathway for biological products shown to be biosimilar to, or interchangeable with, an FDA licensed reference biological product was created by the Biologics Price Competition and Innovation Act of 2009. This amendment to the PHSA, in part, attempts to minimize duplicative testing. Biosimilarity, which requires that the biological product be highly similar to the reference product notwithstanding minor differences in clinically inactive components and that there be no clinically meaningful differences between the product and the reference product in terms of safety, purity and potency, can be shown through analytical studies, animal studies and a clinical trial or trials.
Interchangeability requires that a biological product be biosimilar to the reference product and that the product can be expected to produce the same clinical results as the reference product in any given patient and, for products administered multiple times to an individual, that the product and the reference product may be alternated or switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biological product without such alternation or switch.
A reference biological product is granted 12 years of data exclusivity from the time of first licensure of the product, and the FDA will not accept an application for a biosimilar or interchangeable product based on the reference biological product until four years after the date of first licensure of the reference product. “First licensure” typically means the initial date the particular product at issue was licensed in the United States. Date of first licensure does not include the date of licensure of (and a new period of exclusivity is not available for) a biological product if the licensure is for a supplement for the biological product or for a subsequent application by the same sponsor or manufacturer of the biological product (or licensor, predecessor in interest, or other related entity) for a change (not including a modification to the structure of the biological product) that results in a new indication, route of administration, dosing schedule, dosage form, delivery system, delivery device or strength, or for a modification to the structure of the biological product that does not result in a change in safety, purity, or potency.
Pediatric exclusivity is another type of regulatory market exclusivity in the United States, available under the Best Pharmaceuticals for Children Act by way of its application to biologics through the Biologics Price Competition and Innovation Act. Pediatric exclusivity, if granted, adds six months to existing regulatory exclusivity periods, which must be in place in order for pediatric exclusivity to apply. This six-month exclusivity may be granted based on the voluntary completion of a pediatric trial in accordance with an FDA issued “Written Request” for such a trial, although FDA may issue such a Written Request at the request of the sponsor.
Companion Diagnostics
We may employ companion diagnostics to identify the most suitable phage to treat a specific patient under our personalized phage treatments and to help more accurately identify patients sensitive to our phage cocktails, during our clinical trials and potentially also in connection with the commercialization of our product candidates that we are developing or may in the future develop. Companion diagnostics can identify patients who are most likely to benefit from a particular therapeutic product; identify patients likely to be at increased risk for serious side effects as a result of treatment with a particular therapeutic product; or monitor response to treatment with a particular therapeutic product for the purpose of adjusting treatment to achieve improved safety or effectiveness. Companion diagnostics are regulated as medical devices by the FDA and, as such, require either clearance or approval prior to commercialization. The level of risk combined with available controls to mitigate risk determines whether a companion diagnostic device requires Premarket Approval Application approval or is cleared through the 510(k) premarket notification process. For a novel therapeutic product for which a companion diagnostic device is essential for the safe and effective use of the product, the companion diagnostic device should be developed and approved or 510(k)-cleared contemporaneously with the therapeutic. The use of the companion diagnostic device will be stipulated in the labeling of the therapeutic product.
15
Government Regulation Outside of the United States
In addition to regulations in the United States, we will be subject to a variety of regulations in other jurisdictions governing, among other things, clinical trials of drug products as well as the approval, manufacture and distribution of our product candidates. Because biologically sourced raw materials are subject to unique contamination risks, their use may be restricted in some countries. Whether or not we obtain FDA approval for a product candidate, we must obtain the requisite approvals from regulatory authorities in foreign countries prior to the commencement of clinical trials or marketing of the product in those countries. If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Clinical Trials
Certain countries outside of the United States have a regulatory process similar to the U.S process that requires the submission of a clinical trial application much like the IND prior to the commencement of human clinical trials. In the European Union, for example, a clinical trial application, or CTA, must be submitted for each clinical trial to the relevant national health authority and an independent ethics committee in each country in which the trial is to be conducted through a single EU portal for harmonized assessment, much like the FDA and an IRB, respectively. CTAs must be accompanied by an investigational medicinal product dossier with supporting information prescribed by the Clinical Trials Directive (and corresponding national laws of the member states) and further detailed in applicable guidance documents. Once the CTA is approved in accordance with a country’s requirements, the clinical trial may proceed. A similar process to the one described for the European Union is required in Israel for initiation of clinical trials. The requirements and process governing the conduct of clinical trials vary from country to country. In all cases, the clinical trials must be conducted in accordance with GCP and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
Approval Process
In order to market our products, we must obtain a marketing approval for each product and comply with numerous and varying regulatory requirements. The approval procedure varies among countries and can involve additional testing in comparison to the testing carried out for the U.S. approval. The time required to obtain approval in foreign countries may differ substantially from that required to obtain FDA approval. Clinical trials conducted in one country may not be accepted by regulatory authorities in other countries. The regulatory approval process outside the United States generally is subject to all of the same risks associated with obtaining FDA approval. In addition, in many countries outside the United States, it is required that the product be approved for reimbursement before the product can be approved for sale in that country.
To obtain marketing approval of a medicinal product under the European Union regulatory system, an applicant must submit a marketing authorization application, or MAA, under either a centralized or a decentralized procedure. The decentralized procedure is based on a collaboration among the member states selected by the applicant. In essence, the applicant chooses a ‘lead’ member state that will carry out the scientific assessment of the MAA and review the product information. The other member states must recognize the outcome of such assessment and review except in case of a “serious potential risk to public health.” The decentralized procedure results in the grant of a national marketing authorization in each selected country. That procedure is available for all medicinal products unless they fall into the mandatory scope of the centralized procedure. In practice, it is used for OTC, not highly innovative products, generic products and, increasingly, for biosimilars.
The centralized procedure provides for the grant of a single marketing authorization by the European Commission that is valid for all European Union member states. The centralized procedure is compulsory for certain medicinal products, including for medicinal products produced by certain biotechnological processes, products designated as orphan medicinal products, advanced therapy medicinal products, or ATMPs, and products with a new active substance and indicated for the treatment of certain diseases. For products with a new active substance and indicated for the treatment of other diseases, products that are highly innovative or for which a centralized process is in the interest of patients, the centralized procedure is optional.
16
Under the centralized procedure, the Committee for Medicinal Products for Human Use, or CHMP, the main scientific committee established at the European Medicines Agency, or EMA, is responsible for conducting the scientific assessment of the future medicinal product. The CHMP is also responsible for several post-authorization and maintenance activities, such as the assessment of modifications or extensions to an existing marketing authorization. The maximum timeframe for the evaluation of an MAA is 210 days, excluding clock stops. The European Commission grants or refuses the marketing authorization, following a procedure that involves representatives of the member states. The European Commission’s decision is in accordance with the CHMP scientific assessment except in very rare cases.
Pursuant to Regulation (EC) 1394/2007, specific rules apply to ATMPs, a category that is comprised of gene therapy medical products, somatic cell therapy medicinal products, and tissue-engineered medicinal products. Those rules have triggered the adoption of guidelines on manufacturing, clinical trials and pharmacovigilance that adapt the general regulatory requirements to the specific characteristics of ATMPs. Regulation (EC) 1394/2007 introduced a “hospital exemption”, which authorizes hospitals to develop ATMP for their internal use without having obtained a marketing authorization and to complying with European Union pharmaceutical law. The hospital exemption, which is in essence a compounded ATMP, has been transposed in all Member States, sometimes in such a way that the ATMPs under the hospital exemption are competitive alternatives to ATMPs with marketing authorization. The broad use of the hospital exemption by national hospitals led the European Commission to discuss with the Member States a more reasonable application of the hospital exemption that would not undermine the common legal regime for ATMP.
Marketing authorization is valid for five years in principle and the marketing authorization may be renewed after five years on the basis of a re-evaluation of the risk-benefit balance by the EMA or the competent authority of the authorizing member state. To this end, the marketing authorization holder must provide the EMA or the competent authority with a consolidated version of the file in respect of quality, safety and efficacy, including all variations introduced since the marketing authorization was granted, at least six months before the marketing authorization ceases to be valid. Once renewed, the marketing authorization is valid for an unlimited period, unless the European Commission or the national competent authority decides, on justified grounds relating to pharmacovigilance, to proceed with one additional renewal. Any authorization which is not followed by the actual placing of the medicinal product on the European Union market (in case of centralized procedure) or on the market of the authorizing member state within three years after authorization ceases to be valid (the so-called sunset clause).
Orphan Designation
Countries other than the United States have adopted a specific legal regime to support the development and marketing of drugs and biologics for rare diseases.
For example, in the European Union, Regulation 141/2000 organizes the grant of orphan drug designations to promote the development of products that are intended for the diagnosis, prevention or treatment of life threatening or chronically debilitating conditions affecting not more than five in 10,000 persons in the European Economic Area (the European Union, plus Iceland, Liechtenstein and Norway), or EEA, (or where it is unlikely that the development of the medicine would generate sufficient return to justify the investment) and for which no satisfactory method of diagnosis, prevention or treatment has been authorized or, if a method exists, the product would be of significant benefit to those affected. The EMA’s Committee for Orphan Medicinal Products, or COMP, examines if the orphan criteria are met and gives opinions thereon, and the orphan status is granted by the European Commission. The meeting of the criteria for orphan designation is examined again by the COMP at the time of approval of the medicinal product, which typically occurs several years after the grant of the orphan designation. If the criteria for orphan designation are no longer met at that time, the European Commission withdraws the orphan status.
In the European Union, orphan drug designation entitles the sponsor to financial incentives such as reduction of fees or fee waivers and to ten years of market exclusivity granted following medicinal product approval. Market exclusivity precludes the EMA or a national regulatory authority from validating another MAA, and the European Commission or a national regulatory authority from granting another marketing authorization, for a same or similar medicinal product and a same therapeutic indication, for that time period. This 10-year period may be reduced to six years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity. The orphan exclusivity may be lost vis-à-vis another medicinal product in cases the manufacturer is unable to assure sufficient quantity of the medicinal product to meet patient needs or if that other product is proved to be clinically superior to the approved orphan product. A drug is clinically superior if it is safer, more effective or makes a major contribution to patient care. Orphan drug designation must be requested before submitting a MAA. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process, and it does not afford any regulatory exclusivity until a marketing authorization is granted.
17
Expedited Development and Approval
Mechanisms are in place in many jurisdictions that allow an earlier approval of the drug so that it reaches patients with unmet medical needs earlier. The European Union, for example, has instituted several expedited approval mechanisms including two mechanisms that are specific to the centralized procedure:
| ● | the accelerated approval: the EMA may reduce the maximum timeframe for the evaluation of an MAA from 210 days to 150 days when the future medicinal product is of major interest from the point of view of public health, in particular from the viewpoint of therapeutic innovation. |
| ● | the conditional marketing authorization: as part of its marketing authorization process, the European Commission may grant marketing authorizations on the basis of less complete data than is normally required. |
A conditional marketing authorization may be granted when the CHMP finds that, although comprehensive clinical data referring to the safety and efficacy of the medicinal product have not been supplied, all the following requirements are met:
| ● | the risk/benefit balance of the medicinal product is positive; |
| ● | it is likely that the applicant will be in a position to provide the comprehensive clinical data; |
| ● | unmet medical needs will be addressed; and |
| ● | the benefit to public health of the immediate availability on the market of the medicinal product concerned outweighs the risk inherent in the fact that additional data is still required. |
The granting of a conditional marketing authorization is typically restricted to situations in which only the clinical part of the application is not yet fully complete. Incomplete preclinical or quality data may however be accepted if duly justified and only in the case of a product intended to be used in emergency situations in response to public health threats.
Conditional marketing authorizations are valid for one year, on a renewable basis. The conditions to which approval is subject will typically require the holder to complete ongoing trials or to conduct new trials with a view to confirming that the benefit-risk balance is positive and to collect pharmacovigilance data. Once the conditions to which the marketing authorization is subject are fulfilled, the conditional marketing authorization is transformed into a regular marketing authorization. If, however, the conditions are not fulfilled with the timeframe set by EMA, the conditional marketing authorization ceases to be renewed.
The EMA has also implemented the so-called “PRIME” (PRIority MEdicines) status in order support the development and accelerate the approval of complex innovative medicinal products addressing an unmet medical need. PRIME status enables early dialogue with the relevant EMA scientific committees and, possibly, some payors and thus reinforces the EMA’s scientific and regulatory support. It also opens accelerated assessment of the MAA as PRIME status, is normally reserved for medicinal products that may benefit from accelerated assessment, i.e., medicines of major interest from a public health perspective, in particular from a therapeutic innovation perspective.
Finally, all medicinal products (i.e. decentralized and centralized procedures) may benefit from an MA “under exceptional circumstances.” This marketing authorization is close to the conditional marketing authorization as it is reserved to medicinal products to be approved for severe diseases or unmet medical needs and the applicant does not hold the complete data set legally required for the grant of a marketing authorization. However, unlike the conditional marketing authorization, the applicant does not have to provide the missing data and will never have to. The risk-benefit of the medicinal product is reviewed annually. As a result, although the MA “under exceptional circumstances” is granted definitively, the risk-benefit balance of the medicinal product is reviewed annually and the marketing authorization is withdrawn in case the risk-benefit ratio is no longer favorable.
18
Pediatrics
Mandatory testing in the pediatric population is required in more and more jurisdictions. The European Union has enacted a complex and very stringent system that has inspired other jurisdictions, including the United States and Switzerland. Any application for approval of (i) a medicinal product containing a new active substance or (ii) a new therapeutic indication, pharmaceutical form or route of administration of an already authorized medicinal product which contains an active substance still protected by a supplementary protection certificate, or SPC, or a patent that qualifies for an SPC, must include pediatric data. Otherwise, the application is not validated by the competent regulatory authority. The submission of pediatric data is mandatory in those cases, even if the application concerns an adult use. Submission of pediatric data is not required or fully required if the EMA granted, respectively, a full or partial waiver to pediatric development. Moreover, that submission can be postponed if the EMA grants a deferral in order not to delay the submission of the MAA for the adult population.
The pediatric data are generated through the implementation of a pediatric investigation plan, or PIP, that is proposed by the company after completion of the PK studies in adults and agreed upon by the EMA, typically after some modifications. The PIP lists all the studies to conduct and measures to take in order to prove the safety and efficacy of the future medicinal product when used in children. The EMA may agree to modify the PIP at the company’s request. The scope of the PIP is the adult therapeutic indication or the condition of which the adult application is part or even the mechanism of action of the active substance, at the EMA’s quasi-discretion. This very broad discretion enables the EMA to require companies to develop children indications that are different from the adult indications.
Completion of a PIP renders the company eligible for a pediatric reward, which can be six-month extension of the term of the SPC or, in the cases of orphan medicinal products, two additional years of market exclusivity. The reward is subject, among other conditions, to the PIP being fully completed, to the pediatric medicinal product being approved in all the member states, and to the results of the pediatric studies being mentioned, in one way or another (for example, the approval of a pediatric indication), in the summary of product characteristics of the product.
Post-Marketing Requirements
Many countries impose post-marketing requirements similar to those imposed in the United States, in particular safety monitoring or pharmacovigilance. In the European Union, pharmacovigilance data are the basis for the competent regulatory authorities imposing the conduct of post-approval safety or efficacy study, including on off-label use. Non-compliance with those requirements can result in significant financial penalties as well as the suspension or withdrawal of the marketing authorization.
Supplementary Protection Certificate and Regulatory Exclusivities
In some countries other than the United States, some of our patents may be eligible for limited patent term extension, depending upon the timing, duration and specifics of the regulatory approval of our product candidates and any future product candidates. Furthermore, authorized drugs and biologics may benefit from regulatory exclusivities (in additional to patent protection resulting from patents).
In the European Union, Regulation (EC) 469/2009 institutes SPCs. An SPC is an extension of the term of a patent that compensates for the patent protection lost because of the legal requirements to conduct safety and efficacy tests and to obtain a marketing authorization before placing a medicinal product on the market. An SPC may be applied for any active substance that is protected by a “basic patent” (a patent chosen by the patent holder, which can be a product, process or application patent) and has not been placed on the market as a medicinal product before having obtained a marketing authorization in accordance with European Union pharmaceutical law. The term of the SPC is maximum five years, and the combined patent and SPC protection may not exceed fifteen years from the date of the first marketing authorization in the EEA. SPC rights are restricted by both the basic patent and the marketing authorization, i.e., the SPC grants the same rights as those conferred by the basic patent but limited to the active substance covered by the marketing authorization (and any use as medicinal product approved afterwards).
19
While SPC are regulated at the European level, they are granted by the national patent offices. The grant of an SPC requires a basic patent granted by the national patent office and a marketing authorization, which is the first marketing authorization for the active substance as a medicinal product in the country. Furthermore, no SPC must have already been granted to the active substance, and the application for the SPC must be filed with the national patent office within six months of the first marketing authorization in the EEA or the grant of the basic patent, whichever is the latest.
In the future, we may apply for an SPC for one or more of our currently owned or licensed European patents to add patent life beyond their current expiration date, depending on the expected length of the clinical trials and other factors involved in the filing of the relevant MAA.
Furthermore, in the European Union, medicinal products may benefit from the following regulatory exclusivities: data exclusivity, market protection, market exclusivity, and pediatric reward.
A medicinal product that contains a new active substance (reference medicinal product) is granted eight years of data exclusivity followed by two years of market protection. Data exclusivity prevents other companies from referring to the non-clinical and clinical data in marketing authorization dossier of the reference medicinal product for submission of generic MAA purposes, and market protection prevents other companies from placing generics on the market. Pursuant to the concept of global marketing authorization, any further development of that medicinal product (e.g., new indication, new form, change to the active substance) by the marketing authorization holder does not trigger any new or additional protection. The authorization of any new development is considered as “falling” into the initial marketing authorization with regard to regulatory protection; hence, the new development only benefits from the regulatory protection that remains when it is authorized. The only exception is a new therapeutic indication that is considered as bringing a significant clinical benefit in comparison to the existing therapies. Such new indication will add one-year of market protection to the global marketing authorization, provided that it is authorized within the first eight years of authorization (i.e., during the data exclusivity period). Moreover, a new therapeutic indication of a “well-established substance” benefits from one-year data exclusivity but limited to the non-clinical and clinical data supporting the new indication. Any active substance approved for at least ten years in the EEA qualifies as well-established substance.
Biosimilars may be approved through an abbreviated approval pathway after the expiration of the eight-year data exclusivity period and may be marketed after the 10 or 11-year market protection period. The approval of biosimilars requires the applicant to demonstrate similarity between the biosimilar and the biological medicinal product and to submit the non-clinical and clinical data defined by the EMA. The biosimilar legal regime has been mainly developed through EMA’s scientific guidelines applicable to categories of biological active substances. Unlike in the United States, interchangeability is regulated by each member state.
Market exclusivity is a regulatory protection exclusively afforded to medicinal products with an orphan status. Market exclusivity precludes the EMA or a national regulatory authority from validating another MAA, and the European Commission or a national regulatory authority from granting another marketing authorization, for a same or similar medicinal product and a same therapeutic indication, for a period of ten years from approval (see above).
Pediatric reward is another regulatory exclusivity. Completion of a PIP renders the company eligible for a pediatric reward, which can be six-month extension of the term of the SPC or, in the cases of orphan medicinal products, two additional years of market exclusivity (see above). In case a PIP is completed on a voluntary basis, i.e., for an approved medicinal product that is not or no longer protected by an SPC or a basic patent, the pediatric reward takes the form of a “pediatric use marketing authorization”, or PUMA. That special authorization does not fall into the global marketing authorization and thus benefits from eight years of data exclusivity followed by two or three years of market protection.
20
Other U.S. Healthcare Laws and Compliance Requirements
In addition to FDA restrictions on the marketing of pharmaceutical products, we may be subject to various federal and state laws targeting fraud and abuse in the healthcare industry. These laws may impact, among other things, our business or financial arrangements and relationships through which we market, sell and distribute the products, if any, for which we obtain approval. The laws that may affect our ability to operate include:
| ● | the federal Anti-Kickback Statute, which prohibits, among other things, knowingly and willfully soliciting, receiving, offering or paying any remuneration (including any kickback, bribe, or rebate), directly or indirectly, overtly or covertly, in cash or in kind, to induce, or in return for, either the referral of an individual, or the purchase, lease, order or recommendation of any good, facility, item or service for which payment may be made, in whole or in part, under a federal healthcare program, such as the Medicare and Medicaid programs; a person or entity does not need to have actual knowledge of the federal Anti-Kickback Statute or specific intent to violate it to have committed a violation. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act, or FCA, or federal civil money penalties statute; |
| ● | federal civil and criminal false claims laws and civil monetary penalties laws, such as the FCA, which impose criminal and civil penalties and authorize civil whistleblower or qui tam actions, against individuals or entities for, among other things: knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent; making, using or causing to be made or used, a false statement or record material to a false or fraudulent claim or obligation to pay or transmit money or property to the federal government; or knowingly concealing or knowingly and improperly avoiding or decreasing an obligation to pay money to the federal government; |
| ● | the civil monetary penalties law, which prohibits, among other things, the offering or giving of remuneration, which includes, without limitation, any transfer of items or services for free or for less than fair market value (with limited exceptions), to a Medicare or Medicaid beneficiary that the person knows or should know is likely to influence the beneficiary’s selection of a particular supplier of items or services reimbursable by a federal or state governmental program; |
| ● | the Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters; similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation; |
| ● | the federal transparency requirements under the Affordable Care Act, or ACA, including the provision commonly referred to as the Physician Payments Sunshine Act, which requires manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program to report annually to the U.S. Department of Health and Human Services information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician practitioners (physician assistants, nurse practitioners, clinical nurse specialists, anesthesiologist assistants, certified registered nurse anesthetists and certified nurse midwives) and teaching hospitals, as well as ownership and investment interests held by the physicians described above and their immediate family members; |
| ● | federal government price reporting laws, which require us to calculate and report complex pricing metrics in an accurate and timely manner to government programs; and |
| ● | federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers. |
21
Additionally, we are subject to state and foreign equivalents of each of the healthcare laws described above, among others, some of which may be broader in scope and may apply regardless of the payor. Many U.S. states have adopted laws similar to the federal Anti-Kickback Statute, some of which apply to the referral of patients for healthcare services reimbursed by any source, not just governmental payors, including private insurers. In addition, some states have passed laws that require pharmaceutical companies to comply with the April 2003 Office of Inspector General Compliance Program Guidance for Pharmaceutical Manufacturers and/or the Pharmaceutical Research and Manufacturers of America’s Code on Interactions with Healthcare Professionals. Several states also impose other marketing restrictions or require pharmaceutical companies to make marketing or price disclosures to the state. There are ambiguities as to what is required to comply with these state requirements and if we fail to comply with an applicable state law requirement we could be subject to penalties. Finally, there are state and foreign laws governing the privacy and security of health information, many of which differ from each other in significant ways and often are not pre-empted by HIPAA, thus complicating compliance efforts.
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws.
Violations of fraud and abuse laws may be punishable by criminal and/or civil sanctions, including penalties, fines, imprisonment and/or exclusion or suspension from federal and state healthcare programs such as Medicare and Medicaid and debarment from contracting with the U.S. government. In addition, private individuals have the ability to bring actions on behalf of the U.S. government under the federal FCA, as well as under the false claims laws of several states.
Law enforcement authorities are increasingly focused on enforcing fraud and abuse laws, and it is possible that some of our practices may be challenged under these laws. Efforts to ensure that our current and future business arrangements with third parties, and our business generally, will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices, including our arrangements with physicians and other healthcare providers, some of whom receive stock options as compensation for services provided, may not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of civil, criminal and administrative penalties, damages, disgorgement, monetary fines, imprisonment, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations. In addition, the approval and commercialization of any of our product candidates outside the United States will also likely subject us to foreign equivalents of the healthcare laws mentioned above, among other foreign laws.
If any of the physicians or other healthcare providers or entities with whom we expect to do business are found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs, which may also adversely affect our business.
Much like the federal Anti-Kickback Statute in the United States, the provision of benefits or advantages to physicians to induce or encourage the prescription, recommendation, endorsement, purchase, supply, order or use of medicinal products is also prohibited in the European Union. The provision of benefits or advantages to physicians is mainly governed by the national anti-bribery laws of the member states, such as the UK Bribery Act 2010, or national anti-kickback provisions (France, Belgium, etc.). Infringement of these laws could result in substantial fines and imprisonment. In certain member states, payments made to physicians must be publicly disclosed. Moreover, agreements with physicians often must be the subject of prior notification and approval by the physician’s employer, his or her competent professional organization and/or the regulatory authorities of the individual member states. These requirements are provided in the national laws, industry codes or professional codes of conduct, applicable in the member states. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines or imprisonment.
22
Additional Regulation
In addition to the foregoing, state and federal laws regarding environmental protection and hazardous substances, including the Occupational Safety and Health Act, the Resource Conservancy and Recovery Act and the Toxic Substances Control Act, affect our business. These and other laws govern our use, handling and disposal of various biological, chemical and radioactive substances used in, and wastes generated by, our operations. If our operations result in contamination of the environment or expose individuals to hazardous substances, we could be liable for damages and governmental fines. We believe that we are in material compliance with applicable environmental laws and that continued compliance therewith will not have a material adverse effect on our business. We cannot predict, however, how changes in these laws may affect our future operations.
U.S. Foreign Corrupt Practices Act
The U.S. Foreign Corrupt Practices Act, to which we are subject, prohibits corporations and individuals from engaging in certain activities to obtain or retain business or to influence a person working in an official capacity. It is illegal to pay, offer to pay or authorize the payment of anything of value to any foreign government official, government staff member, political party or political candidate in an attempt to obtain or retain business or to otherwise influence a person working in an official capacity. Similar rules apply to many other countries worldwide such as France (“Loi Sapin”) or the United Kingdom (UK Bribery Act).
U.S. Healthcare Reform
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medical products. For example, in March 2010, the ACA was enacted, which, among other things, increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program; introduced a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care plans; imposed mandatory discounts for certain Medicare Part D beneficiaries as a condition for manufacturers’ outpatient drugs coverage under Medicare Part D; subjected drug manufacturers to new annual fees based on pharmaceutical companies’ share of sales to federal healthcare programs; created a new Patient Centered Outcomes Research Institute to oversee, identify priorities in and conduct comparative clinical effectiveness research, along with funding for such research; and established the Center for Medicare & Medicaid Innovation at the CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending.
Since its enactment, there have been a number of significant changes to the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA without specifically ruling on the constitutionality of the ACA. Prior to the U.S. Supreme Court’s decision, President Biden issued an executive order initiating a special enrollment period from February 15, 2021 through August 15, 2021 for purposes of obtaining health insurance coverage through the ACA marketplace. The executive order also instructed certain governmental agencies to review and reconsider their existing policies and rules that limit access to healthcare. More recently, on March 11, 2021, President Biden signed the American Rescue Plan Act of 2021 into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug’s average manufacturer price, beginning January 1, 2024.
23
In addition, the Budget Control Act of 2011 and the Bipartisan Budget Act of 2015 led to aggregate reductions of Medicare payments to providers of 2% per fiscal year that will remain in effect through 2030, unless additional Congressional action is taken. Further, on January 2, 2013, the American Taxpayer Relief Act was signed into law, which, among other things, reduced Medicare payments to several types of providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. More recently, there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which have resulted in several recent Congressional inquiries and proposed bills designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for pharmaceutical products. In August 2022, the Inflation Reduction Act authorized Medicare to negotiate drug prices for certain high expenditure, single source Medicare part B or D drugs. Individual states in the United States have also become increasingly active in passing legislation and implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing.
We expect that additional foreign, federal and state healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in limited coverage and reimbursement and reduced demand for our products, once approved, or additional pricing pressures.
Coverage and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any products for which we obtain regulatory approval. In the United States, cosmetics are not generally eligible for coverage and reimbursement and thus any products that are marketed as cosmetics will not be covered or reimbursed. In the United States and markets in other countries, sales of any products for which we receive regulatory approval for commercial sale will depend, in part, on the availability of coverage and reimbursement from third-party payors. Third-party payors include government authorities, managed care providers, private health insurers and other organizations. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the reimbursement rate that the payor will pay for the product. Third-party payors may limit coverage to specific products on an approved list, or formulary, which might not include all of the FDA-approved products for a particular indication. A decision by a third-party payor not to cover our products could reduce physician utilization of our products once approved and have a material adverse effect on our sales, results of operations and financial condition. Moreover, a payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
In addition, coverage and reimbursement for products can differ significantly from payor to payor. One third-party payor’s decision to cover a particular medical product or service does not ensure that other payors will also provide coverage for the medical product or service, or will provide coverage at an adequate reimbursement rate.
As a result, the coverage determination process will require us to provide scientific and clinical support for the use of our products to each payor separately and will be a time-consuming process.
Third-party payors are increasingly challenging the price and examining the medical necessity and cost-effectiveness of medical products and services, in addition to their safety and efficacy. In order to obtain and maintain coverage and reimbursement for any product, we may need to conduct expensive clinical trials in order to demonstrate the medical necessity and cost-effectiveness of such product, in addition to the costs required to obtain regulatory approvals. If third-party payors do not consider a product to be cost-effective compared to other available therapies, they may not cover the product as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow a company to sell its products at a profit.
24
Outside of the United States, the pricing of pharmaceutical products is subject to governmental control in many countries. For example, in the European Union, pricing and reimbursement schemes vary widely from member state to member state. Some countries provide that products may be marketed only after a reimbursement price has been agreed. Some countries may require the completion of additional studies that compare the cost-effectiveness of a particular therapy to currently available therapies or so-called health technology assessments, in order to obtain reimbursement or pricing approval. Other countries may allow companies to fix their own prices for products, but monitor and control product volumes and issue guidance to physicians to limit prescriptions. Efforts to control prices and utilization of pharmaceutical products and medical devices will likely continue as countries attempt to manage healthcare expenditures.
Data Privacy and Security Laws
Numerous state, federal and foreign laws, including consumer protection laws and regulations, govern the collection, dissemination, use, access to, confidentiality and security of personal information, including health-related information. In the United States, numerous federal and state laws and regulations, including data breach notification laws, health information privacy and security laws, including HIPAA, and federal and state consumer protection laws and regulations (e.g., Section 5 of the FTC Act), that govern the collection, use, disclosure, and protection of health-related and other personal information could apply to our operations or the operations of our partners. In addition, certain state and non-U.S. laws, such as the California Consumer Protection Act, the California Privacy Rights Act, and the General Data Protection Regulation, or GDPR, govern the privacy and security of personal information, including health-related information in certain circumstances, some of which are more stringent than HIPAA and many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. Failure to comply with these laws, where applicable, can result in the imposition of significant civil and/or criminal penalties and private litigation. Privacy and security laws, regulations, and other obligations are constantly evolving, may conflict with each other to complicate compliance efforts, and can result in investigations, proceedings, or actions that lead to significant civil and/or criminal penalties and restrictions on data processing.
Material Agreements
License Agreements
License Agreement with Yeda
On June 22, 2015, BiomX Ltd. entered into a Research and License Agreement, with Yeda , or, as amended, the Yeda 2015 License Agreement, pursuant to which BiomX Ltd. received an exclusive worldwide license to certain know-how and research information related to the development, testing, manufacturing, production and sale of microbiome-based therapeutic product candidates, including candidates specified in the agreement, which are used in our phage discovery platform, as well as patents, research and other rights to phage product candidates resulting from the work of the consultants identified in the agreement and further research conducted at the WIS which BiomX Ltd. funded.
In connection with this license, BiomX Ltd. agreed to pay a non-refundable license fee of $10,000 per year. In addition, BiomX Ltd. contributed an aggregate of approximately $2.0 million to the research budget agreed upon in the Yeda 2015 License Agreement. BiomX Ltd is also required to pay tiered royalties in the low single digits on net sales of products and diagnostic kits covered by the Yeda 2015 License Agreement, subject to reductions as described therein. The products and diagnostic kits covered by the license agreement include those directed to CF and any other indication that may be treated by phage-based therapies, as well as related technology platforms. If BiomX Ltd. sublicenses its rights under the Yeda 2015 License Agreement, BiomX Ltd. will be obligated to pay Yeda additional sublicense royalties expressed as a percentage of the sublicensing receipts described in the agreement received ranging from the mid-teens to the mid-twenties. BiomX Ltd. is obligated to pay filing and maintenance expenses in respect of patents licensed under the Yeda 2015 License Agreement. In connection with the Yeda 2015 License Agreement, BiomX Ltd. also issued certain ordinary shares which were subsequently converted to 193,406 shares of our Common Stock as part of the Business Combination. In the event of certain mergers and acquisitions we are party to, we are obligated to pay Yeda an amount equivalent to approximately 1% of the consideration received under such transaction.
Unless terminated earlier by either party, the license granted will remain in effect in each country and for each product developed based on the license until the later of the expiration of the last licensed patent (which is expected to be in 2039) in such country for such product, and eleven years from the date of first commercial sale of such product in such country for such product. The Yeda 2015 License Agreement terminates upon the later of the expiration of the last of the patents covered under the agreement, and the expiry of a continuous 15-year period during which there has not been a first commercial sale of any product in any country. Yeda may also terminate the agreement if BiomX Ltd. fails to observe certain diligence and development requirements and milestones as described in the Yeda 2015 License Agreement. BiomX Ltd. or Yeda may terminate the Yeda 2015 License Agreement for the material uncured breach of the other party after a notice period, or the other party’s winding up, bankruptcy, insolvency, dissolution or other similar discontinuation of business. Upon termination of the Yeda 2015 License Agreement, other than due to the passage of time, BiomX Ltd. is required to grant to Yeda a non-exclusive, irrevocable, perpetual, fully paid-up, sublicensable, worldwide license in respect of our rights in know-how and research results as described in the Yeda 2015 License Agreement, provided that if Yeda subsequently grants a license to a third party that utilizes our rights, BiomX Ltd. is entitled to share in the net proceeds actually received by Yeda arising out of that license, subject to a cap based on the development expenses that BiomX incurs in connection with the Yeda 2015 License Agreement.
25
BiomX Ltd. consults with Yeda with respect to patent prosecution and maintenance decisions. Yeda is primarily responsible for prosecution and maintenance with respect to Licensed Information (as defined in the license) and we are responsible for prosecution and maintenance with respect to Subsequent Results (as defined in the license). BiomX Ltd. and Yeda are both entitled to consultation rights. BiomX is responsible for costs associated with prosecution and maintenance of all patents and applications.
BiomX Ltd. is entitled to enforce the patent rights under the license upon approval by Yeda. Yeda may elect to join the lawsuit, but we are responsible for all litigation-related expenses. Yeda reserves the right to bring its own actions if we do not notify Yeda of our intent to enforce a right or bring an action after we initially notified Yeda of the potential action.
Exclusive License with United States Navy
On March 16, 2017, APT entered into an exclusive license (as amended on January 10, 2019, or the USN License Agreement, with the United States of America, as represented by the Secretary of the Navy or the USN, pursuant to which APT received an exclusive license throughout the territory encompassing the United States, Canada and Europe to an invention entitled “Bacteriophage Compositions and Method of Selection of Components Against Specific Bacteria or the USN Licensed Patent, as well as associated materials, including approximately 350 phage (or collectively with the USN Licensed Patent, the USN Materials), in the field of treating and/or eliminating multi-drug resistant bacteria for all uses, including industrial or medical uses. Pursuant to the USN License Agreement, APT agreed to carry out a commercial development plan or the Commercial Development Plan, for the development and marketing of an invention claimed or disclosed in the USN Licensed Patent or a Licensed Invention, to bring a Licensed Invention to practical application consistent with the milestones provided in the Commercial Development Plan by December 31, 2022, and, thereafter, to continue to make the benefits of a Licensed Invention reasonably accessible to the public for the remainder of the term of the USN License Agreement. For the term of the license, any Licensed Invention or product produced through the use of a Licensed Invention for use or sale in the United States must be manufactured substantially in the United States. The Company uses the phage provided in connection with the USN License Agreement as a potential source of phage for the development of its phage treatments.
In connection with the USN License Agreement, APT paid the USN a license execution royalty of $5,000. We are also required to pay royalties expressed as a percentage in the high single digits on net sales of products, or Royalty-Bearing Products (i) defined by or containing a composition defined by any claim of the USN Licensed Patent, (ii) made by a method claimed in a Licensed Invention, (iii) based on, originating from or containing USN Materials, or (iv) based on, originating from or supported by USN-created information not found within the USN Licensed Patent and used to support the commercialization or regulatory approval of a Royalty-Bearing Product, including, DNA sequence data, clinical trial data and detailed laboratory methods, related to the Licensed Invention.
APT agreed to pay minimum annual royalties in the amount of $5,000 from 2018 to 2020 and $20,000 thereafter. APT also agreed to pay (a) a regulatory approval royalty in the low $100,000s within 180 days of receiving FDA approval to market a Royalty-Bearing Product and (b) a revenue milestone royalty in the low $100,000s when certain revenue thresholds have been met. Additionally, we agreed to pay royalties expressed as a percentage in the mid-twenties of all revenue received from sublicensing any Royalty-Bearing Product.
We are responsible for controlling and diligently prosecuting the USN Licensed Patent and paying all costs associated with prosecuting and maintaining the USN Licensed Patent in the United States and in foreign jurisdictions. We agreed to submit annual progress reports on our efforts to achieve a practical application of the Licensed Invention by January 1, 2021, and thereafter until such practical application has been received.
We may terminate the USN License Agreement upon 120 days’ written notice, and the USN may terminate the USN License Agreement if (i) the USN determines we are not executing the Commercial Development Plan, (ii) the USN determines such termination is necessary to meet requirements for public use specified by U.S. federal regulations issued after the date of the USN License Agreement and not reasonably satisfied by us, (iii) in the event we willfully made a material false statement or omitted a material fact in our application for the USN License Agreement or any report required thereby, or (iv) we commit a substantial material breach of the USN License Agreement that has not been remedied within 30 days of written notice.
26
License Agreement with Walter Reed Army Institute of Research
On August 24, 2021, APT entered into a Biological Materials License Agreement (or, as modified on August 31, 2022, the WRAIR License Agreement) with Walter Reed Army Institute of Research or WRAIR, pursuant to which APT received a nonexclusive worldwide license to certain materials and information, including approximately 100 phage, or WRAIR Materials, to develop and commercialize phage products to treat/prevent Pseudomonas aeruginosa, Acinebactor baumannii, Staphylococcus aureus, Klebsiella pneumonia, wound and UTI Escherichia coli and Enterobacter cloacae bacterial infections. The Company uses the phage provided in connection with the WRAIR License Agreement as a potential source of phage for the development of its phage treatments.
In connection with the WRAIR License Agreement, APT paid WRAIR an initial execution fee in the mid-thousands of dollars and agreed to pay a maintenance fee in the mid-thousands of dollars per year. We are also required to pay royalties expressed as a percentage in the low single digits on net sales of products that incorporate the WRAIR Materials, or the WRAIR Licensed Products, subject to reductions as described in the WRAIR License Agreement. In addition, if we sublicense our rights under the WRAIR License Agreement we are obligated to pay WRAIR additional sublicense royalties expressed as a percentage in the low teens of the sublicensing receipts we receive from any such sublicense royalties. In addition, additional royalties in the low teens may be assessed on any overdue royalty payments.
We are obligated to make written annual progress reports to WRAIR, detailing our efforts to bring any inventions licensed under WRAIR License Agreement to the point of practical application, together with any additional information requested by WRAIR or as contemplated or required under the development plan. As part of our performance under the WRAIR License Agreement, we have agreed to dose the first patient in a clinical trial with a WRAIR Licensed Product within four years from the effective date of the WRAIR License Agreement.
In the event WRAIR files a non-provisional patent application covering the WRAIR Materials and/or the use thereof, provided as part of this License Agreement, WRAIR is obligated to notify us, and we and WRAIR will assess the need and/or desirability of a patent license. In such case, we will have the first right of refusal to negotiate a non-exclusive or exclusive license.
The WRAIR License Agreement will expire as to each WRAIR Material ten years from the date that such WRAIR Material was added to the WRAIR License Agreement unless earlier terminated in accordance with its terms. We may terminate the WRAIR License Agreement upon 60 days’ written notice, and WRAIR may terminate if we are in default and such default has not been remedied within 90 days after written notice of such default.
Employees
As of December 31, 2023, we had 58 full-time employees and 13 part time employees. 21 of our employees have Ph.D. or M.D. degrees and 53 of our employees are currently engaged in research and development and clinical activities. None of our employees is represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be strong.
Corporate Information
The mailing address of our principal executive office is 22 Einstein St., Floor 4, Ness Ziona, Israel 7414003 and the telephone number is (972) 72-394-2377. Our corporate website address is www.biomx.com. The content of our website is not intended to be incorporated by reference into this Annual Report or in any other report or document we file and any references to these websites are intended to be inactive textual references only.
27
ITEM 1A. RISK FACTORS
You should carefully consider the risks and uncertainties described below and the other information in this Annual Report before making an investment in our securities. Our business, financial condition, results of operations, or prospects could be materially and adversely affected if any of these risks occurs, and as a result, the market price of our securities could decline and you could lose all or part of your investment. This Annual Report also contains forward-looking statements that involve risks and uncertainties. See “Cautionary Statement Regarding Forward-Looking Statements.” Our actual results could differ materially and adversely from those anticipated in these forward-looking statements as a result of certain factors, including those set forth below.
Risks Related to Our Business, Technology and Industry
We are a clinical-stage company with limited operating history and have incurred losses since our inception. We anticipate that we will continue to incur significant expenses, and we will continue to incur significant losses for the foreseeable future.
We are a clinical-stage biopharmaceutical company with limited operating history. We have incurred losses in each year since BiomX Ltd.’s inception in 2015. As of December 31, 2023, our accumulated deficit was $163 million, and we expect to incur increasingly significant losses for the foreseeable future. Preclinical development and clinical trials and activities are costly. We have devoted, and will continue to devote for the foreseeable future, substantially all of our resources to research and development and clinical trials for our product candidates. We do not expect to generate any revenue from the commercial sales of our product candidates in the near term. In addition, as a result of the Acquisition, our future business, prospects, financial position and operating results could be significantly different than those in historical periods or projected by our management.
For the years ended December 31, 2023 and 2022, we had losses from operations of $25.3 million and $27.2 million, respectively. We anticipate that the level of our expenses is expected to increase as a result of the recent acquisition of APT, and will continue to be significant if and as we:
| ● | initiate and continue research, preclinical and clinical development efforts for any future product candidates; |
| ● | seek to discover and develop additional product candidates and further expand our clinical product pipeline; |
| ● | seek marketing and regulatory approvals for any product candidates that successfully complete clinical trials; |
| ● | require the manufacture of larger quantities of product candidates for clinical development and, potentially, commercialization; |
| ● | maintain, expand and protect our intellectual property portfolio; |
| ● | expand our research and development infrastructure, including hiring and retaining additional personnel, such as clinical, quality control and scientific personnel; |
| ● | establish sales, marketing, distribution and other commercial infrastructure in the future to commercialize products for which we obtain marketing approval, if any; and |
| ● | add operational, financial and management information systems and personnel, including personnel to support our product development and commercialization and help us comply with our obligations as a public company. |
28
We will need to raise additional capital in the future to support our operations which may not be available at terms that are favorable to us and might cause significant dilution to our stockholders or increase our debt towards third parties.
As of December 31, 2023, we had cash, cash equivalents and restricted cash of $15.9 million, and we have had recurring losses from operations and negative operating cash flows since inception. We will need to raise additional capital in the future to support our operations and product development activities. In the near term, we expect to continue to fund our operations and other development activities relating to additional product candidates from the cash held by us, governmental and other grants and through future equity and debt financing. In addition, on December 7, 2023, we entered into an At the Market Offering Agreement, or the ATM Agreement, with H.C. Wainwright & Co., LLC, or Wainwright, as manager, pursuant to which we may issue and sell shares of our Common Stock having an aggregate offering price of up to $7.5 million from time to time through Wainwright. We are not obligated to make any sales of Common Stock under the ATM Agreement. On May 4, 2023, subsequent to the approval of the Company’s stockholders, the Company completed the second closing of the February 2023 PIPE for an additional $6 million in gross proceeds. On December 7, 2023, we filed a shelf registration statement on Form S-3, which was declared effective by the SEC on January 2, 2024. Additionally, on March 15, 2024, concurrently with the consummation of the Acquisition, we consummated a private placement, or the March 2024 PIPE, pursuant to an exemption from registration requirements under the Securities Act with certain investors pursuant to which such investors purchased an aggregate of 216,417 shares of our Series X non-voting convertible preferred stock, par value $0.0001 per share, or the Convertible Preferred Stock, and warrants, or Private Placement Warrants, to purchase up to an aggregate of 108,208,500 shares of the Company’s Common Stock, for aggregate gross proceeds of approximately $50 million. Each share of Convertible Preferred Stock is convertible into an aggregate of 1,000 shares of Common Stock.
Subject to restrictions pursuant to the March 2024 PIPE, we may continue to sell shares under the ATM Agreement and otherwise to use our shelf registration statement to raise additional funds from time to time. We may also raise funds privately, as we did in February 2023 and the March 2024 PIPE. We may also seek funds through arrangements with collaborators or others that may require us to relinquish rights to the product candidates that we might otherwise seek to develop or commercialize independently.
If we enter into a collaboration for one or more of our current or future product candidates at an earlier development stage, the terms of such a collaboration will likely be less favorable than if we were to enter the collaboration in later stages or if we commercialized the product independently. If we raise additional funds through equity offerings, the terms of these securities may include liquidation or other preferences that adversely affect our stockholders’ rights or cause significant dilution to our stockholders. If we raise additional capital through debt financing, it would be subject to fixed payment obligations and may be subject to covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures, declaring dividends or acquiring or licensing intellectual property rights.
Developing drugs and conducting clinical trials is expensive. Our future funding requirements will depend on many factors, including:
| ● | the costs, timing and progress of our research and development and clinical activities; |
| ● | manufacturing costs associated with our targeted bacteriophage, or phage, therapies strategy and other research and development activities; |
| ● | the terms and timing of any collaborative, licensing, acquisition or other arrangements that we may establish; |
| ● | employee-related expenses, as well as external costs such as fees paid to outside consultants; |
| ● | the costs and timing of seeking regulatory approvals and related to compliance with regulatory requirements; and |
| ● | the costs of filing, prosecuting, defending and enforcing any patent applications, claims, patents and other intellectual property rights. |
Domestic and international equity and debt markets have experienced and may continue to experience heightened volatility and turmoil based on domestic and international economic conditions and concerns. In the event these economic conditions and concerns continue or worsen and the markets continue to remain volatile, or a bear market, or recession, ensues in the U.S. stock market, or the markets are negatively impacted by factors such as the Israel-Hamas War, the Russian invasion of Ukraine and the resulting world sanctions on Russia, Belarus, and related parties or other sources of geopolitical uncertainty and instability, our operating results and liquidity could be affected adversely by those factors in many ways, including making it more difficult for us to raise funds if necessary and our stock price may decline.
29
There can be no assurance that sufficient funds will be available to us when required or on acceptable terms, if at all. Our inability to obtain additional funds could have a material adverse effect on our business, financial condition and results of operations. Moreover, if we are unable to obtain additional funds on a timely basis, there will be substantial doubt about our ability to continue as a going concern and increased risk of insolvency and up to a total loss of investment by our stockholders.
Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern, which could prevent us from obtaining new financing on reasonable terms or at all.
Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern. We have concluded that there is substantial doubt about our ability to continue as a going concern. We have accumulated a deficit of $163 million since our inception. To date, we have not generated revenue from our operations and we do not expect to generate any significant revenues from sales of products in the next twelve months. Our cash needs may increase in the foreseeable future. As of December 31, 2023, we had $15.9 million of cash and cash equivalents.
We believe our cash and cash equivalents and short-term deposits on hand will be sufficient to meet our working capital and capital expenditure requirements for at least 12 months. However, since there is a risk of our stockholders not approving the conversion of the Convertible Preferred Stock that was issued in connection with the March 2024 PIPE and the Acquisition, which could result in us being required to cash settle the Convertible Preferred Stock, there is substantial doubt about our ability to continue as a going concern for at least 12 months from April 3, 2024. Our continuation as a going concern is dependent upon many factors, including our ability to receive the approval of our shareholders to convert the Convertible Preferred Stock within 5 months, raise additional funds, the success of our clinical trial for CF, our ability to successfully integrate the business of APT and our ability to repay our obligations when due. We cannot be sure that we will be able to obtain any future funding, and any such funding we may obtain may not be sufficient to finance our operations. If we are unable to obtain sufficient funds, we may be unable to continue as a going concern.
There is no guarantee that our acquisition of APT will increase stockholder value.
In March 2024, we acquired APT in the Acquisition. We cannot guarantee that implementing the Acquisition and related transactions will not impair stockholder value or otherwise adversely affect our business. The Acquisition could result in integration challenges between our businesses and management teams which could cause management and business disruptions, any of which could impact our results of operation and business prospects and impair the value of such acquisition to our stockholders.
We are required to use reasonable best efforts to solicit stockholder approval for the conversion of shares of Convertible Preferred Stock and Warrants issued in the Acquisition and the March 2024 PIPE. If we do not obtain such approval within 150 days of the initial issuance of the Convertible Preferred Stock, we could be required to cash settle the Convertible Preferred Stock.
Pursuant to the Merger Agreement, we are required to hold a meeting of stockholders (the “Stockholder Meeting”) for the purpose of obtaining stockholder approval of (i) the conversion of the Convertible Preferred Stock and the exercise of the Warrants (as defined below) into shares of Common Stock in excess of 19.9% of the outstanding shares of Common Stock for purposes of the NYSE American Stock Market Rules, (ii) adoption of a new stock incentive plan or amendment of the Company’s current stock incentive plan (the “2024 Incentive Plan”), and (iii) if necessary, the amendment of our certificate of incorporation to authorize sufficient additional shares of Common Stock to allow for conversion of the Convertible Preferred Stock and exercise of the Warrants. If such stockholder approval is not received, we are required to convene additional stockholder meetings at least every 90 days thereafter until such approval is obtained, which could result in substantial costs and be a distraction to management. Furthermore, if our stockholders do not approve the conversion of the Convertible Preferred Stock within 150 days of the initial issuance of the Convertible Preferred Stock, then upon written request by the holders of 70% of the Convertible Preferred Stock, we will be required to pay to each holder of Convertible Preferred Stock an amount in cash equal to the fair value of the shares of Convertible Preferred Stock held by such holder, as described in the Certificate of Designation for the Convertible Preferred Stock. We do not expect that we would have sufficient liquidity to settle a significant amount of the Convertible Preferred Stock if required to do so. The cash settlement is not in our control and raises substantial doubt about our ability to continue as a going concern.
30
We are seeking to develop product candidates using phage technology, an approach for which it is difficult to predict the time and cost of development. To our knowledge, as of the date of this Annual Report, no bacteriophage has thus far been approved as a drug in the United States or in the European Union.
We are developing our product candidates with phage technology. We have not, nor to our knowledge has any other company, received regulatory approval from the FDA, or equivalent foreign regulatory agencies for a product candidate based on this approach. While in vitro and in vivo studies have characterized the behavior of phage in cell cultures and animal models and there exists a body of literature regarding the use of phage therapy in humans, the safety and efficacy of phage therapy in humans has not been extensively studied in well-controlled modern clinical trials. Most of the prior research on phage-based therapy was conducted in the former Soviet Union prior to and immediately after World War II and lacked appropriate control group design or lacked control groups at all. Furthermore, the standard of care has changed substantially during the ensuing decades since those studies were performed, diminishing the relevance of prior claims of improved cure rates. Any product candidates that we develop may not demonstrate in patients the therapeutic properties ascribed to them in laboratory and other preclinical studies, and they may interact with human biological systems in unforeseen, ineffective or even harmful ways. We cannot be certain that our approach will lead to the development of approvable or marketable products. Furthermore, the bacterial targets of phage may develop resistance to our product candidates over time, which we may or may not be able to overcome with the development of new phage cocktails or we may not be able to construct a cocktail with sufficient coverage of our target pathogen universe.
If our product candidates receive regulatory approval but do not achieve an adequate level of acceptance by physicians, healthcare payors and patients, we may not generate product revenue sufficient to attain profitability. Our success will depend upon physicians who specialize in the treatment of diseases targeted by our product candidates that we pursue as drugs, prescribing potential treatments that involve the use of our product candidates in lieu of, or in addition to, existing treatments with which they are more familiar and for which greater clinical data may be available. Our success will also depend on consumer acceptance and adoption of our products that we commercialize. Adverse events in preclinical studies and clinical trials of our product candidates or in clinical trials of others developing similar products and the resulting publicity, as well as any other adverse events in the field of phage therapeutics, could result in a decrease in demand for any product that we may develop. The degree of market acceptance of any approved products will depend on a number of factors, including:
| ● | the effectiveness of the product; |
| ● | the prevalence and severity of any side effects; |
| ● | potential advantages or disadvantages over alternative treatments; |
| ● | relative convenience and ease of administration; |
| ● | the strength of marketing and distribution support; |
| ● | the price of the product, both in absolute terms and relative to alternative treatments; and |
| ● | sufficient third-party coverage or reimbursement. |
Developing our product candidates on a commercial scale will require substantial technical, financial and human resources. We and our third-party collaborators may experience delays in developing manufacturing capabilities for our product candidates, and may not be able to do so at the scale required to efficiently conduct the clinical trials required to obtain regulatory approval of those of our product candidates that require it, or to manufacture commercial quantities of our products, if approved or otherwise permitted to be marketed.
31
Our product candidates must undergo clinical testing which may fail to demonstrate the requisite safety and efficacy for drug products, or safety, purity, and potency for biologics, and any of our product candidates could cause adverse effects, which would substantially delay or prevent regulatory approval and/or commercialization.
Before we can obtain regulatory approval for a product candidate or otherwise obtain evidence allowing us to market the product as a drug or biologic, we must undertake extensive preclinical and clinical testing in humans to demonstrate safety and efficacy or in the case of biologics, safety, purity, and potency, to the satisfaction of the FDA or other regulatory agencies. Clinical trials of product candidates sufficient to obtain regulatory marketing approval or otherwise demonstrate safety prior to marketing, are expensive and take years to complete. Furthermore, results from these clinical trials may not show safety or efficacy of our product candidates sufficient to lead to approval, or to warrant further development. Our approach is intended to design phage combinations, or cocktails, to target specific strains of pathogenic bacteria in order to alter microbiome composition and confer potential therapeutic or cosmetic benefit to patients. However, there can be no assurance that the eradication of the selected targets will result in a clinically meaningful effect on the underlying disease, such as in cases where the pathology of the disease is not well-defined. In addition, the bacteria that we target may be associated with the disease, but may not be causative or contributive to the pathology of the disease, or there may be other bacteria that our product candidates do not target that are more meaningful drivers of the underlying disease. In addition, our product candidates require the use of effective delivery vehicles to reach the target organ or tissue, and there can be no assurance that our intended delivery systems will allow our product candidates to reach the desired locations in a patient. Safety must first be established through preclinical testing and early clinical trials, before efficacy can be evaluated and established and thereby lead to FDA or other regulatory agencies marketing approval. Our clinical trials may produce undesirable side effects or negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical and/or preclinical testing or to abandon programs.
Ongoing geopolitical instability have adversely affected and may continue to adversely affect our business, including our clinical trials.
General economic, political, demographic and business conditions worldwide, including geopolitical uncertainty and instability, such as the Israel- Hamas War and the Russia-Ukraine conflict, might adversely affect our business, through indirect disruption to our supply chain, harming our ability to raise funds at terms acceptable to us among other affects. We may further experience additional disruptions that could severely impact our business, preclinical studies and clinical trials, including:
| ● | delays or difficulties in enrolling patients in our clinical trials; |
| ● | delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff; |
| ● | interruption of, or delays in receiving, supplies of our product candidates from our contract manufacturing organizations due to staffing shortages, production slowdowns or stoppages and disruptions in delivery systems; and |
| ● | interruptions or delays to our sourced discovery and clinical activities. |
32
If we are not able to obtain, or if there are delays in obtaining, required regulatory approvals for our product candidates for therapeutic indications, we will not be able to commercialize, or will be delayed in commercializing, our product candidates, and our future ability to generate revenue will be materially impaired.
Our product candidates and the activities associated with their development and commercialization for therapeutic indications, including their design, testing, manufacture, safety, efficacy, recordkeeping, labeling, storage, approval, advertising, promotion, sale, distribution, import and export are subject to regulation by the FDA and other regulatory agencies in the United States and by equivalent foreign regulatory authorities. Before we can commercialize any of our product candidates for therapeutic indications, we must obtain marketing approval. We have not received approval to market any of our product candidates from regulatory authorities in any jurisdiction, and it is possible that none of our product candidates or any product candidates we may seek to develop in the future will ever obtain regulatory approval.
The process of obtaining regulatory approvals for therapeutic indications, both in the United States and in other countries, is expensive, may take many years if additional clinical trials are required, and can vary substantially based upon a variety of factors, including the type, complexity and novelty of the product candidates involved. Changes in marketing approval policies during the development period, changes in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted IND, or equivalent application types, may cause delays in the approval or rejection of an application. The FDA and equivalent foreign regulatory authorities have substantial discretion in the approval process and may refuse to accept any application or may decide that our data is insufficient for approval and require additional preclinical, clinical or other studies. Our product candidates could be delayed in receiving, or fail to receive, regulatory approval for many reasons, including the following:
| ● | the FDA or equivalent foreign regulatory authorities may disagree with the design, including study population, dose level, dose regimen, and bioanalytical assay methods, or implementation of our clinical trials; |
| ● | we may be unable to demonstrate to the satisfaction of the FDA or equivalent foreign regulatory authorities that a drug candidate is safe and effective for its proposed indication or a related companion diagnostic is suitable to identify appropriate patient populations; |
| ● | the results of clinical trials may not meet the level of statistical significance required by the FDA or equivalent foreign regulatory authorities for approval, such as was the case with our acne product candidate; |
| ● | we may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks; |
| ● | the FDA or equivalent foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials; |
| ● | the data collected from clinical trials of our product candidates may not be sufficient to support the submission of a marketing application or other submission or to obtain regulatory approval in the United States or elsewhere; |
| ● | the FDA or equivalent foreign regulatory authorities may fail to approve the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; and |
| ● | the approval policies or regulations of the FDA or equivalent foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
Of the large number of drugs in development, only a small percentage successfully complete the FDA or equivalent foreign regulatory approval processes and are commercialized. The lengthy approval process as well as the unpredictability of future clinical trial results may result in us failing to obtain regulatory approval to market its product candidates, which would significantly harm our business, results of operations and prospects.
33
The FDA may also require a panel of experts, referred to as an Advisory Committee, to deliberate on the adequacy of the safety and efficacy data to support approval for therapeutic indications. The opinion of the Advisory Committee, although not binding, may have a significant impact on our ability to obtain approval of any product candidates that we develop based on the completed clinical trials. In the European Union, the safety and efficacy data of any product candidate considered by the EMA to qualify as an advanced therapy medicinal product must be reviewed by the EMA’s, Committee for Advanced Therapies, a group of experts in advanced therapy medicinal products.
Moreover, under PREA, in the United States, and the Pediatric Regulation, in the European Union, the FDA or equivalent foreign regulatory authority could require mandatory testing in the pediatric population. Applications for approval in the United States or in the European Union must contain data to assess the safety and efficacy of the biologic for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA or equivalent foreign regulatory authority may, in its discretion, grant full or partial waivers, or deferrals, for submission of data in pediatric subjects. If the FDA requires data in pediatric patients, significantly more capital will have to be invested in order to conduct the mandatory pediatric clinical trials and studies, but the approval of the medicinal products for the adult population should normally not be affected. If the results of such pediatric studies are not positive, our product candidates will not be approved for children.
In addition, even if we were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited therapeutic indications than our requests, may include limitations for use or contraindications that limit the suitable patient population, may not approve the price we intend to charge for our products, may grant approval contingent on the performance of costly post-marketing clinical trials or may approve a product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate. Any of the foregoing scenarios could materially harm the commercial prospects for our product candidates.
If we experience delays in obtaining approval or if we fail to obtain approval of our product candidates, the commercial prospects for our product candidates may be harmed and our future ability to generate revenues will be materially impaired.
We have never generated any revenue from product sales and may never be profitable or, if achieved, may not sustain profitability.
Our ability to generate meaningful revenue and achieve profitability depends on our ability, and the ability of any third party with which we may partner, to successfully complete the development of, and meet regulatory requirements, including (but not limited to) obtaining any necessary regulatory approvals, to commercialize our product candidates. We do not currently meet regulatory requirements or have the required approvals to market our product candidates and may never meet or receive them. We do not anticipate generating revenue from product sales for the foreseeable future, if ever. If any of our product candidates fail in clinical trials or if any of our product candidates do not meet regulatory requirements, including gaining regulatory approval when needed, or if any of our product candidates, if marketed, fail to achieve market acceptance, we may never become profitable. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Our ability to generate future revenue from product sales depends heavily on our success in:
| ● | completing research and preclinical and clinical development of our product candidates; |
| ● | seeking and obtaining regulatory and marketing approvals for product candidates for which we complete clinical trials; |
| ● | meeting regulatory requirements for marketing the products; |
| ● | developing a sustainable, scalable, reproducible and transferable manufacturing process for our product candidates; |
34
| ● | launching and commercializing product candidates for which we obtain regulatory and marketing approval or are otherwise permitted to market, either by establishing a sales force, marketing and distribution infrastructure or by collaborating with a partner; |
| ● | obtaining market acceptance of any approved products; |
| ● | addressing any competing technological and market developments; |
| ● | implementing additional internal systems and infrastructure, as needed; |
| ● | identifying and validating new product candidates; |
| ● | negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter; |
| ● | maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how; and |
| ● | attracting, hiring and retaining qualified personnel. |
Even if one or more of the product candidates that we develop is approved for commercial sale or otherwise permitted for marketing, we anticipate incurring significant costs associated with commercializing any approved product. Our expenses could increase beyond expectations if we are required by the FDA, or the EMA, or other equivalent foreign regulatory agencies to perform clinical trials and other studies in addition to those that we currently anticipate. Even if we are able to generate revenue from the sale of any approved products, we may not become profitable and may need to obtain additional funding to continue operations. If we fail to become profitable, or if we are unable to fund our continuing losses, our business, financial condition and results of operations may be materially adversely impacted.
We are seeking to develop product candidates to treat medical conditions related to the presence of certain bacteria. Our success is largely dependent on a broad degree of market acceptance, and in the case of drug products, physician adoption and use, which are necessary for commercial success.
Even if we obtain FDA or foreign regulatory approvals for our drug product candidates, the commercial success of our product candidates will depend on consumer acceptance and adoption of products that we commercialize. Adverse events in preclinical studies and clinical trials of our product candidates or in clinical trials of others developing similar products and the resulting publicity could result in a decrease in demand for any product that we may develop.
In addition, the commercial success of our drug product candidates will depend significantly on their broad adoption and use by pediatricians and other physicians for approved therapeutic indications, as well as any other indications for which we may seek approval. We cannot be certain that our approach will lead to the development of approvable or marketable products.
Obtaining high titers for specific phage cocktails necessary for our preclinical and clinical testing may be difficult and time-consuming.
Our product candidates are phage cocktails that we have designed to meet specific characteristics. We and our contract manufacturers produce a cocktail of multiple phage and it may be difficult or time-consuming to achieve high titers, or levels, of phage sufficient for our preclinical and clinical testing. In some cases, it may require multiple product runs in order for us to obtain the amounts necessary for its clinical testing. This may result in delays in our clinical trial timelines, and it may increase production costs and associated expenses. Also, it may be difficult to reproduce the manufacturing process to the extent that more significant quantities are required as our product candidates advance through the clinical development process.
35
Results from preclinical studies of our product candidates may not be predictive of the results of clinical trials or later stage clinical development.
Preclinical studies of our product candidates, such as BX004 and BX005, including studies in animal disease models may not accurately predict the safety of the product candidate such that further human clinical trials would be allowed to proceed. In particular, promising preclinical testing suggesting the potential efficacy of prototype phage products may not predict the ability of these products to address conditions in the human clinical settings. For example, while we have studied phage activity in vitro and in vivo, these results may not be replicated when our phage cocktails are administered to human subjects. Despite promising data in any preclinical studies, our phage technology may be found not to be efficacious when studied in clinical trials.
To satisfy FDA or equivalent foreign regulatory approval standards, we must demonstrate in adequate and well controlled clinical trials that our drug product candidates are safe and effective for their intended use. Success in preclinical testing and early-stage clinical trials does not ensure that later clinical trials will be successful. Our initial results from preclinical testing also may not be confirmed by later analysis or subsequent larger clinical trials. A number of companies in the pharmaceutical industry have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier clinical trials, and most product candidates that commence clinical trials are never approved for commercial sale.
If we encounter difficulties enrolling patients in our clinical trials, our clinical development activities could be delayed or otherwise adversely affected.
Completion of clinical trials depends, among other things, on our ability to enroll a sufficient number of patients, which is a function of many factors, including:
| ● | the therapeutic endpoints chosen for evaluation; |
| ● | the eligibility criteria defined in the protocol; |
| ● | the perceived benefit of the product candidate under study; |
| ● | the size of the patient population required for analysis of the clinical trial’s therapeutic endpoints; |
| ● | our ability to recruit clinical trial investigators and sites with the appropriate competencies and experience; |
| ● | our ability to obtain and maintain patient consents; and |
| ● | competition for patients from clinical trials for other treatments. |
We have experienced and may continue to experience difficulties in enrolling patients in our clinical trials, which could increase the costs or affect the timing or outcome of these clinical trials. This is particularly true with respect to diseases with relatively small patient populations. In addition, potential patients for our trials may not be adequately diagnosed or identified with the diseases that we are targeting or may not meet the entry criteria for our studies.
We may not be able to initiate or continue clinical trials if we are unable to locate a sufficient number of eligible patients to participate in the clinical trials required by the FDA or equivalent foreign regulatory agencies. In addition, the process of finding and diagnosing patients may prove costly. Our inability to enroll a sufficient number of patients for any of our clinical trials would result in significant delays or may require us to abandon one or more clinical trials.
36
Delays in our clinical trials could result in us not achieving anticipated developmental milestones when expected, increased costs and delays in our ability to obtain regulatory approval for and commercialization of our product candidates.
Delays in our clinical trials could result in us not meeting anticipated clinical milestones and could materially impact our product development costs and delay regulatory approval of our product candidates. Planned clinical trials may not be commenced or completed on schedule, or at all.
Clinical trials can be delayed for a variety of reasons, including:
| ● | delays in the development of manufacturing capabilities for our product candidates to enable their consistent production at clinical trial scale; |
| ● | failures in our internal manufacturing operations that result in our inability to consistently and timely produce bacteriophage in sufficient quantities to support our clinical trials; |
| ● | the availability of financial resources to commence and complete our planned clinical trials; |
| ● | delays in reaching a consensus with clinical investigators on study design; |
| ● | delays in reaching a consensus with regulatory agencies on trial design or in obtaining regulatory approval to commence a trial; |
| ● | delays in obtaining clinical materials; |
| ● | slower than expected patient recruitment for participation in clinical trials; |
| ● | regulatory constraints or injunctions (for example, from supervisory authorities in case of noncompliance with cybersecurity and data privacy laws); |
| ● | failure by clinical trial sites, other third parties or us to adhere to clinical trial agreements and/or the trial protocol; |
| ● | delays in reaching agreement on acceptable clinical trial agreement terms with prospective sites or obtaining IRB or independent ethics committee approval; and |
| ● | adverse safety events experienced during our clinical trials. |
If we do not successfully commence or complete our clinical trials on schedule, the price of our securities may decline. Significant preclinical or clinical trial delays could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do, potentially impairing our ability to successfully commercialize our product candidates and harming our business and results of operations.
37
Our current or future product candidates may cause adverse effects that could halt their clinical development, prevent their approval or marketing, limit their commercial potential or result in significant negative consequences.
Adverse effects could occur and cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of marketing approval by the FDA or equivalent foreign regulatory agencies. Results of our trials could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics.
If adverse effects arise in the development of our product candidates, we, the FDA or equivalent foreign regulatory agencies, the IRBs or independent ethics committees at the institutions in which our studies are conducted, or the Data Safety Monitoring Board could suspend or terminate our clinical trials or the FDA or equivalent foreign regulatory agencies could deny approval of our product candidates for any or all targeted indications.
We intend to continue to evaluate our product candidates for safety and tolerability in the form of Phase 1 clinical trials. While our current and future product candidates will undergo safety testing to the extent possible and, where applicable, under such conditions discussed with regulatory authorities, not all adverse effects of drugs can be predicted or anticipated. Unforeseen adverse effects could arise either during clinical development or, if such adverse effects are more rare, after our products have been approved by regulatory authorities and the approved product has been marketed, resulting in the exposure of additional patients. For example, while we screen our phage in attempts to minimize safety issues, there can be no assurance that we will eliminate the risk of the appearance of virulence genes, antibiotic resistance genes, lysogenic genes, integrase genes, or other toxic genes in our phage, or of adverse reactions to our phage in a patient’s immune system. So far, we have not demonstrated, and we cannot predict, if ongoing or future clinical trials will demonstrate that any of our product candidates are safe in humans. Moreover, clinical trials of our product candidates are conducted in carefully defined sets of patients who have agreed to enter into clinical trials. Consequently, it is possible that our clinical trials may indicate an apparent positive effect of a product candidate that is greater than the actual positive effect, if any, or alternatively fail to identify undesirable adverse effects.
Ultimately, some or all of our product candidates may prove to be unsafe for human use. Moreover, we could be subject to significant liability if any volunteer or patient suffers, or appears to suffer, adverse health effects as a result of participating in our clinical trials. Any of these events could prevent us from achieving or maintaining market acceptance of our product candidates and could substantially increase commercialization costs.
We have not completed composition development of our product candidates.
The development of our product candidates requires that we isolate, select, optimize and combine a number of phage that target the desired bacteria for that product candidate. The selection of phage for any of our product candidates is based on a variety of factors, including, without limitation, the ability of the selected phage, in combination, to successfully kill the targeted bacteria, the degree of cross-reactivity of the individual phage with the same part of the bacterial targets, the ability of the combined phage to satisfy regulatory requirements, our ability to manufacture sufficient quantities of the phage, intellectual property rights of third parties, and other factors. While we have selected an initial formulation of BX004, there can be no assurance that this initial formulation will be the final formulations of this product candidate for commercialization if approved. If we are unable to complete formulation development of our product candidates in the time frame that we have anticipated, then our product development timelines, and the regulatory approval of our product candidates, could be delayed.
38
We must continue to develop manufacturing processes for our product candidates, and any delay in doing so, or our inability to do so, would result in delays in our clinical trials.
The manufacturing processes for our product candidates, and the scale-up of such processes for clinical trials, may present challenges, and there can be no assurance that we will be able to complete this work in a timely manner, if at all. Any delay in the development or scale-up of these manufacturing processes could delay the start of clinical trials and harm our business. In order to scale-up our manufacturing capacity, we need to either build additional internal manufacturing capacity, contract with one or more partners, or both. Our technology and the production process for our equipment and tools are complex and we may encounter unexpected difficulties in manufacturing our product candidates. For example, the manufacturing hosts that we use to produce our phage may contain one or more integrated phage in their genomes that, if we are unable to remove, can present challenges in manufacturing of the produced phage. There is no assurance that we will be able to continue to build manufacturing capacity internally or find one or more suitable partners, or both, to meet the necessary volume and quality requirements. Manufacturing and product quality issues may arise as we increase the scale of our production. Any delay or inability in establishing or expanding our manufacturing capacity could diminish our ability to develop our product candidates.
In the third quarter of 2019, we established our own manufacturing facility at our headquarters in Ness Ziona, Israel and we have executed cGMP manufacturing for our first in human clinical study. In February 2021, APT consolidated its GMP manufacturing, testing and development in its Gaithersburg facility. In March 2021, we moved into a new manufacturing facility at our headquarters in Ness Ziona, Israel. Our new facility undergoes ongoing internal inspections to verify proper manufacturing for Phase I and II clinical studies in accordance with cGMP requirements. In the event these facilities do not comply with cGMP standards for the manufacture of our product candidates, we may need to fund additional modifications to our manufacturing process, conduct additional validation studies or find alternative manufacturing facilities, any of which would result in significant cost to us as well as a delay of up to several years in obtaining approval for such product candidate.
If we submit marketing applications for any of our product candidates manufactured at this facility, this manufacturing facility will be subjected to ongoing periodic inspection for compliance with European, FDA and cGMP regulations. Compliance with these regulations and standards is complex and costly, and there can be no assurance that we will be able to comply. Any failure to comply with applicable regulations could result in sanctions being imposed (including fines, injunctions and civil penalties), failure of regulatory authorities to grant marketing approval of our product candidates, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of product candidates or products, operating restrictions and criminal prosecution.
If our competitors are able to develop and market products that are more effective, safer or more affordable than ours, or obtain marketing approval before we do, our commercial opportunities may be limited.
Competition in the biotechnology and pharmaceutical industries is intense and continues to increase. Some companies that are larger and have significantly more resources than us are aggressively pursuing development programs for indications that we are pursuing, including traditional therapies and therapies with novel mechanisms of action. In addition, other companies are developing phage-based products for therapeutic and non-therapeutic uses, and may elect to use their expertise in phage development and manufacturing to try to develop products that would compete with our products.
We also face potential competition from academic institutions, government agencies and private and public research institutions engaged in the discovery and development of drugs and therapies. Many of our competitors have significantly greater financial resources and expertise in research and development, preclinical testing, conducting clinical trials, obtaining regulatory approvals, manufacturing, sales and marketing than we do. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established pharmaceutical companies.
In the European Union, potential competition also comes from medicinal preparations made by hospitals or pharmacists and administered without marketing authorizations, generally referred to as “compounding.” In some member states, national authorities generally promote compounding in order to reduce healthcare expenses.
Our competitors may succeed in developing products that are more effective, have fewer side effects and are safer or more affordable than our product candidates, which would render our product candidates less competitive or noncompetitive and would prevent the granting or maintenance of an orphan designation. These competitors also compete with us to recruit and retain qualified scientific and management personnel, establish clinical trial sites and patient registration for clinical trials, as well as to acquire technology and technology licenses complementary to our programs or advantageous to our business. Moreover, competitors that are able to achieve patent protection, obtain regulatory approvals and commence commercial sales of their products before we do, and competitors that have already done so may enjoy a significant competitive advantage.
39
We may not be successful in our efforts to identify or discover additional product candidates.
Although we intend to utilize our technology to evaluate other therapeutic opportunities in addition to the product candidates that we are currently developing, we may fail to identify other product candidates for clinical development for a number of reasons. For example, our research methodology may not be successful in identifying potential product candidates, or those we identify may be shown to have harmful side effects or other characteristics that make them unmarketable or unlikely to receive regulatory approval. In addition, we may not be able to identify phage that eradicate the target bacteria, including due to sourcing difficulties such as lack of diversity, inability to obtain samples in a timely manner or at all, or contamination in the samples. We may also encounter difficulties in designing phage cocktails that meet the requirements of an investigational therapy, including due to the build-up of resistances in bacteria to our phage, the range of host bacteria that are affected by our phage, the variety of activity on different bacteria growth states, issues with toxicity in our phage, and the stability, robustness and ease of manufacturing of our product candidates. In addition, the designing of synthetically engineered phage may fail to result in the development of phage with the desired characteristics or behaviors that are suitable for use as viable therapies, or may result in phage that contain undesired features such as immunogenicity, toxicity and other safety concerns.
A key part of our strategy is to utilize our screening technology to identify product candidates to pursue in clinical development. If we fail to identify and develop additional potential product candidates, we may be unable to grow our business and our results of operations could be materially harmed. Such product candidates will require additional, time-consuming development efforts prior to commercial sale, including preclinical studies, clinical trials and approval by the FDA and/or applicable foreign regulatory agencies. All product candidates are prone to the risks of failure that are inherent in pharmaceutical product development.
Legal requirements as well as ethical and social concerns about synthetic biology and genetic engineering could limit or prevent the use of our technologies and limit our revenues.
Our technology may include the use of synthetic biology and genetic engineering. In some countries, drugs made using genetically modified organisms may be subject to a more stringent legal regime, which could prove to be complex and very challenging, especially for a small life sciences company. For example, in the European Union, the rules on genetically modified organisms would apply in addition to the general rules on medicinal products or cosmetic products. The rules on advanced therapy medicinal products may also apply.
Additionally, public perception about the safety and environmental hazards of, and ethical concerns over, synthetic biology and genetic engineering could influence public acceptance of our technologies, product candidates and processes. If we and our collaborators are not able to overcome the legal challenges as well as the ethical and social concerns relating to synthetic biology and genetic engineering, our technologies, product candidates and processes may not be accepted. These challenges and concerns could result in increased expenses, regulatory scrutiny and increased regulation, trade restrictions on imports of our product candidates, delays or other impediments to our programs or the public acceptance and commercialization of our products. We design and produce product candidates with characteristics comparable or superior to those found in naturally occurring organisms or enzymes in a controlled laboratory; however, the release of such organisms into uncontrolled environments could have unintended consequences. Any adverse effect resulting from such a release could have a material adverse effect on our business, financial condition or results of operations, and we may have exposure to liability for any resulting harm.
We may expend our limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and managerial resources, we intend to focus on developing product candidates for specific indications that we identify as most likely to succeed, in terms of both their potential for marketing approval and commercialization. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that may prove to have greater commercial potential. For example, we spent significant time and resources developing BX001, which we discontinued, and our BX005 product candidates and CRC development efforts, which we have paused indefinitely.
Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on current and future research and development programs and product candidates for specific indications may not yield any commercially viable product candidates. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to the product candidate.
We intend to continue to rely on our BOLT proprietary product platform to develop our phage therapies. Our competitive position could be materially harmed if our competitors develop similar platforms and develop rival product candidates.
Our BOLT platform enables us to rapidly develop, manufacture and formulate phage therapy candidates targeting particular pathogenic bacteria and incorporates our experience over the past six years with process refinement and implementation of technological advancements. For a given indication, the platform will typically allow for the completion of a clinical proof of concept study in patients, meaning Phase 2 results, within approximately 12-18 months from project initiation; however in certain indications the length of clinical proof of concept may be longer depending on the indication, identity of target bacteria, recruitment rate, cohort size and other factors, and we may not achieve clinical proof of concept on that timeline, or at all. We are initially aiming to complete a clinical proof of concept study in patients within approximately 12-18 months from project initiation in our CF program. We have limited experience with our BOLT platform and may not achieve the benefits we anticipate. To the extent we utilize our resources to further develop our BOLT platform, we may become more dependent on its success.
40
There is a substantial risk of product liability claims in our business. If we do not obtain sufficient liability insurance, a product liability claim could result in substantial liabilities to us.
Our business exposes us to significant potential product liability risks that are inherent in the development, manufacturing and marketing of human therapeutic products. Regardless of merit or eventual outcome, product liability claims may result in:
| ● | delay or failure to complete our clinical trials; |
| ● | withdrawal of clinical trial participants; |
| ● | decreased demand for our product candidates; |
| ● | injury to our reputation; |
| ● | litigation costs; |
| ● | substantial monetary awards against us; and |
| ● | diversion of management or other resources from key aspects of our operations. |
If we succeed in marketing products, product liability claims could result in an FDA or equivalent foreign regulatory agency investigation of the safety or efficacy of our products, our manufacturing processes and facilities or our marketing programs. Such investigation could also potentially lead to a recall of our products or more serious enforcement actions, or limitations on the indications, for which they may be used, or suspension or withdrawal of approval.
We currently only have limited clinical trials insurance policies that cover clinical trials in certain territories. We intend to expand our insurance coverage to include the sale of commercial products if marketing approval is obtained for our product candidates or any other compound that we may develop. However, insurance coverage is expensive, and we may not be able to maintain insurance coverage at a reasonable cost or at all, and the insurance coverage that we have or obtain may not be adequate to cover potential claims or losses.
41
Our employees, independent contractors, consultants, commercial partners and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We are exposed to the risk of employee fraud or other illegal activity by our employees, independent contractors, consultants, commercial partners and vendors. Misconduct by these parties could include intentional, reckless and/or negligent conduct that fails to comply with the laws of the FDA and other similar foreign regulatory bodies, provide true, complete and accurate information to the FDA and other similar foreign regulatory bodies, comply with manufacturing standards we have established, comply with healthcare fraud and abuse laws in the United States and similar foreign fraudulent misconduct laws or report financial information or data accurately or to disclose unauthorized activities to us. If we obtain FDA approval of any of our product candidates and begin commercializing those products in the United States, our potential exposure under such laws will increase significantly, and our costs associated with compliance with such laws are also likely to increase. These laws may impact, among other things, our current activities with principal investigators and research patients, as well as proposed and future sales, marketing and education programs.
Our limited operating history may make it difficult to evaluate the success of our business to date and to assess our future viability.
Since inception in 2015, BiomX Ltd. has devoted substantially all of its resources to developing product candidates with phage technology through its preclinical programs, building its intellectual property portfolio, developing a supply chain, planning its business, raising capital and providing general and administrative support for these operations. We have not yet demonstrated our ability to successfully complete any clinical study or other pivotal clinical trials, obtain regulatory approvals, manufacture a commercial-scale product, or arrange for a third-party to do so on our behalf, or conduct sales and marketing activities necessary for successful product commercialization. Consequently, any predictions made about our future success or viability may not be as accurate as they could be if we had a longer operating history.
In addition, as an early-stage company, we may encounter unforeseen expenses, difficulties, complications, delays and other known and unknown circumstances. As we advance our product candidates, we will need to transition from a company with a research focus to a company capable of supporting clinical development and, if successful, commercial activities. We may not be successful in such a transition.
We may need to grow the size of our organization and may experience difficulties in managing this growth.
As our research, development, manufacturing and commercialization plans and strategies, we may need additional managerial, operational, sales, marketing, financial and other personnel. Future growth would impose significant added responsibilities on members of management, including:
| ● | identifying, recruiting, compensating, integrating, maintaining and motivating additional employees; |
| ● | managing our internal research and development efforts effectively, including identification of clinical candidates, scaling our manufacturing process and navigating the clinical and FDA review process for our product candidates; and |
| ● | improving our operational, financial and management controls, reporting systems and procedures. |
Our future financial performance and our ability to commercialize our product candidates will depend, in part, on our ability to effectively manage any future growth, and our management may also have to divert a disproportionate amount of our attention away from day-to-day activities in order to devote a substantial amount of time to managing these growth activities.
If we are not able to effectively expand our organization by hiring additional employees and expanding our groups of consultants and contractors, we may not be able to successfully implement the tasks necessary to further develop and commercialize our product candidates and, accordingly, may not achieve our research, development and commercialization goals.
42
Risks Related to Government Regulation
Our product candidates are subject to significant regulatory approval requirements, which could delay, prevent or limit our ability to market or develop our product candidates.
Our research and development activities, preclinical studies, clinical trials and the anticipated manufacturing and marketing of our drug product candidates are subject to extensive regulation by the FDA and other regulatory agencies in the United States and by comparable authorities in Europe and elsewhere. To satisfy FDA or equivalent foreign regulatory approval standards, we must demonstrate in adequate and well controlled clinical trials that our drug product candidates are safe and effective for their intended use. The regulatory approval process is expensive and time-consuming, and the timing of receipt of regulatory approval is difficult to predict. Given the uncertainties around phage therapy, our product candidates could require a significantly longer time to gain regulatory approval than expected or may never gain approval. We cannot be certain that, even after expending substantial time and financial resources, we will obtain regulatory approval for any of our product candidates. A delay or denial of regulatory approval could delay or prevent our ability to generate product revenue and to achieve profitability.
Regulatory requirements for development of our product candidates are uncertain and evolving. Changes in these laws or the current interpretation or application of these laws would have a significant adverse impact on our ability to develop and commercialize our product candidates. The legal and regulatory status of phage therapy remains unclear in many countries, including the European Union. Changes in regulatory approval policies during the development period of any of our product candidates, changes in, or the enactment of, additional regulations or statutes, or changes in regulatory review practices for a submitted product application may cause a delay in obtaining approval or result in the rejection of an application for regulatory approval.
Regulatory approval, if obtained, may be made subject to limitations on the indicated uses for which we may market a product, as well as the approved labeling for the product. These limitations could adversely affect our potential product revenue. Regulatory approval may also be conditioned on costly post-marketing follow-up studies. In addition, the labeling, packaging, adverse event reporting, storage, advertising, promotion and recordkeeping related to the product will be subject to extensive ongoing regulatory requirements. Furthermore, for any marketed product, our manufacturer and our manufacturing facilities will be subject to registration and listing requirements and continual review and periodic inspections by the FDA or other regulatory authorities. Failure to comply with applicable regulatory requirements may, among other things, result in fines, suspensions of regulatory approvals, product recalls, product seizures, operating restrictions and criminal prosecution.
Breakthrough Therapy Designation or Fast Track Designation by the FDA, even if granted for any of our product candidates developed for therapeutic indications, may not lead to a faster development, regulatory review or approval process, and it does not increase the likelihood that any of our product candidates will receive marketing approval in the United States.
In the United States, we may seek a Breakthrough Therapy Designation for some of our product candidates, including BX004 or another product candidate under development. A breakthrough therapy is defined as a therapy that is intended, alone or in combination with one or more other therapies, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the therapy may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. For therapies that have been designated as breakthrough therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Breakthrough designation also provides sponsors with the potential for rolling review of a BLA. Designation as a breakthrough therapy is within the discretion of the FDA.
In the European Union, the PRIME (PRIority MEdicines) status is similar to the Breakthrough Therapy Designation. The EMA has implemented the PRIME status to support the development and accelerate the approval of complex, innovative medicinal products addressing an unmet medical need. The PRIME status enables early dialogue with the relevant EMA scientific committees and, possibly, some payors and thus reinforces the EMA’s scientific and regulatory support. The PRIME status, which is granted at the EMA’s discretion, focuses on medicinal products the marketing authorization of which qualifies for accelerated assessment (medicinal products of major interest from a public health perspective, in particular from a therapeutic innovation perspective).
Accordingly, even if we believe one of our product candidates meets the criteria for designation as a breakthrough therapy or for PRIME status, the FDA or EMA, respectively, may disagree and instead determine not to make such designation. In any event, the receipt of a Breakthrough Therapy Designation or PRIME status for a product candidate may not actually result in a faster development process, review or approval compared to therapies considered for approval under conventional procedures and does not assure ultimate approval. In addition, even if one or more of our product candidates qualify as breakthrough therapies or is granted PRIME status, the FDA or EMA, respectively, may later decide that such product candidates no longer meet the conditions for qualification or decide that the time period for review or approval will not be shortened.
In the United States, we may seek Fast Track Designation for some of our product candidates for therapeutic indications. If a therapy is intended for the treatment of a serious or life-threatening condition and the therapy demonstrates the potential to address unmet medical needs for this condition, the therapy sponsor may apply for Fast Track Designation. The FDA has broad discretion whether or not to grant this designation, so even if we believe a particular product candidate is eligible for this designation; we cannot assure you that the FDA would decide to grant it. In August 2023, we obtained Fast Track Designation for BX004 in the United States. Although we received Fast Track Designation, we may not experience a faster development process, review or approval compared to conventional FDA procedures. The FDA may withdraw Fast Track Designation if they believe that the designation is no longer supported by data from our clinical development program. Fast Track Designation alone does not guarantee qualification for the FDA’s priority review procedures.
43
Other countries may have adopted schemes designed to ensure an accelerated approval of drugs that are especially important for patients. For example, in the European Union, the EMA may agree to an accelerated assessment (150 days instead of 210 days) for medicinal products of major interest from a public health perspective, in particular from a therapeutic innovation perspective). Furthermore, competent regulatory authorities may grant market authorizations “under exceptional circumstances,” in cases where all the required safety and efficacy data have not been and will not be collected, to medicinal products designed for unmet needs or orphan medicinal products. Although a marketing authorization under exceptional circumstances is definitive, the risk-benefit balance of the medicinal product must be reviewed annually and the marketing authorization is withdrawn if it becomes negative. Moreover, under the centralized procedure, the European Commission may grant “conditional marketing authorizations” in cases where all the required safety and efficacy data are not yet available. The conditional marketing authorization is subject to conditions to be fulfilled for generating the missing data or ensuring increased safety measures. It is valid for one year and has to be renewed annually until fulfillment of all the conditions. If the conditions are not fulfilled within the timeframe set by the EMA, the marketing authorization ceases to be renewed. As with Fast Track Designation, the competent regulatory authorities in the European Union have broad discretion whether or not to grant such an accelerated assessment or approval and, even if such assessment or approval is granted, we may not experience a faster development process, review or approval compared to conventional procedures.
We may fail to obtain and maintain orphan drug designations from the FDA or equivalent foreign regulatory agencies for our current and future therapeutic product candidates, as applicable.
In the United States, under the Orphan Drug Act, the FDA may grant orphan drug designation to a drug or biologic intended to treat a rare disease or condition, which is defined as one occurring in a patient population of fewer than 200,000 in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the drug or biologic will be recovered from sales in the United States. In December 2023, we obtained orphan drug designation for BX004 in the United States. In the United States, the orphan drug designation entitles a party to financial incentives, such as opportunities for grant funding toward clinical trial costs, tax advantages and user-fee waivers. In addition, if a product that has the orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications, including an NDA, to market the same drug or biologic for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity or where the original manufacturer is unable to assure sufficient product quantity.
In addition, exclusive marketing rights in the United States may be limited if we seek approval for an indication broader than the orphan-designated indication or may be lost if the FDA later determines that the request for designation was materially defective, or if we are unable to assure sufficient quantities of the product to meet the needs of patients with the orphan-designated disease or condition. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties may receive and be approved for the same condition. Even after an orphan-designated product is approved, the FDA can subsequently approve a later drug with the same active moiety for the same condition if the FDA concludes that the later drug is clinically superior if it is shown to be safer, more effective or makes a major contribution to patient care. Orphan drug designation neither shortens the development time or regulatory review time of a drug nor gives the drug any advantage in the regulatory review or approval process. In addition, while we may seek the orphan drug designation for our product candidates, we may never receive such designation.
An orphan drug legal regime also exists in the European Union. The EMA’s Committee for Orphan Medicinal Products, or COMP, gives opinions, and the European Commission takes decisions, on the granting of the orphan drug designation to the development of products that are intended for the diagnosis, prevention or treatment of (i) a life-threatening or chronically debilitating condition affecting not more than five in 10,000 persons in the European Economic Area (comprising the European Union, Iceland, Liechtenstein and Norway); or (ii) a life-threatening, seriously debilitating or serious and chronic condition when, without incentives, it is unlikely that sales of the drug in the European Economic Area would be sufficient to justify the necessary investment in developing the drug or biological product. The granting of the orphan designation requires that there is no satisfactory method of diagnosis, prevention or treatment, or, if such a method exists, that the future medicine is to be of significant benefit to those affected by the condition. The test for that later condition is stringent, because the future product must be compared with all existing therapies for the rare condition, including surgical operations, already authorized medicinal products and compounded preparations (subject to certain conditions). At the time of marketing authorization, the orphan designation is reviewed again by the COMP in view of the maintenance of the orphan status. If the designation criteria are no longer met, the European Commission withdraws the orphan designation. Maintenance of the orphan designation at the time of marketing authorization means that all the drugs/biologicals authorized since the granting of the designation become relevant for determining the lack of satisfactory therapy or the significant benefit.
If obtained, the orphan drug designation would entitle us to financial incentives, such as reductions of fees or fee waivers and 10 years of market exclusivity. Market exclusivity precludes the EMA or the national competent authorities from validating a marketing authorization application, and the European Commission or a national competent authority from granting a marketing authorization, for a same or similar drug/biological and the same therapeutic indication. The 10-year period may be reduced to six years if the orphan designation criteria are no longer met, including where it is shown that the product is not sufficiently profitable to justify maintenance of market exclusivity. The orphan exclusivity may also be lost vis-à-vis another drug/biological in cases where the manufacturer is unable to assure sufficient quantity of the drug to meet patient needs or if that other product is proved to be clinically superior to the approved orphan product. A drug/biological is clinically superior if it is safer, more effective or makes a major contribution to patient care.
44
Failure to comply with health and data protection laws and regulations could lead to claims, government enforcement actions (which could include civil or criminal penalties), regulatory actions, private litigation and/or adverse publicity and could negatively affect our operating results and business.
We may be subject to federal, state and foreign data protection laws and regulations (i.e., laws and regulations that address privacy and security). In the United States, numerous federal and state laws and regulations, including federal health information privacy laws, state consumer privacy laws, state data breach notification laws, state health information privacy laws and federal and state consumer protection laws (e.g., Section 5 of the Federal Trade Commission Act), that govern the collection, use, disclosure and protection of health-related and other personal information could apply to our operations or the operations of our collaborators. In addition, we may obtain health information from third parties (including research institutions from which we obtain clinical trial data) that are subject to privacy and security requirements under HIPAA, as amended by the Health Information Technology for Economic and Clinical Health of 2009. Depending on the facts and circumstances, we could be subject to criminal penalties if we knowingly obtain, use or disclose individually identifiable health information maintained by a HIPAA-covered entity in a manner that is not authorized or permitted by HIPAA.
Additional requirements may also be imposed by international data protection laws. In this context, Regulation 2016/679 of the GDPR (in addition to many other international data protection laws) may have an impact on our operations when we collect and/or process personal data of individuals located in the European Union. The GDPR has applied since May 25, 2018 (replacing previously applicable data protection frameworks) and has an extraterritorial reach. The GDPR allows members states to introduce specific requirements in relation to certain areas, including processing of special categories of data, and we may face further restrictions and non-compliance risks under such national frameworks. We have not yet assessed whether its activities might be caught by the GDPR.
Because of the types of data we collect and process, which may involve health, biometric and genetic data, we may face high risks for non-compliance with the GDPR rules (or local declinations of GDPR-rules across the different European Union Member States), as these types of data are considered as special categories of data and are granted higher protection. The risks are further increased considering the diverging approach in the European Union as to the rules, requirements and frameworks in relation to the processing of personal data in clinical trials (in matters such as the choice of the legal basis for the processing of data, the possible uses of the personal data collected, etc.) and the interplay with other relevant frameworks. The GDPR introduced stringent data protection requirements in the European Union, as well as potential fines for noncompliant companies of up to the greater of €20 million or 4% of annual worldwide turnover. Supervisory authorities also have the ability to restrict our processing activities if those are deemed not to be in compliance with the GDPR (or local declinations); this may significantly impact the way we conduct our activities. The GDPR imposes numerous requirements for the collection, use and disclosure of personal data, including high standards for consent to be valid, and specific information to be provided to individuals about how their personal data is used, the obligation to notify regulators and (in some cases) to communicate to affected individuals of personal data breaches, extensive new internal privacy governance requirements and obligations to allow individuals to exercise their strengthened privacy rights (e.g., the right to access, correct and delete their personal data, to withdraw their consent, etc.), and obligations when contracting with third parties such as service providers, CROs, etc. In addition, the GDPR includes restrictions on data transfers outside the EEA. The actual mechanisms made available under GDPR to transfer such personal data have received heightened regulatory and judicial scrutiny. If we cannot rely on existing mechanisms for transferring personal data from the EEA, the United Kingdom, or other jurisdictions, we may be unable to transfer personal data in those regions. Further, the United Kingdom’s vote in favor of exiting the European Union, often referred to as “Brexit,” has created uncertainty as to whether or not the United Kingdom data protection legislation will depart from the GDPR and how data transfers to and from the United Kingdom will be regulated.
45
Compliance with U.S. and international data protection laws and regulations could require us to take on more onerous obligations in our contracts, restrict our ability to collect, use and disclose data, or in some cases, impact our ability to operate in certain jurisdictions. Such laws and regulations could limit our ability to use and share personal or other data, thereby increasing our costs and harming our business and financial condition. Failure to comply with U.S. and international data protection laws and regulations could result in claims, government enforcement actions (which could include civil or criminal penalties), regulatory actions, private litigation and/or adverse publicity and could negatively affect our operating results and business. Moreover, clinical trial subjects about whom we or our potential collaborators obtain information, as well as the providers who share this information with us, may contractually limit our ability to use and disclose the information. Claims that we have violated individuals’ privacy rights, failed to comply with data protection laws, or breached our contractual obligations, even if we are not found liable, could be expensive and time consuming to defend and could result in adverse publicity that could harm our business. Finally, we may be required to disclose personal data pursuant to demands from government agencies, from law enforcement agencies, and from intelligence agencies. This disclosure may result in a failure or perceived failure by us to comply with data privacy laws, rules, and regulations and could result in proceedings or actions against us in the same or other jurisdictions, and could have an adverse impact on our reputation and brand.
Our relationships with healthcare providers, physicians and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings.
Healthcare providers, physicians and third-party payors in the United States and elsewhere play a primary role in the recommendation and prescription of pharmaceutical products. Arrangements with third-party payors and customers can expose pharmaceutical manufacturers to broadly applicable fraud and abuse and other healthcare laws and regulations, including, without limitation, the federal Anti-Kickback Statute and the FCA, and foreign equivalent legislation, which may constrain the business or financial arrangements and relationships through which such companies sell, market and distribute pharmaceutical products. In particular, the promotion, sales and marketing of healthcare items and services, as well as certain business arrangements in the healthcare industry, are subject to extensive laws designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, structuring and commissions, certain customer incentive programs and other business arrangements generally. Activities subject to these laws also involve the improper use of information obtained in the course of patient recruitment for clinical trials. The applicable federal, state and foreign healthcare laws and regulations laws that may affect our ability to operate include, but are not limited to:
| ● | the federal Anti-Kickback Statute, which prohibits, among other things, knowingly and willfully soliciting, receiving, offering or paying any remuneration (including any kickback, bribe or rebate), directly or indirectly, overtly or covertly, in cash or in kind, to induce, or in return for, either the referral of an individual, or the purchase, lease, order or recommendation of any good, facility, item or service for which payment may be made, in whole or in part, under a federal healthcare program, such as the Medicare and Medicaid programs. A person or entity can be found guilty of violating the statute without actual knowledge of the statute or specific intent to violate it. In addition, a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the FCA. The Anti-Kickback Statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers and formulary managers on the other hand. There are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution; | |
| ● | federal civil and criminal false claims laws, including the FCA, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, false or fraudulent claims for payment to, or approval by Medicare, Medicaid or other federal healthcare programs, knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim or an obligation to pay or transmit money to the federal government, or knowingly concealing or knowingly and improperly avoiding or decreasing or concealing an obligation to pay money to the federal government. Manufacturers can be held liable under the FCA even when they do not submit claims directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims. The FCA also permits a private individual acting as a “whistleblower” to bring actions on behalf of the federal government alleging violations of the FCA and to share in any monetary recovery; |
46
| ● | HIPAA, which created new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity can be found guilty of violating HIPAA without actual knowledge of the statute or specific intent to violate it; | |
| ● | the federal Physician Payment Sunshine Act, created under the Patient Protection and Affordable Care Act and its implementing regulations, which require manufacturers of drugs, devices, biologicals and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report annually to the United States Department of Health and Human Services information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician practitioners (physician assistants, nurse practitioners, clinical nurse specialists, anesthesiologist assistants, certified registered nurse anesthetists and certified nurse midwives) and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; | |
| ● | federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers; | |
| ● | analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by nongovernmental third-party payors, including private insurers, and may be broader in scope than their federal equivalents; state and foreign laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government or otherwise restrict payments that may be made to healthcare providers; and state and foreign laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; and | |
| ● | European Union and other foreign provisions. |
The distribution of pharmaceutical products is subject to additional requirements and regulations, including extensive recordkeeping, licensing, storage, security requirements intended to prevent the unauthorized sale of pharmaceutical products and, in some foreign countries, including the European Union countries, mandatory anti-counterfeit features.
47
The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of healthcare reform, especially in light of the lack of applicable precedent and regulations. Federal and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry. Ensuring business arrangements comply with applicable healthcare laws, as well as responding to possible investigations by government authorities, can be time- and resource-consuming and can divert a company’s attention from the business.
It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent inappropriate conduct may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. Efforts to ensure that our business arrangements will comply with applicable healthcare laws may involve substantial costs. It is possible that governmental and enforcement authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws and regulations. The failure to comply with any of these laws or regulatory requirements could subject us to possible legal or regulatory action. Depending on the circumstances, failure to meet applicable regulatory requirements can result in civil, criminal and administrative penalties, damages, fines, disgorgement, individual imprisonment, possible exclusion from participation in federal and state funded healthcare programs, contractual damages and the curtailment or restricting of our operations, as well as additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of noncompliance with these laws. Any action for violation of these laws, even if successfully defended, could cause a pharmaceutical manufacturer to incur significant legal expenses and divert management’s attention from the operation of the business. Prohibitions or restrictions on sales or withdrawal of future marketed products could materially affect business in an adverse way.
In addition, the approval and commercialization of any of our product candidates outside the United States will also likely subject us to foreign equivalents of the healthcare laws mentioned above, among other foreign laws.
The FDA and other equivalent foreign regulatory agencies may implement additional regulations or restrictions on the development and commercialization of products which act on the microbiome, which may be difficult to predict.
The FDA and equivalent foreign regulatory agencies in other countries have each expressed interest in further regulating biotechnology products and product candidates, such as those that act on the human microbiome. Agencies at both the federal and state level in the United States, as well as the U.S. congressional committees and other governments or governing agencies, have also expressed interest in further regulating the biotechnology industry. Such action may delay or prevent commercialization of some or all of our product candidates. Adverse developments in non-IND human clinical studies or clinical trials of microbiome products conducted by others may cause the FDA or other oversight bodies to change the requirements for approval of any of our product candidates. These regulatory review agencies and committees and the new requirements or guidelines they promulgate may lengthen the regulatory review process, require us to perform additional studies or trials, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of our product candidates or lead to significant post-approval limitations or restrictions. As we advance our product candidates, we will be required to consult with these regulatory agencies and comply with applicable requirements and guidelines. If we fail to do so, we may be required to delay or discontinue development of such product candidates. These additional processes may result in a review and approval process that is longer than we otherwise would have expected. Delays as a result of an increased or lengthier regulatory approval process or further restrictions on the development of our product candidates can be costly and could negatively impact our ability to complete clinical trials and commercialize our current and future product candidates in a timely manner if at all.
Even if we receive regulatory approval of any product candidates for therapeutic indications, we will be subject to ongoing regulatory compliance obligations and continued regulatory review, which may result in significant additional expense. Additionally, any of our product candidates, if approved, could be subject to labeling and other restrictions and market withdrawal, and we may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our product candidates.
If any of our product candidates is approved for therapeutic indications, we will be subject to ongoing regulatory requirements for manufacturing, labeling, packaging, storage, distribution, advertising, promotion, sampling, recordkeeping, export, import, conduct of post-marketing studies and submission of safety, efficacy and other post-market information, including both federal and state requirements in the United States and requirements of equivalent foreign regulatory agencies. In addition, we will be subject to continued compliance with cGMP and GCP requirements for any clinical trials that we conduct post-approval.
48
Manufacturers and manufacturers’ facilities are required to comply with extensive FDA and equivalent foreign regulatory agency requirements, including ensuring that quality control and manufacturing procedures conform to cGMP regulations. As such, we and our contract manufacturers will be subject to continual review and inspections to assess compliance with cGMP and adherence to commitments made in any NDA, other marketing applications and previous responses to inspection observations. Accordingly, we and others with whom we work must continue to expend time, money, and effort in all areas of regulatory compliance, including manufacturing, production and quality control.
The FDA or equivalent foreign regulatory agencies have significant post-marketing authority, including, for example, the authority to require labeling changes based on new safety information and to require post-marketing studies or clinical trials to evaluate serious safety risks related to the use of a drug. Any regulatory approvals that we receive for our product candidates may be subject to limitations on the approved indicated uses for which the product may be marketed or to the conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase 4 clinical trials and surveillance to monitor the safety and efficacy of the product candidate. The FDA or equivalent foreign regulatory agencies may also require a REMS program as a condition of approval of our product candidates, which could entail requirements for long-term patient follow-up, a medication guide, physician communication plans or additional elements to ensure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. In addition, if the FDA or an equivalent foreign regulatory agency approves our product candidates, we will have to comply with requirements, including submissions of safety and other post-marketing information and reports and registration.
The FDA or equivalent foreign regulatory agencies may impose consent decrees or withdraw approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with our product candidates, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements may result in revisions to the approved labeling to add new safety information, the imposition of post-market studies or clinical trials to assess new safety risks, or the imposition of distribution restrictions or other restrictions under a REMS program. Other potential consequences include, among other things:
| ● | restrictions on the marketing or manufacturing of our products, withdrawal of products from the market, or voluntary or mandatory product recalls; |
| ● | fines, warning or untitled enforcement letters, or holds on clinical trials; |
| ● | refusal by the FDA or equivalent foreign regulatory agencies to approve pending applications or supplements to approved applications filed by us or the suspension or revocation of license approvals; |
| ● | product seizure or detention or refusal to permit the import or export of our product candidates; and |
| ● | injunctions or the imposition of civil or criminal penalties. |
The FDA or equivalent foreign regulatory agencies strictly regulate the marketing, labeling, advertising and promotion of drug products that are placed on the market. Products may be promoted only for the approved indications and in accordance with the provisions of the approved label or other regulatory marketing pathway. The FDA and equivalent foreign regulatory agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. The policies of the FDA or equivalent foreign regulatory agencies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, which would adversely affect our business, prospects and the ability to achieve or sustain profitability.
49
The policies of the FDA or equivalent foreign regulatory agencies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. We also cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may be subject to enforcement action, and we may not achieve or sustain profitability.
Noncompliance by us or any future collaborator with regulatory requirements, including safety monitoring or pharmacovigilance requirements, can also result in significant financial penalties.
We may conduct clinical trials for our product candidates outside the United States, and the FDA may not accept data from such trials.
We have and may continue to conduct certain clinical trials or a portion of our clinical trials for our product candidates outside the U.S. The acceptance of study data from clinical trials conducted outside the U.S. or another jurisdiction by the FDA or comparable foreign regulatory authority may be subject to certain conditions or may not be accepted at all. In cases where data from foreign clinical trials are intended to serve as the sole basis for marketing approval in the U.S., the FDA will generally not approve the application on the basis of foreign data alone unless (i) the data are applicable to the U.S. population and U.S. medical practice; (ii) the trials were performed by clinical investigators of recognized competence and pursuant to GCP regulations; and (iii) the data may be considered valid without the need for an on-site inspection by the FDA, or if the FDA considers such inspection to be necessary, the FDA is able to validate the data through an on-site inspection or other appropriate means. In addition, even where the foreign study data are not intended to serve as the sole basis for approval, the FDA will not accept the data as support for an application for marketing approval unless the study is well-designed and well-conducted in accordance with GCP requirements and the FDA is able to validate the data from the study through an onsite inspection if deemed necessary. Many foreign regulatory authorities have similar approval requirements. In addition, such foreign trials would be subject to the applicable local laws of the foreign jurisdictions where the trials are conducted. There can be no assurance that the FDA or any comparable foreign regulatory authority will accept data from trials conducted outside of the U.S. or the applicable jurisdiction. If the FDA or any comparable foreign regulatory authority does not accept such data, it would result in the need for additional trials, which could be costly and time-consuming, and which may result in current or future product candidates that we may develop not receiving approval for commercialization in the applicable jurisdiction.
Any products that we may develop may become subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, which could make it difficult for us to sell any product candidates or therapies profitably.
The regulations that govern pricing for new medical products vary widely from country to country. As a result, we might obtain regulatory approval for a product in a particular country but then be subject to pricing regulations in that country that delay the commercial launch of the product and negatively impact the revenue we are able to generate from the sale of the product in that country. In addition, our ability to commercialize any approved products successfully will depend in part on the extent to which reimbursement for these products will be available from government health administration authorities, private health insurers and other organizations. Even if we succeed in bringing one or more therapeutic products to market, these products may not be considered cost-effective, and the amount reimbursed for any products may be insufficient to allow us to sell them on a competitive basis. If the price we are able to charge for therapeutic products is inadequate in light of our development and other costs, our future profitability could be adversely affected.
Ongoing health care legislative and regulatory reform measures may have a material adverse effect on our business and results of operations.
Changes in regulations, statutes or the interpretation of existing regulations could impact our business in the future by requiring, for example, (i) changes to our manufacturing arrangements, (ii) additions or modifications to product labeling, (iii) the recall or discontinuation of our products, or (iv) additional record-keeping requirements. If any such changes were to be imposed, they could adversely affect the operation of our business.
In the United States, there have been and continue to be a number of legislative initiatives to contain health care costs. For example, in March 2010, the ACA was passed, which substantially changed the way health care is financed by both governmental and private insurers and significantly impacted the United States pharmaceutical industry. The ACA, among other things, subjected biological products to potential competition by lower-cost biosimilars; addressed a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; increased the minimum Medicaid rebates owed by manufacturers under the Medicaid Drug Rebate Program; and extended the rebate program to individuals enrolled in Medicaid managed care organizations. It also established annual fees and taxes on manufacturers of certain branded prescription drugs and creates a new Medicare Part D coverage gap discount program in which manufacturers must now agree to offer 50% point of sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D.
50
Since its enactment, there have been judicial, executive and Congressional challenges to certain aspects of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA without specifically ruling on the constitutionality of the ACA. Prior to the Supreme Court’s decision, President Biden issued an executive order initiating a special enrollment period from February 15, 2021 through August 15, 2021 for purposes of obtaining health insurance coverage through the ACA marketplace. The executive order also instructed certain governmental agencies to review and reconsider their existing policies and rules that limit access to healthcare. More recently, on March 11, 2021, President Biden signed the American Rescue Plan Act of 2021 into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug’s average manufacturer price, beginning January 1, 2024. It is unclear how other healthcare reform measures of the Biden administration, if any, will impact our business.
These laws and future state and federal health care reform measures may be adopted in the future, any of which may result in additional reductions in Medicare and other health care funding and otherwise affect the prices we may obtain for any of our product candidates for which we may obtain regulatory approval or the frequency with which any such product candidate is prescribed or used.
A similar movement is observed in the European Union countries. Criteria for pricing and reimbursement, which vary from country to country, are regularly amended and tightened in order to reduce the draw on the budget allocated to national health insurance systems. Moreover, the system of reference pricing (the price in a country calculated on the basis of prices in other countries with typically lower prices) leads to price reductions in countries that traditionally granted high prices.
Disruptions at the FDA and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire, retain or deploy key leadership and other personnel, or otherwise prevent new or modified products from being developed, approved or commercialized in a timely manner or at all, which could negatively impact our business.
The ability of the FDA to review and or approve new products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy changes, the FDA’s ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect the FDA’s ability to perform routine functions. Average review times at the FDA have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable. Disruptions at the FDA and other regulatory authorities may also slow the time necessary for new drugs and biologics to be reviewed and/or approved by necessary regulatory authorities, which would adversely affect our business. For example, over the last several years, the U.S. government has shut down several times and certain regulatory authorities, such as the FDA, have had to furlough critical FDA employees and stop critical activities.
We are subject to certain U.S. and foreign anticorruption, anti-money laundering, export control, sanctions and other trade laws and regulations. We can face serious consequences for violations.
Among other matters, U.S. and foreign anticorruption, anti-money laundering, export control, sanctions and other trade laws and regulations, which are collectively referred to as Trade Laws, prohibit companies and their employees, agents, clinical research organizations, legal counsel, accountants, consultants, contractors and other partners from authorizing, promising, offering, providing, soliciting or receiving, directly or indirectly, corrupt or improper payments or anything else of value to or from recipients in the public or private sector. Violations of Trade Laws can result in substantial criminal fines and civil penalties, imprisonment, the loss of trade privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences. We have direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities and other organizations. We also expect our non-U.S. activities to increase over time. We plan to engage third parties for clinical trials and/or to obtain necessary permits, licenses, patent registrations and other regulatory approvals, and we can be held liable for the corrupt or other illegal activities of our personnel, agents or partners, even if we do not explicitly authorize or have prior knowledge of such activities.
51
Risks Related to our Licensed and Co-Owned Intellectual Property
The license agreements we maintain, including the Yeda 2015 License Agreement, with Yeda are important to our business. If we or the other parties to our license agreements fail to adequately perform under the license agreements, or if we or they terminate the license agreements, the development, testing, manufacture, production and sale of our phage-based therapeutic product candidates would be delayed or terminated, and our business would be adversely affected.
The Yeda 2015 License Agreement provides for an exclusive worldwide license to certain know-how and research information related to the development, testing, manufacture, production and sale of phage-based therapeutic product candidates, including candidates specified in the agreement, which are used in our phage discovery platform, as well as patents, research and other rights to phage product candidates resulting from the work of the consultants identified in the agreement and further research that we funded. The Yeda 2015 License Agreement terminates upon the later of the expiration of the last of the patents covered under the Yeda 2015 License Agreement and the expiry of a continuous 15-year period during which there has not been a first commercial sale of any product in any country. Yeda may also terminate the agreement if we fail to observe certain diligence and development requirements and milestones as described in the Yeda 2015 License Agreement. We or Yeda may terminate the agreement for the material uncured breach of the other party after a notice period or the other party’s winding up, bankruptcy, insolvency, dissolution or other similar discontinuation of business. Upon termination of the agreement, other than due to the passage of time, we are required to grant to Yeda a nonexclusive, irrevocable, perpetual, fully paid-up, sublicensable, worldwide license in respect of our rights in know-how and research results as described in the Yeda 2015 License Agreement, provided that, if Yeda subsequently grants a license to a third party that utilizes our rights, we are entitled to share in the net proceeds actually received by Yeda arising out of that license, subject to a cap based on the development expenses that we incur in connection with the Yeda 2015 License Agreement. For more information on the Yeda 2015 License Agreement, see “Business—Material Agreements—License Agreements—License Agreement with Yeda.”
Termination of our license agreements could cause significant delays in our product and commercialization efforts that could prevent us from commercializing our product candidates, including our phage-based therapeutic product candidates, without first expanding our internal capabilities or entering into other agreements with third parties. Any alternative collaboration or license could also be on less favorable terms to us.
We are highly dependent on intellectual property licensed from third parties, and termination or limitation of any of these licenses could result in the loss of significant rights and materially harm our business.
We currently rely on licenses from third-party collaborators for certain aspects of our technology and for certain of our existing programs. In particular, we received exclusive, royalty-bearing licenses to certain patents held by third parties, including Yeda. The Yeda 2015 License Agreement provide license to certain know-how and research information related to the development, testing, manufacture, production and sale of phage-based therapeutic product candidates that are used in our phage discovery platform, as well as patents, research and other rights to phage product candidates resulting from the work of the consultants identified in the agreement and further research that we funded.
If we fail to comply with our obligations under our license agreements, including payment terms, our licensors may have the right to terminate our license agreements, in which event we may not be able to develop, manufacture, market or sell the products covered by those license agreements. We may also face other penalties under our license agreements if we do not meet our contractual obligations. Such an occurrence could materially adversely affect the value of our products being developed under any such license agreements. Termination of one or more of our license agreements, or reduction or elimination of our rights under these license agreements, may result in us having to negotiate new or reinstated license agreements, which may not be available to us on equally favorable terms, or at all, which may mean we are unable to commercialize the affected product candidates.
In the future, we may rely upon additional licenses to certain patent rights and proprietary technology from third parties that are important or necessary to the development of our product candidates and proprietary product platform. Patent rights that we in-license in the future may be subject to a reservation of rights by one or more third parties. As a result, any such third party may have certain rights to such intellectual property.
52
In addition, subject to the terms of any such license agreements, we may not have the right to control the preparation, filing, prosecution and maintenance, and we may not have the right to control the enforcement and defense, of patents and patent applications covering the technology that we license from third parties. We cannot be certain that our in-licensed patent applications (and any patents issuing therefrom) that are controlled by our licensors will be prepared, filed, prosecuted, maintained, enforced and defended in a manner consistent with the best interests of our business. If our licensors fail to prosecute, maintain, enforce and defend such patents rights, or lose rights to those patent applications (or any patents issuing therefrom), the rights we have licensed may be reduced or eliminated, our right to develop and commercialize any of our product candidates and proprietary product platform technology that are subject of such licensed rights could be adversely affected, and we may not be able to prevent competitors from making, using and selling competing products. Moreover, we cannot be certain that such activities by our potential future licensors will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents or other intellectual property rights. In addition, even where we may have the right to control the prosecution of patents and patent applications that we may license to and from third parties, we may still be adversely affected or prejudiced by actions or inactions of our potential future licensees, licensors and their counsel that took place prior to the date of assumption of control over patent prosecution.
The patent position of biopharmaceutical companies, including ours and our licensors’, is generally uncertain and involves complex legal and factual considerations and, therefore, validity and enforceability cannot be predicted with certainty. Our licensed and co-owned intellectual property may be challenged, deemed unenforceable, invalidated or circumvented. We and our licensors will be able to protect our intellectual property rights from unauthorized use by third parties only to the extent that these rights (and the products and services they cover) are protected by valid and enforceable patents, copyrights or trademarks, or are effectively maintained as trade secrets.
Any patents obtained by our licensors or us, may be challenged by re-examination or otherwise invalidated or eventually found unenforceable. Both the patent application process and the process of managing patent disputes can be time consuming and expensive. If we or one of our licensors were to initiate legal proceedings against a third party to enforce a patent relating to one of our products, the defendant in such litigation could counterclaim that the asserted patents are invalid and/or unenforceable. In patent litigation in the U.S., defendant counterclaims alleging invalidity or unenforceability are common, as are validity challenges by the defendant against the subject patent or related patents before the USPTO. Grounds for a validity challenge could be an alleged failure to meet any of several statutory patentability requirements, including lack of novelty, obviousness, non-enablement, failure to meet the written description requirement, indefiniteness, and/or failure to claim patentable subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected to prosecution of the patent/s at issue intentionally withheld material information from the USPTO or made a misleading statement during prosecution. Additional grounds for an unenforceability assertion include an allegation of misuse or anticompetitive use of patent rights, and an allegation of incorrect inventorship with deceptive intent. Third parties may also raise similar claims before the USPTO, even outside the context of litigation. The outcome of any assertion of invalidity and/or unenforceability is unpredictable. If a defendant or third party were to prevail on a legal assertion of invalidity and/or unenforceability, we and our licensors would lose at least part, and perhaps all, of the claims of the challenged patent/s. Such a loss of patent protection could have a material adverse impact on our business.
We are dependent on patents and proprietary technology. If we fail to adequately protect this intellectual property or if we otherwise do not have exclusivity for the marketing of our products, our ability to commercialize products could suffer.
Our commercial success will depend in part on our ability to obtain and maintain patent protection sufficient to prevent others from marketing our product candidates, as well as to defend and enforce these patents against infringement and to operate without infringing the proprietary rights of others. Protection of our product candidates from unauthorized use by third parties will depend on having valid and enforceable patents that cover our product candidates or their manufacture or use or on having effective trade secret protection. If our patent applications do not result in issued patents or if our patents are found to be invalid, we will lose the ability to exclude others from making, using or selling the inventions claimed therein. We have a limited number of patents and pending patent applications.
53
The patent positions of biotechnology companies can be uncertain and involve complex legal and factual questions. This is due to inconsistent application of policies and changes in policy relating to the examination and enforcement of biotechnology patents to date on a global scale. The laws of some countries may not protect intellectual property rights to the same extent as the laws of countries having well-established patent systems, and those countries may lack adequate rules and procedures for defending our intellectual property rights. Also, changes in either patent laws or in the interpretations of patent laws may diminish the value of our intellectual property. We are not able to guarantee that all of our patent applications will result in the issuance of patents, and we cannot predict the breadth of claims that may be allowed in our patent applications or in the patent applications we may license from others.
The Leahy-Smith America Invents Act provides for proceedings involving post-issuance patent review procedures, such as inter partes review, or IPR, and post-grant review, that allow third parties to challenge the validity of an issued patent in front of the USPTO Patent Trial and Appeal Board. Each proceeding has different eligibility criteria and different patentability challenges that can be raised. IPRs permit any person (except a party who has been litigating the patent for more than a year) to challenge the validity of the patent on the grounds that it was anticipated or made obvious by prior art. Patents covering pharmaceutical products have been subject to attack in IPRs from generic drug companies and from hedge funds. If it is within nine months of the issuance of the challenged patent, a third party can petition the USPTO for post-grant review, which can be based on any invalidity grounds and is not limited to prior art patents or printed publications.
In post-issuance proceedings, USPTO rules and regulations generally tend to favor patent challengers over patent owners. For example, unlike in district court litigation, claims challenged in post-issuance proceedings are given their broadest reasonable meaning, which increases the chance a claim might be invalidated by prior art or lack support in the patent specification. As another example, unlike in district court litigation, there is no presumption of validity for an issued patent, and thus a challenger’s burden to prove invalidity is by a preponderance of the evidence, as opposed to the heightened clear and convincing evidence standard. As a result of these rules and others, statistics released by the USPTO show a high percentage of claims being invalidated in post-issuance proceedings. Moreover, with few exceptions, there is no standing requirement to petition the USPTO for inter partes review or post-grant review. In other words, companies that have not been charged with infringement or that lack commercial interest in the patented subject matter can still petition the USPTO for review of an issued patent. Thus, even where we have issued patents, our rights under those patents may be challenged and ultimately not provide us with sufficient protection against competitive products or processes.
The degree of future protection for our proprietary rights is uncertain, because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
| ● | we might not be the first to file patent applications for our inventions; |
| ● | others may independently develop similar or alternative product candidates to any of our product candidates that fall outside the scope of our patents; |
| ● | our pending patent applications may not result in issued patents; |
| ● | our issued patents may not provide a basis for commercially viable products or may not provide us with any competitive advantages or may be challenged by third parties; |
| ● | others may design around our patent claims to produce competitive products that fall outside the scope of our patents; |
| ● | we may not develop additional patentable proprietary technology related to our product candidates; and |
| ● | we are dependent upon the diligence of our appointed agents in national jurisdictions, acting for and on our behalf, which control the prosecution of pending domestic and foreign patent applications and maintain granted domestic and foreign patents. |
54
An issued patent does not guarantee us the right to practice the patented technology or commercialize the patented product. Third parties may have blocking patents that could be used to prevent us from commercializing our patented products and practicing our patented technology. Our issued patents and those that may be issued in the future may be challenged, invalidated or circumvented, which could limit our ability to prevent competitors from marketing the same or related product candidates or could limit the length of the term of patent protection of our product candidates. Moreover, because of the extensive time required for development, testing and regulatory review of a potential product, it is possible that, before any of our product candidates can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby reducing any advantage of the patent. Patent term extensions may not be available for these patents.
Our rights to develop and commercialize our product candidates and proprietary product platform may be subject, in part, to the terms and conditions of current and future licenses granted to us by others.
Some of our licensed rights could provide us with freedom to operate for aspects of our products and services. We may need to obtain additional licenses from others to advance our research, development and commercialization activities.
Disputes may arise between us and our licensors regarding intellectual property subject to a license agreement, including:
| ● | the scope of rights granted under the license agreement and other interpretation-related issues; |
| ● | whether, and the extent to which, our products, services, technology and processes infringe on the intellectual property of the licensor that is not subject to the license agreement; |
| ● | our right to sublicense patent and other rights to third parties under collaborative development relationships; |
| ● | our diligence obligations under the license agreement and what activities satisfy those diligence obligations; |
| ● | the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our collaborators; and |
| ● | the priority of invention of patented technology. |
If we do not prevail in such disputes, we may lose any or all of our rights under such license agreements.
In addition, the agreements under which we currently license intellectual property or technology from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology or could increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects. Moreover, if disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize any affected products or services, which could have a material adverse effect on our business, financial conditions, results of operations and prospects.
Absent the license agreements, we may infringe patents subject to those agreements, and, if the license agreements are terminated, we may be subject to litigation by the licensor. Litigation could result in substantial costs to us and distract our management. If we do not prevail, we may be required to pay damages, including treble damages, attorneys’ fees, costs and expenses and royalties. We may also be enjoined from selling our products or services, which could adversely affect our ability to offer products or services, our ability to continue operations, and our financial condition.
55
If we infringe the rights of third parties, we could be prevented from selling products, forced to pay damages and/or royalties, and forced to defend against litigation.
We do not believe that the products we are currently developing infringe upon the rights of any third parties or are infringed upon by third parties. However, there can be no assurance that our technology will not be found in the future to infringe upon the rights of others or be infringed upon by others. Moreover, patent applications are in some cases maintained in secrecy until patents are issued. The publication of discoveries in the scientific or patent literature frequently occurs much later than the date on which the underlying discoveries were made and patent applications were filed. Because patents can take many years to issue, there may be currently pending applications of which we are unaware that may later result in issued patents that our products or product candidates infringe. For example, pending patent applications may exist that provide support or can be amended to provide support for a claim that results in an issued patent that is infringed by one or more of our products. In such a case, others may assert infringement claims against us, and should we be found to infringe these patents or impermissibly use their intellectual property, we might be forced to pay damages, potentially including treble damages, if we are found to have willfully infringed on such third parties’ patent rights.
In addition to any damages we might have to pay, we may also be required to obtain licenses from the holders of this intellectual property, enter into royalty agreements, or redesign our products so as not to use this intellectual property. Each of these penalties may prove to be uneconomical or otherwise impossible. We may fail to obtain any such licenses or intellectual property rights on commercially reasonable terms. Even if we are able to obtain a license, it may be non-exclusive, thereby giving our competitors access to the same licensed technologies. In that event, we may be required to spend significant time and resources to develop or license replacement technologies. If we are unable to do so, we may be unable to develop or commercialize the affected products, which could materially harm our business. Conversely, we may not be able to pursue claims against third parties that infringe on our licensed or co-owned technology. Thus, our licensed and co-owned technology may not provide adequate protection against competitors.
The pharmaceutical industry is characterized by extensive litigation regarding patents and other intellectual property rights. Moreover, the cost to us of any litigation or other proceeding relating to our licensed and/or co-owned intellectual property rights, even if resolved in our favor, could be substantial. Any such litigation would divert our management efforts, and we may not have sufficient resources to bring any such action to a successful conclusion. Uncertainties resulting from the initiation and continuation of any litigation could limit our ability to continue operations.
Additionally, because our pipeline may involve additional development candidates that could require the use of proprietary rights held by third parties, the growth of our business could depend in part on our ability to acquire, in-license or use these proprietary rights. In addition, our development candidates may require specific formulations to work effectively and efficiently and these rights may be held by others. We may be unable to acquire or in-license any compositions, methods of use, processes or other third-party intellectual property rights from third parties that we identify. The licensing and acquisition of third-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to license or acquire third-party intellectual property rights that we may consider attractive. These established companies may have a competitive advantage over us due to their size, cash resources, and greater clinical development and commercialization capabilities.
For example, we sometimes collaborate with U.S. and foreign academic institutions to accelerate our preclinical research or development under written agreements with these institutions. Typically, these institutions provide us with an option to negotiate a license to any of the institution’s rights in technology resulting from the collaboration. Regardless of such right of first negotiation for intellectual property, we may be unable to negotiate a license within the specified time frame or under terms that are acceptable to us. If we are unable to do so, the institution may offer the intellectual property rights to other parties, potentially blocking our ability to pursue our program.
In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment. If we are unable to successfully obtain rights to require third-party intellectual property rights, our business, financial condition and prospects for growth could suffer.
56
We may not be successful in obtaining, through acquisitions, in-licenses or otherwise, necessary rights to our product candidates, proprietary product platform technologies or other technologies.
We currently have rights to certain intellectual property, through licenses from third parties, to develop our product candidates and proprietary product platform technologies. Some healthcare companies and academic institutions are competing with us in the field of phage-based therapies and may have patents and/or have filed and are likely filing patent applications potentially relevant to our business. In order to avoid infringing these third-party patents, we may find it necessary or prudent to obtain licenses to such patents from such third-party intellectual property holders. We may also require licenses from third parties for certain technologies that we may be evaluating for use with our current or future product candidates. However, we may be unable to secure such licenses or otherwise acquire or in-license any compositions, methods of use, processes or other intellectual property rights from third parties that we identify as necessary for our current or future product candidates and our proprietary product platform at a reasonable cost or on reasonable terms, if at all. The licensing or acquisition of third-party intellectual property rights is a competitive area, and several more established companies may pursue strategies to license or acquire third-party intellectual property rights that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, capital resources and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment or at all.
In the event that we try to obtain rights to required third-party intellectual property rights and are ultimately unsuccessful, we may be required to expend significant time and resources to redesign our technology, product candidates or the methods for manufacturing them or to develop or license replacement technology, all of which may not be feasible on a technical or commercial basis. If we are unable to do so, we may be unable to develop or commercialize the affected product candidates or continue to utilize our existing proprietary product platform technology, which could significantly harm our business, financial condition, results of operations and prospects.
We rely on our proprietary product platform to identify phage-based therapies. Our competitive position could be materially harmed if our competitors develop a similar platform and develop rival product candidates.
We rely on know-how, inventions and other proprietary information to strengthen our competitive position. We consider know-how to be our primary intellectual property with respect to our proprietary product platform. Our clinical trials allow us to collect clinical data, which we use as a feedback loop to make improvements to our proprietary product platform. In particular, we anticipate that, with respect to this proprietary product platform, this data may over time be disseminated within the industry through independent development, the publication of journal articles describing the method and the movement of skilled personnel.
We cannot rule out that our competitors may have or obtain the knowledge necessary to analyze and characterize similar data to our known data for the purpose of identifying and developing products that could compete with any of our product candidates. Our competitors may also have significantly greater financial, product development, technical and human resources access to date. Further, our competitors may have significantly greater experience in using translational science methods to identify and develop product candidates.
We may not be able to prohibit our competitors from using technology or methods that are the same as or similar to our proprietary product platform to develop their own product candidates. If our competitors develop associated therapies, our ability to develop and market a promising product or product candidate may diminish substantially, which could have a material adverse effect on our business, financial condition, prospects and results of operations.
57
We rely on trade secrets and other forms of non-patent intellectual property protection. If we are unable to protect our trade secrets, other companies may be able to compete more effectively against us.
We rely on trade secrets to protect certain aspects of our technology, including our proprietary processes for manufacturing and purifying bacteriophage. Trade secrets are difficult to protect, especially in the pharmaceutical industry, where much of the information about a product must be made public during the regulatory approval process. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, outside scientific collaborators and other advisors may unintentionally or willfully disclose our information to competitors. Enforcing a claim that a third party illegally obtained and is using our trade secret information is expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the United States may be less willing to or may not protect trade secrets. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
If we are sued for infringing intellectual property rights of third parties or if we are forced to engage in an interference proceeding, it will be costly and time-consuming, and an unfavorable outcome in that litigation or interference would have a material adverse effect on our business.
Our ability to commercialize our product candidates depends on our ability to develop, manufacture, market and sell our product candidates without infringing the proprietary rights of third parties. Numerous U.S. and foreign patents and patent applications, which are owned by third parties, exist in the general field of anti-infective products or in fields that otherwise may relate to our product candidates. If we are shown to infringe, we could be enjoined from the use or sale of the claimed invention if we are unable to prove that the patent is invalid. In addition, because patent applications can take many years to issue, there may be currently pending patent applications, unknown to us, that may later result in issued patents that our product candidates may infringe or that may trigger an interference proceeding regarding one of our owned or licensed patents or applications. There could also be existing patents of which we are not aware that our product candidates may inadvertently infringe or that may become involved in an interference proceeding.
The biotechnology and pharmaceutical industries are characterized by the existence of a large number of patents and frequent litigation based on allegations of patent infringement. For so long as our product candidates are in clinical trials, we believe our clinical activities fall within the scope of the exemptions provided by 35 U.S.C. Section 271(e) in the United States, which exempts from patent infringement liability activities reasonably related to the development and submission of information to the FDA. As our clinical investigational drug product candidates progress toward commercialization, the possibility of a patent infringement claim against us increases. While we attempt to ensure that our active clinical investigational drugs and the methods we employ to manufacture them, as well as the methods for their use we intend to promote, do not infringe other parties’ patents and other proprietary rights, we cannot be certain they do not, and competitors or other parties may assert that we infringe their proprietary rights in any event.
We may be exposed to future litigation based on claims that our product candidates, the methods we employ to manufacture them or the uses for which we intend to promote them infringe the intellectual property rights of others. Our ability to manufacture and commercialize our product candidates may depend on our ability to demonstrate that the manufacturing processes we employ and the use of our product candidates do not infringe third-party patents. If third-party patents were found to cover our product candidates or their use or manufacture, we could be required to pay damages or be enjoined and therefore unable to commercialize our product candidates, unless we obtained a license. A license may not be available to us on acceptable terms, if at all.
We may become subject to claims for remuneration or royalties for assigned service invention rights by our employees, which could result in litigation and adversely affect our business.
A significant portion of our intellectual property has been developed by our employees in the course of their employment for us. Under the Israeli Patent Law, 5727-1967, or the Patent Law, inventions conceived by an employee during the term and as part of the scope of his or her employment with a company are regarded as “service inventions,” which belong to the employer, absent a specific agreement between the employee and employer giving the employee service invention rights. The Patent Law also provides that, if there is no such agreement between an employer and an employee, the Israeli Compensation and Royalties Committee, a body constituted under the Patent Law, shall determine whether the employee is entitled to remuneration for his or her inventions. We generally enter into assignment of invention agreements with our employees pursuant to which such individuals assign to us all rights to any inventions created in the scope of their employment or engagement with us. Although our employees have agreed to assign to our service invention rights, we may face claims demanding remuneration in consideration for assigned inventions. As a consequence of such claims, we could be required to pay additional remuneration or royalties to our current or former employees or be forced to litigate such claims, which could negatively affect our business.
58
Risks Related to Our Reliance on Third Parties
We rely, and continue to rely, on third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such trials.
We continue to rely on third parties, such as contract research organizations, or CROs, and clinical investigators, to conduct and manage our clinical trials.
Our reliance on these third parties for research and development activities will reduce our control over these activities but does not relieve us of our responsibilities. For example, we remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the trial. Moreover, the FDA requires us to comply with GCPs for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, safety and welfare of trial participants are protected. Other countries’ regulatory agencies also have requirements for clinical trials with which we must comply. We are also required to register ongoing clinical trials and post the results of completed clinical trials in a government-sponsored database, clinicaltrials.gov, within specified time frames. Failure to do so can result in fines, adverse publicity, and civil and criminal sanctions.
Furthermore, these third parties may also have relationships with other entities, some of which may be our competitors. If these third parties do not successfully carry out their contractual duties, do not meet expected deadlines, experience work stoppages, terminate their agreements with us or need to be replaced, or do not conduct our clinical trials in accordance with regulatory requirements or our stated protocols, we may need to enter into new arrangements with alternative third parties, which could be difficult, costly or impossible, and our clinical trials may be extended, delayed, terminated or need to be repeated. If any of the foregoing occurs, we may not be able to obtain, or may be delayed in obtaining, marketing approvals for our product candidates and may not be able to, or may be delayed in our efforts to, successfully commercialize our product candidates.
We also rely on other third parties to store and distribute drug supplies for our clinical trials. Any performance failure on the part of our distributors could delay clinical development or marketing approval of our product candidates or commercialization of our products, producing additional losses and depriving us of potential product revenue.
Third-party relationships are important to our business. If we are unable to maintain our collaborations or enter into new relationships, or if these relationships are not successful, our business could be adversely affected.
We have limited capabilities for product development and do not yet have any capability for sales, marketing or distribution. Accordingly, we enter into relationships with other companies and academic institutions to provide us with important technology, and we may receive additional technology and funding under these and other collaborations in the future. The relationships we enter into may pose a number of risks, including the following:
| ● | third parties have, and future third-party collaborators may have, significant discretion in determining the efforts and resources that they will apply; |
| ● | current and future third parties may not perform their obligations as expected; |
| ● | current and future third parties may not pursue development and commercialization of any product candidates that achieve regulatory approval or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the third parties’ strategic focus or available funding, or external factors, such as a strategic transaction that may divert resources or create competing priorities; |
59
| ● | third parties may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
| ● | current and future third parties could independently develop, or develop with third parties, products that compete directly or indirectly with our products and product candidates if the third parties believe that the competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| ● | product candidates discovered in collaboration with us may be viewed by our current or future third parties as competitive with their own product candidates or products, which may cause such third parties to cease to devote resources to the commercialization of our product candidates; |
| ● | current and future third parties may fail to comply with applicable regulatory requirements regarding the development, manufacture, distribution or marketing of a product candidate or product; |
| ● | current and future third parties with marketing and distribution rights to one or more of our product candidates that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of such product or products; |
| ● | disagreements with current or future third parties, including disagreements over proprietary rights, contract interpretation or the preferred course of development, might cause delays or terminations of the research, development or commercialization of product candidates, might lead to additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration, any of which would be time-consuming and expensive; |
| ● | current and future third parties may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| ● | current and future third parties may infringe the intellectual property rights of others, which may expose us to litigation and potential liability; |
| ● | current and future third parties may infringe regulatory frameworks (such as but not limited to cybersecurity and/or privacy frameworks), which may expose us to litigation and potential liability or require or lead us to terminate relationships with them; |
| ● | if a current or future third party is involved in a business combination, the collaborator might deemphasize or terminate the development or commercialization of any product candidate licensed to it by us; and |
| ● | current and future relationships may be terminated by the collaborator, and, if terminated, we could be required to raise additional capital to pursue further development or commercialization of the applicable product candidates. |
If our relationships do not result in the successful discovery, development and commercialization of products or if one of our third-party collaborators terminates its agreement with us, we may not receive any future research funding or milestone or royalty payments under the collaboration. If we do not receive the funding we expect under these agreements, our development of our technology and product candidates could be delayed, and we may need additional resources to develop product candidates and our technology. Additionally, if any of our current or future third-party collaborators terminates its agreement with us, we may find it more difficult to attract new collaborators, and our reputation in the business and financial communities could be adversely affected.
60
Relationships are complex and time-consuming to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators. We face significant competition in seeking appropriate collaborators. Our ability to reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of a collaborator’s resources and expertise, the terms and conditions of a proposed collaboration and a proposed collaborator’s evaluation of a number of factors.
We may not be successful in maintaining or establishing collaborations, which could adversely affect our ability to develop and, if required regulatory approvals are obtained, commercialize our product candidates.
In the future, in order to advance our clinical development, or in connection with any potential out-licensing of product candidates or technologies, we may seek to enter into collaboration agreements. In addition, we may consider entering into collaboration arrangements with medical technology, pharmaceutical or biotechnology companies and/or seek to establish strategic relationships with marketing partners for the development, sale, marketing and/or distribution of our product candidates within or outside of the United States. If we are unable to reach agreements with potential collaborators, then we may fail to meet our business objectives for the affected product candidates or programs. Collaboration arrangements are complex and time-consuming to negotiate, document and implement, and we may not be successful in our efforts, if any, to establish and implement collaborations or other alternative arrangements. The terms of any collaboration or other arrangements that we establish may not be favorable to us, and the success of any such collaboration will depend heavily on the efforts and activities of our collaborators. Moreover, our collaboration agreement could be terminated or not renewed by a third party at a time that is costly or damaging to us. Any failure to engage successful collaborators could cause delays in our product development and/or commercialization efforts, which could harm our financial condition and operational results.
Risks Related to Our Operations in Israel
Our headquarters, research and development and other significant operations are located in Israel, and, therefore, our results may be adversely affected by political, economic and military instability in Israel, including the recent war with Hamas and other terrorist organizations from the Gaza Strip.
Our executive offices and research and development facilities are located in Israel. In addition, the majority of our key employees and all of our officers are residents of Israel. Accordingly, political, geopolitical, economic and military conditions in Israel may directly affect our business. Since the establishment of the State of Israel in 1948, a number of armed conflicts have taken place between Israel and its neighboring Arab countries, the Hamas (an Islamist terrorist militia and political group that controls the Gaza strip), the Hezbollah (an Islamist terrorist militia and political group based in Lebanon) and other terrorist organizations active in the region. These conflicts have involved missile strikes, hostile infiltrations and terrorism against civilian targets in various parts of Israel, which have negatively affected business conditions in Israel. Any hostilities involving Israel or the interruption or curtailment of trade between Israel and its trading partners could negatively affect business conditions in Israel in general and our business in particular, and adversely affect our product development, operations and results of operations.
In October 2023, Hamas terrorists infiltrated Israel’s southern border from the Gaza Strip and conducted a series of attacks on civilian and military targets. Hamas also launched extensive rocket attacks on Israeli population and industrial centers located along Israel’s border with the Gaza Strip and in other areas within the State of Israel. These attacks resulted in extensive deaths, injuries and kidnapping of civilians and soldiers. Following the attack, Israel’s security cabinet declared war against Hamas and a military campaign against these terrorist organizations commenced in parallel to their continued rocket and terror attacks. In addition, since the commencement of these events, there have been continued hostilities along Israel’s northern border with Lebanon (with the Hezbollah terror organization) and southern border (with the Houthi movement in Yemen, as described below). It is possible that hostilities with Hezbollah in Lebanon will escalate, and that other terrorist organizations, including Palestinian military organizations in the West Bank as well as other hostile countries, such as Iran, will join the hostilities. Such clashes may escalate in the future into a greater regional conflict.
In connection with the Israeli security cabinet’s declaration of war against Hamas and possible hostilities with other organizations, several hundred thousand Israeli military reservists were drafted to perform immediate military service, including 9 employees, none of whom are management or key employees, who were called up for reserve service, of which 4 have since returned to work full time and their pre-war military reserve duty. So long as the war continues, our personnel may be called up for reserve service, whether for an extended periods or periodically for short-term periods. Military service call ups that result in absences of personnel for an extended period of time may materially and adversely affect our business, prospects, financial condition and results of operations.
61
Since the war broke out on October 7, 2023, our operations have not been adversely affected by this situation, and we have not experienced disruptions to our business operations. As such, our product and business development activities remain on track. However, the intensity and duration of Israel’s current war against Hamas is difficult to predict at this stage, as are such war’s economic implications on our business and operations and on Israel’s economy in general. If the war extends for a long period of time or expands to other fronts, such as Lebanon, Syria and the West Bank, our operations may be adversely affected.
Any armed conflicts, terrorist activities or political instability in the region could adversely affect business conditions, could harm our results of operations and the market price of our Common Stock, and could make it more difficult for us to raise capital.
Our commercial insurance does not cover losses that may occur as a result of an event associated with the security situation in the Middle East. Although the Israeli government has in the past covered the reinstatement value of certain damages that were caused by terrorist attacks or acts of war, we cannot assure you that this government coverage will be maintained or, if maintained, will be sufficient to compensate us fully for damages incurred. Any losses or damages incurred by us could have a material adverse effect on our business.
Finally, political conditions within Israel may affect our operations. Israel has held five general elections between 2019 and 2022, and prior to October 2023, the Israeli government pursued extensive changes to Israel’s judicial system, which sparked extensive political debate and unrest. To date, these initiatives have been substantially put on hold. Actual or perceived political instability in Israel or any negative changes in the political environment, may individually or in the aggregate adversely affect the Israeli economy and, in turn, our business, financial condition, results of operations and growth prospects.
Our operations may be disrupted as a result of the obligation of management or key personnel to perform military service.
As of the date hereof, we currently have 99 full-time employees, of which 64 are located in Israel, including 5 members of our senior management. Certain of our employees and consultants in Israel, including members of our senior management, may be obligated to perform military reserve duty generally until they reach the age of 40 (or older, for officers or other citizens who hold certain positions in the Israeli armed forces reserves) and, in the event of a military conflict, may be called to active duty. In response to increases in terrorist activity, there have been periods of significant call-ups of military reservists. It is possible that there will be similar large-scale military reserve duty call-ups in the future. Our operations could be disrupted by the absence of a significant number of our officers, directors, employees and consultants. Such disruption could materially adversely affect our business and operations.
The Israeli government grants we have received for research and development expenditures restrict our ability to manufacture products and transfer technology outside of Israel and require us to satisfy specified conditions. If we fail to satisfy these conditions, we may be required to refund grants previously received, together with interest and penalties.
Our research and development efforts have been financed, in part, through the grants that we have received from the Israeli Innovation Authority, or the IIA. We, therefore, must comply with the requirements of Israel’s Law for the Encouragement of Research and Development in Industries, or the Research Law. For the years ended December 31, 2023 and 2022, we recorded grants totaling $1.0 million and $1.1 million, from the IIA, respectively. The grants represented 7.3% and 6.1% of our gross research and development expenditures for the years ended December 31, 2023 and 2022, respectively.
Under the Research Law, we are required to manufacture the major portion of each of our products developed using these grants in the State of Israel or otherwise ask for special approvals. We may not receive the required approvals for any proposed transfer of manufacturing activities. Even if we receive approval to manufacture products developed with government grants outside of Israel, the royalty rate may be increased, and we may be required to pay up to 300% of the grant amounts, plus interest, depending on the manufacturing volume that is performed outside of Israel. This restriction may impair our ability to outsource manufacturing or engage in our own manufacturing operations for those products or technology.
62
Additionally, under the Research Law, we are prohibited from transferring, including by way of license, the IIA-financed technology and related intellectual property rights and know-how outside of the State of Israel, except under limited circumstances and only with the approval of the IIA Research Committee. We may not receive the required approvals for any proposed transfer, and, even if received, we may be required to pay the IIA a portion, to be set by the IIA, in its discretion and taking into account the circumstances, upon its approval of such transaction, of the consideration or milestone and royalty payments that we receive upon any sale or out-licensing of such technology to a non-Israeli entity, up to 600% of the grant amounts plus interest.
These restrictions may impair our ability to sell our technology assets or to perform or outsource manufacturing outside of Israel or otherwise transfer our know-how outside of Israel and may require us to obtain the approval of the IIA for certain actions and transactions and pay additional royalties and other amounts to the IIA. In addition, any change of control and any change of ownership of our Common Stock that would make a non-Israeli citizen or resident an “interested party,” as defined in the Research Law, requires prior written notice to the IIA, and our failure to comply with this requirement could, under certain circumstances, result in criminal liability.
These restrictions will continue to apply even after we have repaid the full amount of royalties on the grants.
We have received, and may continue to receive, Israeli governmental grants to assist in the funding of our research and development activities. If we lose our funding from these research and development grants, we may encounter difficulties in the funding of future research and development projects and implementing technological improvements, which would harm our operating results.
Through December 31, 2023, we had received an aggregate of $8.0 million in the form of grants from the IIA. BiomX Ltd. was formed as an incubator company as part of the FutuRx incubator, and, until 2017, the majority of our funding was from IIA grants and funding by the incubator, which is supported by the IIA. We continued to apply for and receive IIA grants after we left the incubator. The requirements and restrictions for such grants are found in the Research Law. Under the Research Law, royalties of 3% to 3.5% on the revenue derived from sales of products or services developed in whole or in part using these IIA grants are payable to the Israeli government. We developed both of our platform technologies, at least in part, with funds from these grants, and, accordingly, we would be obligated to pay these royalties on sales of any of our product candidates that achieve regulatory approval. As long as the manufacturing of our product candidates takes place in Israel and no technology funded with IIA grants is sold or out licensed to a non-Israeli entity, the maximum aggregate royalties paid generally would not exceed 100% of the grants made to us, plus annual interest equal to the 12-month Secured Overnight Financing Rate, or SOFR, applicable to dollar deposits, as published on the first trading day of each calendar year. As of December 31, 2023, the balance of the principal and interest in respect of our commitments for future payments to the IIA totaled approximately $7.9 million. As part of funding our current and planned product development activities, we may submit follow-up grant applications for additional grants.
These grants have funded some of our personnel, development activities with subcontractors, and other research and development costs and expenses. However, if these awards are not funded in their entirety or if additional grants are not awarded in the future, due to, for example, IIA budget constraints or governmental policy decisions, our ability to fund future research and development and implement technological improvements would be impaired, which would negatively impact our ability to develop our product candidates.
63
Exchange rate fluctuations between the U.S. Dollar, the New Israeli Shekel, the Euro and other foreign currencies, may negatively affect our future expenses.
Our proceeds from sales of our securities are generally received in U.S. Dollars. Our headquarters are located in Israel, where the majority of our general and administrative expenses and research and development costs are incurred in the New Israeli Shekel, or NIS. Future expenses may be incurred in foreign currencies such as the Euro or British Pound. As a result, our financial results may be affected by fluctuations in the exchange rates of currencies in the countries. For example, during 2020, we witnessed a strengthening of the average exchange rate of the NIS against the U.S. Dollar, which increased the U.S. Dollar value of Israeli expenses. If the NIS strengthens against the U.S. Dollar, as it did in 2020 and 2021, the U.S. Dollar value of our Israeli expenses, mainly personnel and facility-related, will increase. We use foreign exchange contracts (mainly option and forward contracts) to hedge balance sheet items from currency exposure. However, these foreign exchange contracts are not designated as hedging instruments for accounting purposes and they may not be effective. Although exposure to currency fluctuations to date has not had a material adverse effect on our business, there can be no assurance that fluctuations in the future will not have a material adverse effect on our operating results and financial condition.
Under applicable employment laws, we may not be able to enforce covenants not to compete.
We generally enter into noncompetition agreements with our employees. These agreements prohibit our employees, if they cease working for us, from competing directly with us or working for our competitors or clients for a limited period. We may be unable to enforce these agreements under the laws of the jurisdictions in which our employees work, and it may be difficult for us to restrict our competitors from benefitting from the expertise our former employees or consultants developed while working for us. For example, Israeli labor courts have required employers seeking to enforce noncompete undertakings of a former employee to demonstrate that the competitive activities of the former employee will harm one of a limited number of material interests of the employer that have been recognized by the courts, such as the protection of a company’s trade secrets or other intellectual property.
The tax benefits that are available to us if and when we generate taxable income require us to meet various conditions and may be prevented or reduced in the future, which could increase our costs and taxes.
If and when we generate taxable income, we would be eligible for certain tax benefits provided to “Technologic Preferred Enterprise” and/or “Preferred Enterprise” as defined under the Encouragement of Capital Investment Law -1959, the Law, and its regulations, as amended and, accordingly, could be subject to a reduced corporate tax rate on its income that will meet the provisions of the Law (ranging between 7.5%-16%). To the extent that we are not eligible to obtain such statuses, our Israeli taxable income would be subject to regular Israeli corporate tax rates. The standard corporate tax rate for Israeli companies is 23%. The benefits available to us in accordance to the Law and its regulations are subject to the fulfillment of conditions stipulated in the Law and the regulations. Further, in the future, these tax benefits may be reduced or discontinued.
It may be difficult to enforce a U.S. judgment against us or our officers and directors in Israel or the United States or to assert U.S. securities laws claims in Israel or serve process on our officers and directors.
Not all of our directors or officers are residents of the United States, and most of their and our assets are located outside the United States. Service of process upon us or our non-U.S. resident directors and officers may be difficult to obtain within the United States. Israeli courts may refuse to hear a claim based on a violation of U.S. securities laws against us or our non-U.S. officers and directors, because Israel may not be the most appropriate forum to bring such a claim. In addition, even if an Israeli court agrees to hear a claim, it may determine that Israeli law, and not U.S. law, is applicable to the claim. If U.S. law is found to be applicable, the content of applicable U.S. law must be proved as a fact, which can be a time-consuming and costly process. Certain matters of procedure will also be governed by Israeli law. There is little binding case law in Israel addressing the matters described above. Additionally, Israeli courts might not enforce judgments obtained in the United States against us or our non-U.S. directors and executive officers, which may make it difficult to collect on judgments rendered against us or our non-U.S. officers and directors.
64
Moreover, an Israeli court will not enforce a non-Israeli judgment if it was given in a state whose laws do not provide for the enforcement of judgments of Israeli courts (subject to exceptional cases), if its enforcement is likely to prejudice the sovereignty or security of the State of Israel, if it was obtained by fraud or in the absence of due process, if it is at variance with another valid judgment that was given in the same matter between the same parties, or if a suit in the same matter between the same parties was pending before a court or tribunal in Israel at the time the foreign action was brought.
Risks Related to Manufacturing and Supply
We rely on third parties to manufacture our clinical supply of product candidates and we intend to rely on third parties to produce and process our products, if approved.
We currently rely on outside vendors to supply raw materials and other important components, such as lab equipment. We have not yet caused any product candidates to be manufactured or processed on a commercial scale and may not be able to do so for any of our product candidates. We will make changes as it works to optimize the manufacturing process for our product candidates, and we cannot be sure that even minor changes in the process will result in therapies that are safe and effective.
The facilities used to manufacture our product candidates must be approved by the FDA or equivalent foreign regulatory agencies pursuant to inspections that will be conducted after we submit a marketing application to the FDA or equivalent foreign regulatory agency. Additionally, any facilities used for the manufacture of product candidates commercialized for non-therapeutic uses will be subject to inspection by the FDA and foreign regulatory agencies. We do not currently control all aspects of the manufacturing process of, and are currently largely dependent on, our contract manufacturing partners for compliance with regulatory requirements, known as cGMP requirements, for manufacture of our product candidates. If and when our manufacturing facility becomes operational, we will be responsible for compliance with cGMP requirements. If we or our contract manufacturers cannot successfully manufacture in conformance with our specifications and the strict regulatory requirements of the FDA or other regulatory authorities, we and they will not be able to secure and/or maintain regulatory approval for their manufacturing facilities with respect to the manufacture of our product candidates. In addition, we have no control over the ability of our contract manufacturers to maintain adequate quality control, quality assurance and qualified personnel. If the FDA or an equivalent foreign regulatory agency does not approve these facilities for the manufacture of our product candidates or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved.
We have limited experience manufacturing our product candidates for purposes of clinical trials for therapeutic indications or for non-therapeutic clinical studies or trials. We opened our own manufacturing facility at our headquarters in Ness Ziona, Israel in 2019. We cannot assure you that we can manufacture our product candidates in compliance with regulations at a cost or in quantities necessary to make them commercially viable.
Our product candidates rely on the availability of specialty raw materials, which may not be available to us on acceptable terms or at all.
Our product candidates require certain specialty raw materials, some of which we obtain from small companies with limited resources and experience to support a commercial product. These third-party suppliers may be ill-equipped to support our needs, especially in non-routine circumstances like an FDA inspection or medical crisis, such as widespread contamination. We do not currently have contracts in place with all of the suppliers that we may need at any point in time and, if needed, may not be able to contract with them on acceptable terms or at all. Accordingly, we may experience delays in receiving key raw materials to support clinical or commercial manufacturing.
65
Risks Related to Our Common Stock
A significant number of shares of our Common Stock are subject to issuance upon exercise of outstanding warrants and options or conversion of our Convertible Preferred Stock, which upon such exercise or conversion, as applicable, may result in dilution to our security holders.
As of December 31, 2023, we had an aggregate of 25,363,688 warrants outstanding to purchase an aggregate of up to 20,926,189 shares of Common Stock with a weighted average exercise price of $2.60, certain of which, or the Unit Warrants are included in our outstanding units, each consisting of one share of Common Stock and one warrant exercisable for one-half of one share of Common Stock, or the Units, certain of which were issued in private placements, or Private Warrants, certain of which, or the Pre-funded Warrants, were issued in the February 2023 PIPE, and certain of which, or the Public Warrants, were previously traded on NYSE American under the symbol “PHGE.WS,” and are currently quoted on OTC Pink under the symbol “PHGEW”. Unit Warrants, Private Warrants, Pre-funded Warrants and Public Warrants, collectively, or the Outstanding Warrants, in each case subject to adjustment. To the extent such Outstanding Warrants are exercised, additional shares of our Common Stock will be issued, which will result in dilution to the then existing holders of Common Stock and increase the number of shares eligible for resale in the public market. Sales of substantial numbers of such shares in the public market could adversely affect the market price of our Common Stock.
In addition, as of December 31, 2023, we had outstanding vested and unvested options to purchase 5,280,711 shares of our Common Stock. To the extent any of these options are exercised, additional shares of Common Stock will be issued that will generally be eligible for resale in the public market (subject to limitations under Rule 144 under the Securities Act with respect to shares held by our affiliates), which will result in dilution to our security holders.
Furthermore, (i) in connection with the Acquisition, in addition to issuance of Common Stock, we issued (a) an aggregate of 40,470 shares of Convertible Preferred Stock, which, in the event our stockholders approve the conversion of the Convertible Preferred Stock at the Stockholder Meeting, will be convertible into an aggregate of up to 40,470,000 shares of our Common Stock, and (b) warrants, or Merger Warrants, which, in the event our stockholders approve the exercise of the Merger Warrants at the Stockholder Meeting, will be exercisable for an aggregate of 2,416,497 shares of Common Stock, and (ii) in connection with the March 2024 PIPE, we issued (x) an aggregate of 216,417 shares of Convertible Preferred Stock, which, in the event our stockholders approve the conversion of the Convertible Preferred Stock at the Stockholder Meeting, will be convertible into an aggregate of up to 216,417,000 shares of Common Stock, and (y) Private Placement Warrants, which, in the event our stockholders approve the exercise of the Private Placement Warrants at the Stockholder Meeting, will be exercisable for an aggregate of 108,208,500 shares of our Common Stock. Additionally, as partial compensation to the placement agents for the March 2024 PIPE, we issued warrants, or Placement Agent Warrants, and collectively with the Merger Warrants and the Private Placement Warrants, the Warrants, which, in the event our stockholders approve the conversion of the Placement Agent Warrants at the Stockholder Meeting, will be exercisable for up to an aggregate of 9,523,809 shares of Common Stock. To the extent any of the Convertible Preferred Stock is converted or any of the Warrants are exercised, additional shares of Common Stock will be issued that, subject to applicable securities laws, will generally be eligible for resale in the public market (subject to limitations under Rule 144 under the Securities Act with respect to shares held by our affiliates). Sales of substantial numbers of such shares in the public market could adversely affect the market price of our Common Stock.
We plan to grant additional options, subject to stockholder approval at the Stockholder Meeting and may issue additional warrants and shares of preferred stock in the future. Furthermore, the issuance of additional shares of our Common Stock upon exercise of such securities, as applicable, will result in dilution to the then existing holders of Common Stock and could also have an adverse effect on the market price of our Common Stock.
We have never paid dividends on our Common Stock, and we do not anticipate paying any cash dividends on our Common Stock in the foreseeable future.
We have never declared or paid cash dividends on our Common Stock. We do not anticipate paying any cash dividends on our Common Stock in the foreseeable future. We currently intend to retain all available funds and any future earnings to fund the development and growth of our business. As a result, capital appreciation, if any, of our Common Stock will be our stockholders’ sole source of gain for the foreseeable future.
Our Public Warrants have been delisted, and we may be unable to maintain the listing of our securities in the future.
Our Common Stock and Units trade on NYSE American. Our Public Warrants previously traded on NYSE American but were delisted in June 2023 and since then have been quoted on OTC Pink. As a result of the delisting of our Public Warrants, the holders thereof have experienced a limited availability of market quotations and reduced liquidity with respect to their Public Warrants. If our Common Stock or Units are subsequently delisted, we could face significant material adverse consequences, including:
| ● | a limited availability of market quotations for our securities; |
| ● | reduced liquidity with respect to our securities; |
| ● | a determination that our shares are a “penny stock,” which will require brokers trading in our securities to adhere to more stringent rules, possibly resulting in a reduced level of trading activity in the secondary trading market for our securities; |
| ● | a limited amount of news and analyst coverage for the Company; and |
| ● | a decreased ability to issue additional securities or obtain additional financing in the future. |
66
The market price of our Common Stock and other securities may be volatile and fluctuate substantially, which could result in substantial losses for purchasers of our Common Stock.
The stock markets in general and the markets for biotechnology stocks have experienced extreme volatility. The market for the common stock of smaller companies such as ours is characterized by significant price volatility when compared to the shares of larger, more established companies that trade on a national securities exchange and have large public floats, and our share price is more volatile than the shares of such larger, more established companies for the indefinite future.
In addition to the factors discussed in this “Risk Factors” section, price declines in our Common Stock (and other securities) could also result from general market and economic conditions and a variety of other factors, including:
| ● | adverse results or delays in our clinical trials; | |
| ● | adverse actions taken by regulatory agencies with respect to our product candidates, clinical trials or the manufacturing processes of our product candidates; | |
| ● | announcements of technological innovations, patents or new products by our competitors; | |
| ● | regulatory developments in the United States and foreign countries; | |
| ● | any lawsuit involving us or our product candidates; | |
| ● | announcements concerning our competitors, or the biotechnology or pharmaceutical industries in general; | |
| ● | developments concerning any strategic alliances or acquisitions we may enter into; | |
| ● | actual or anticipated variations in our operating results; | |
| ● | changes in recommendations by securities analysts or lack of analyst coverage; | |
| ● | deviations in our operating results from the estimates of analysts; | |
| ● | our inability, or the perception by investors that we will be unable, to continue to meet all applicable requirements for continued listing of our Common Stock on NYSE American, and the possible delisting of our Common Stock; | |
| ● | sales of our Common Stock by our executive officers, directors and principal stockholders or sales of substantial amounts of Common Stock; and | |
| ● | loss of any of our key scientific or management personnel. |
Additionally, market prices for securities of biotechnology companies historically have been very volatile. The market for these securities has from time to time experienced significant price and volume fluctuations for reasons unrelated to the operating performance of any one company. Furthermore, our business may be adversely impacted by risks, or the public perception of the risks, related to a pandemic or other health crisis, such as the COVID-19 or as a result of the Israel-Hamas War or the Russian invasion of Ukraine and the resulting world sanctions on Russia, Belarus, and related parties. A significant outbreak of contagious diseases could result in a widespread health crisis that could adversely affect the economies and financial markets of many countries, resulting in an economic downturn.
In the past, following periods of volatility in the market price of a particular company’s securities, litigation has often been brought against that company. Any such lawsuit could consume resources and management time and attention, which could adversely affect our business.
67
As a “smaller reporting company” we are permitted to provide less disclosure than larger public companies, which may make our Common Stock less attractive to investors.
We are currently a “smaller reporting company,” as defined by Rule 12b-2 of the Exchange Act. As a smaller reporting company, we are eligible to take advantage of certain exemptions from various reporting requirements applicable to other public companies. Consequently, it may be more challenging for investors to analyze our results of operations and financial prospects which may result in less investor confidence. Investors may find our Common Stock less attractive as a result of our smaller reporting company status. If some investors find our Common Stock less attractive, there may be a less active trading market for our Common Stock and our stock price may be more volatile.
General Risk Factors
Our success depends, in part, on our ability to retain key executives and to attract, retain and motivate qualified personnel.
We are highly dependent on Jonathan Solomon, our chief executive officer, as well as the other principal members of our management, scientific and clinical team. Although we have entered into employment agreements with our executive officers, each of them may terminate their employment with us at any time. We do not maintain “key person” insurance for any of our executives or other employees. The loss of the services of any of our executive officers, other key employees, and other scientific and medical advisors, and our inability to find suitable replacements could result in delays in product development and harm our business. Additionally, our recent acquisition of APT and its integration into the Company’s business may increase the likelihood that employees depart in the foreseeable future.
Our continued ability to attract, retain and motivate highly qualified management, clinical and scientific personnel and our ability to develop and maintain important relationships with leading academic institutions, clinicians and scientists is critical to our success. Competition for qualified personnel in the biotechnology field is intense, particularly in Israel where our headquarters are located. We face competition for personnel from other biotechnology and pharmaceutical companies, universities, public and private research institutions and other organizations. We also face competition from other more well-funded and well-established businesses, and we may also be viewed as a riskier choice from a job stability perspective due to our relatively newer status than longer existing biotech and pharmaceutical companies. We may not be able to attract and retain qualified personnel on acceptable terms given the competition for such personnel. If we are unsuccessful in our retention, motivation and recruitment efforts, we may be unable to execute our business strategy.
Expectations relating to environmental, social and governance (ESG) programs may impose additional costs and expose us to new risks.
There is an increasing focus from certain investors and other key stakeholders concerning corporate responsibility, specifically related to environmental, social and governance, or ESG, factors. As a result, there is an increased emphasis on corporate responsibility ratings and a number of third parties provide reports on companies in order to measure and assess corporate responsibility performance. In addition, the ESG factors by which companies’ corporate responsibility practices are assessed may change, which could result in greater expectations of us and cause us to undertake costly initiatives to satisfy such new criteria. Alternatively, if we are unable to satisfy such new criteria, investors may conclude that our policies with respect to corporate responsibility are inadequate. We risk damage to our brand and reputation if our corporate responsibility procedures or standards do not meet the standards set by various constituencies. We may be required to make investments in matters related to ESG, which could be significant and adversely impact our results of operations. Furthermore, if our competitors’ corporate responsibility performance is perceived to be greater than ours, potential or current investors may elect to invest with our competitors instead. In addition, if we communicate certain initiatives and goals regarding ESG matters, we could fail, or be perceived to fail, in our achievement of such initiatives or goals, or we could be criticized for the scope of such initiatives or goals. If we fail to satisfy the expectations of investors and other key stakeholders or our initiatives are not executed as planned, our reputation and financial results could be materially and adversely affected.
68
If we engage in future acquisitions or strategic partnerships, this may increase our capital requirements, dilute our stockholders, cause us to incur debt or assume contingent liabilities, and subject us to other risks.
On March 15, 2024, we acquired APT. We may evaluate various additional acquisition opportunities and strategic partnerships, including licensing or acquiring complementary products, intellectual property rights, technologies or businesses. Any potential acquisition or strategic partnership may entail numerous risks, including:
| ● | increased operating expenses and cash requirements; |
| ● | the assumption of additional indebtedness or contingent liabilities; |
| ● | the issuance of our equity securities; |
| ● | assimilation of operations, intellectual property and products of an acquired company, including difficulties associated with integrating new personnel; |
| ● | the diversion of our management’s attention from our existing product programs and initiatives in pursuing such a strategic merger or acquisition; |
| ● | retention of key employees, the loss of key personnel and uncertainties in our ability to maintain key business relationships; |
| ● | risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing products or product candidates and marketing approvals; and |
| ● | our inability to generate revenue from acquired technology and/or products sufficient to meet our objectives in undertaking the acquisition or even to offset the associated acquisition and maintenance costs. |
Our business and operations would suffer in the event of computer system failures, cyber-attacks or deficiencies in our cyber-security.
Despite the implementation of security measures, our internal computer systems, and those of third parties on which we rely, are vulnerable to damage from computer viruses, malware, natural disasters, terrorism, war, telecommunication and electrical failures, cyber-attacks or cyber-intrusions over the Internet, attachments to emails, persons inside our organization, or persons with access to systems inside our organization. The risk of a security breach or disruption, particularly through cyber-attacks or cyber intrusion, including by computer hackers, foreign governments, and cyber terrorists, has generally increased as the number, intensity and sophistication of attempted attacks and intrusions from around the world have increased. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our product development programs. For example, the loss of clinical trial data from completed or ongoing or planned clinical trials could result in delays in our clinical trial efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach was to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur material legal claims and liability, and damage to our reputation, and the further development of our product candidates could be delayed. We also maintain compliance programs to address the potential applicability of restrictions against trading while in possession of material, nonpublic information generally and in connection with a cyber-security breach. However, a breakdown in existing controls and procedures around our cyber-security environment may prevent us from detecting, reporting or responding to cyber incidents in a timely manner and could have a material adverse effect on our financial position and value of our stock.
We incur significant costs operating as a public company.
As a public company, we incur significant costs in connection with our directors and officers insurance, paying for service providers such as legal and accounting as well as other expenses. We are subject to the reporting requirements of the Exchange Act, which require, among other things, that we file with the SEC annual, quarterly and current reports with respect to our business and financial condition. In addition, the Sarbanes-Oxley Act, as well as rules subsequently adopted by the SEC and NYSE American to implement provisions of the Sarbanes-Oxley Act, the Dodd-Frank Wall Street Reform and Consumer Protection Act, or the Dodd-Frank Act, and the Public Company Accounting Oversight Board impose significant requirements on public companies, including requiring the establishment and maintenance of effective disclosure and financial controls and changes in corporate governance practices. These expenses will likely increase in the future, particularly if we cease to be a “smaller reporting company”, as a result of additional corporate governance and disclosure requirements under the Sarbanes-Oxley Act, the Dodd-Frank Act, and SEC rules and regulations.
The rules and regulations applicable to public companies result in us continuing to incur substantial legal and financial compliance costs. These costs increase our net loss or decrease any net income and may require us to reduce costs in other areas of our business.
69
ITEM 1B. UNRESOLVED STAFF COMMENTS
Not applicable.
ITEM 1C. CYBERSECURITY
We recognize the critical importance of developing, implementing, and maintaining cybersecurity measures to safeguard our information systems and protect the confidentiality, integrity, and availability of our data. We address cybersecurity risks by implementing security measures on our internal computer systems and ensuring that third parties and business partners implement similar measures. These security measures include firewalls, intrusion prevention and detection systems, antimalware functionality and access controls, which are evaluated by our external IT consultant and improved through vulnerability assessments and cybersecurity threat intelligence.
Our senior director of operation is responsible for day-to-day assessment and management of risks from cybersecurity threats, including the prevention, mitigation, detection, and remediation of cybersecurity incidents.
The Audit Committee is responsible for reviewing our policies with respect to cybersecurity risks and relevant contingent liabilities and risks that may be material to the Company, including risks from third parties and business partners. The Audit Committee receives quarterly updates from management with respect to risks from cybersecurity threats. Such updates cover the Company’s information technology security program, including its current status, capabilities, changes during the last quarter, objectives and plans, as well as the evolving cybersecurity threat landscape.
To date, risks from cybersecurity threats have not materially affected us and we do not currently believe any risks from cybersecurity threats are reasonably likely to affect the Company, including our business strategy, results of operations or financial condition. For further information, see “Risk Factors — Our business and operations would suffer in the event of computer system failures, cyber-attacks or deficiencies in our cyber-security.” in Item 1A of this Annual Report. We maintain a cyber liability insurance policy. However, our cyber liability insurance policy may not cover all claims made against us, and defending a suit, regardless of its merit, could be costly and divert management’s attention from our business and operations.
ITEM 2. PROPERTIES
Our corporate headquarters are located in Ness Ziona, Israel. During the second quarter of 2021, we moved into a new 28,610 square feet facility of office and laboratory space, including a new 6,500 square feet manufacturing facility. The lease agreement expires in November 2025, with an option to extend the term by five years. This facility has been designed with the capacity to produce clinical quantities of our product candidates required for clinical development. In August 2022, BiomX Israel entered into a sublease agreement for a portion of its office space in Ness Ziona, Israel. The agreement is for a period of two years beginning on August 15, 2022.
In addition to our premises in Israel, we are leasing a 25,894 square feet facility of office and laboratory space in Gaithersburg, Maryland, including 6,100 square feet manufacturing facility. The lease agreement expires in July 2034, with an option to terminate in February 2029, subject to 12 months’ notice and early termination fee.
We believe our facilities are sufficient to meet our current needs.
ITEM 3. LEGAL PROCEEDINGS
We may be subject to legal proceedings, investigations and claims incidental to the conduct of our business from time to time. We are not currently a party to any material litigation or other material legal proceedings brought against us.
ITEM 4. MINE SAFETY DISCLOSURES
Not Applicable.
70
part II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Our shares of Common Stock, and Units, are traded on NYSE American under the symbols PHGE, and PHGE.U, respectively. Our Public Warrants are quoted on the OTC Pink under the symbol “PHGEW”.
Holders of Record
As of March 28, 2024, there were 55,220,707 issued and outstanding shares of our Common Stock held by 75 stockholders of record. The number of record holders was determined from the records of our transfer agent and does not include beneficial owners of shares of Common Stock whose shares are held in the names of various security brokers, dealers, and registered clearing agencies.
Dividends
We have not paid any cash dividends on our Common Stock to date and do not intend to pay cash dividends. The payment of cash dividends in the future will be dependent upon our revenues and earnings, if any, capital requirements and general financial condition. The payment of any cash dividends will be within the discretion of our Board of Directors at such time. Further if we incur indebtedness, our ability to declare dividends may be further limited by restrictive covenants we may agree to in connection therewith.
ITEM 6. [RESERVED.]
71
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our financial statements and the notes thereto contained elsewhere in this Annual Report. The analysis of the financial condition and results of operations excludes APT as it was acquired after December 31, 2023. Certain information contained in the discussion and analysis set forth below includes forward-looking statements that involve risks and uncertainties. Our actual results may differ materially from those discussed in any forward-looking statement because of various factors, including those described in the sections titled “Cautionary Statement Regarding Forward-Looking Statements” and “Risk Factors” in this Annual Report.
Overview
We are a clinical stage product discovery company developing products using both natural and engineered phage technologies designed to target and kill specific harmful bacteria associated with chronic diseases, such as CF and DFO. Bacteriophage or phage are bacterial, species-specific, strain-limited viruses that infect, amplify and kill the target bacteria and are considered inert to mammalian cells. By utilizing proprietary combinations of naturally occurring phage and by creating novel phage using synthetic biology, we develop phage-based therapies intended to address both large-market and orphan diseases.
Since BiomX Ltd.’s inception in 2015, we have devoted substantially all our resources to organizing and staffing our company, raising capital, acquiring rights to or discovering product candidates, developing our technology platforms, securing related intellectual property rights, and conducting discovery, research and development and clinical activities for our product candidates. We do not have any products approved for sale, and we have not generated any revenue from product sales. As we advance our product candidates, we expect our expenses to remain significant. To date, we have funded our operations with proceeds from sales of our Common Stock, preferred shares and warrants, governmental grants, collaboration agreements and debt. As of December 31, 2023, we had received gross proceeds of approximately $154 million from sales of our securities. In addition, as of December 31, 2023, we have received $2.0 million from our collaboration agreements and recorded a reduction from research and development expenses of $2.2 million. The remainder of $0.2 million was received in January 2024. In addition, through December 31, 2023, we had received an aggregate of $8.0 million in the form of grants from the IIA, of which $1.1 million had been received as of December 31, 2023.
In addition, we have incurred significant operating losses. Our ability to generate revenue from product sales sufficient to achieve profitability will depend on the successful development of, the receipt of regulatory approval for, and eventual commercialization of one or more of our product candidates. Our net losses were approximately $26.2 million and $28.3 million for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023, we had an accumulated deficit of $163 million and expect that for the foreseeable future we will continue to incur significant expenses as we advance our product candidates from discovery through preclinical development and clinical trials and seek regulatory approval of our product candidates. In addition, if we obtain regulatory approval for any of our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution. We may also incur expenses in connection with in-licensing or acquiring additional product candidates.
Because of the numerous risks and uncertainties associated with product development, we are unable to predict the timing or amount of increased expenses or when or if we will be able to achieve or maintain profitability. Even if we are able to generate product sales, we may not become profitable. If we fail to become profitable or are unable to sustain profitability on a continuing basis, we may be unable to continue our operations at planned levels and be forced to reduce or terminate our operations. We may implement cost reduction strategies, which may include amending, delaying, limiting, reducing or terminating one or more of our programs or ongoing or planned clinical trials of our product candidates. In May 2022, we announced, as part of our corporate restructuring plan (the “Corporate Restructuring”), our intention to reduce our operating costs, including a 50% reduction in personnel, while prioritizing our ongoing CF program.
On December 31, 2023, we had cash, cash equivalents and restricted cash of $15.9 million. Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern for at least one year until April 3, 2025, as discussed further below under “-Liquidity and Capital Resources”.
72
On March 6, 2024 we entered into a merger agreement with APT and certain other parties, as a result of which APT became our wholly-owned subsidiary, effective as of March 15, 2024, or the Acquisition. The Acquisition was structured as a stock-for-stock transaction whereby all outstanding equity interests of APT were exchanged in a merger for an aggregate of 9,164,968 shares of BiomX common stock, 40,470 shares of Series X Preferred Stock, convertible upon stockholder approval into 40,470,000 shares of BiomX common stock, and warrants, or the Merger Warrants, exercisable for 2,166,497 shares of BiomX common stock. Upon the consummation of the Acquisition, a successor-in-interest of APT became a wholly-owned subsidiary of BiomX. The Merger Warrants will be exercisable at any time after the date of the receipt of BiomX stockholder approval of their exercise at an exercise price of $5.00 per share and will expire on January 28, 2027.
Concurrently with the consummation of the Acquisition, BiomX consummated a private placement financing, or the March 2024 PIPE, with existing and new investors, resulting in aggregate gross proceeds of approximately $50 million, in which the investors purchased (i) an aggregate of 216,417 shares of Series X Preferred Stock, convertible upon stockholder approval into an aggregate of up to 216,417,000 shares of BiomX common stock, and (ii) warrants, or the Private Placement Warrants, to purchase up to an aggregate of 108,208,500 shares of BiomX common stock, at a combined purchase price of $231.10 per share of Series X Preferred Stock and an accompanying Private Placement Warrant to purchase 500 shares of BiomX common stock. The Private Placement Warrants will be exercisable any time after the date of the receipt of BiomX stockholder approval, at an exercise price of $0.2311 per share, and will expire on the 24-month anniversary of the initial exercisability date.
Immediately following the Acquisition, and without taking into account the shares of Convertible Preferred Stock issued in the March 2024 PIPE, and assuming conversion of all of the Convertible Preferred Stock into Common Stock, our stockholders (including holders of the Pre-Funded Warrants, as defined below) prior to the Acquisition owned approximately 55% of the share capital of the Company and APT’s stockholders prior to the Acquisition owned approximately 45% of the share capital of the Company.
Components of Our Consolidated Results of Operations
Revenue
To date, we have not generated any revenue from product sales and do not expect to generate any revenue from product sales in the near future. If development efforts for our product candidates are successful and result in any necessary regulatory approvals or otherwise lead to any commercialized products or additional license agreements with third parties, we may generate revenue in the future from product sales or payments from collaboration or license agreements with third parties.
Operating Expenses
Research and Development Expenses, net
Research and development expenses consist primarily of costs incurred in connection with the discovery and development of our product candidates. We expense research and development costs as incurred, offset by IIA grants and, to a lesser degree, income from research and development collaboration agreements. These expenses include:
| ● | development and operation of our proprietary platform; | |
|
|
● | expenses incurred in connection with the preclinical and clinical development of our product candidates, including under agreements with third parties, such as CROs and contract manufacturing organizations, as well as consultants, subcontractors and key opinion leaders providing scientific development services; |
| ● | manufacturing scale-up expenses and the cost of acquiring and manufacturing preclinical and clinical trial materials; | |
| ● | license maintenance fees and milestone fees incurred in connection with various license agreements; | |
| ● | employee-related expenses, including salaries, related benefits, travel and stock-based compensation expenses for employees engaged in research and development functions, as well as external costs, such as fees paid to outside consultants engaged in such activities; | |
| ● | costs related to compliance with regulatory requirements and legal fees relating to patent matters; and | |
| ● | depreciation and other expenses. |
We recognize external development costs based on an evaluation of the progress to completion of specific tasks using information provided to us by our service providers.
We do not allocate employee costs or facility expenses, including depreciation or other indirect costs, to specific programs because these costs are deployed across multiple programs and, as such, are not separately classified. We use internal resources primarily to oversee the research and discovery as well as for managing our preclinical development, process development, manufacturing and clinical development activities. These employees work across multiple programs and, therefore, we do not track their costs by program.
73
The table below summarizes our research and development expenses incurred by program:
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| USD In thousands | ||||||||
| BX004 | 8,853 | 3,499 | ||||||
| BX005 | 81 | 1,011 | ||||||
| Salaries and related benefits (including stock-based compensation) | 6,004 | 9,130 | ||||||
| Depreciation | 782 | 909 | ||||||
| Rent and related expenses | 905 | 1,101 | ||||||
| Infrastructure & other unallocated or R&D expenses | 2,410 | 2,017 | ||||||
| Less grants from the IIA and consideration from collaboration agreements | (2,337 | ) | (1,423 | ) | ||||
| Total research and development expenses, net | 16,698 | 16,244 | ||||||
Research and development activities are central to our business. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. Our research and development expenses reflect, among other things, programs that were discontinued or put on hold as well as new development programs. As a result, we expect that our research and development expenses will increase substantially over the next several years, particularly as we increase personnel costs, including stock-based compensation, contractor costs and facilities costs, as we continue to advance the development of our product candidates. We also may incur additional expenses related to milestone and royalty payments payable to third parties with whom we have entered into license agreements to acquire the rights to our product candidates.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries, related benefits and stock-based compensation expenses for personnel in executive, finance, corporate, business development and administrative functions. General and administrative expenses also include legal fees relating corporate and securities matters; professional fees for accounting, tax and audit services; insurance costs; travel expenses; and facility-related expenses, including rent, as well as operating related costs.
We believe that our general and administrative expenses may increase in the future as we integrate the APT operations and support our continued research activities and development of our product candidates. We also anticipate that we will continue to incur significant accounting, audit, legal, regulatory, compliance, directors’ and officers’ insurance costs as well as investor and public relations expenses associated with being a public company. We anticipate the additional costs for these services will increase our general and administrative expenses in the future. Additionally, if and when we believe a regulatory approval of a product candidate appears likely, we anticipate an increase in payroll and expenses as a result of our preparation for commercial operations, especially as it relates to the sales and marketing of our product candidate.
Amortization of intangible assets
Intangible assets consist of in-process research and development, amortized for a period of three years, that started on January 1, 2020 and ended on December 31, 2022.
Other income
Other income consists of proceeds from sub-leasing a portion of our office space in Ness Ziona, Israel starting in August 2022.
Interest expenses
Interest expense consists of interest incurred under the Hercules Loan Agreement (as defined below(. We entered into a Loan and Security Agreement with Hercules Capital, Inc., or Hercules, with respect to a venture debt facility, or the Hercules Loan Agreement. Under the Hercules Loan Agreement, Hercules provided the Company with access to a term loan with an aggregate principal amount of up to $30 million, or the Term Loan Facility. On March 19, 2024, the Company prepaid all of the remaining loan under the Term Loan Facility in a total of $10,428 thousands. The prepayment included an end of term charge of $983 thousands and accrued interest of $69 thousands.
Financial expenses, net
Financial expenses, net consist primarily of income or expenses related to revaluation of foreign currencies and interest income on our bank deposits and money market funds.
74
Results of Operations
Comparison of the Years Ended December 31, 2023 and 2022
The following table summarizes our consolidated results of operations for the years ended December 31, 2023 and 2022:
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| USD In thousands | ||||||||
| R&D expenses, net | 16,698 | 16,244 | ||||||
| Amortization of intangible assets | - | 1,519 | ||||||
| General and administrative expenses | 8,650 | 9,456 | ||||||
| Operating loss | 25,348 | 27,219 | ||||||
| Interest expenses | 2,404 | 2,069 | ||||||
| Finance income, net | (1,249 | ) | (902 | ) | ||||
| Other income | (357 | ) | (134 | ) | ||||
| Tax expenses | 23 | 65 | ||||||
| Net Loss | 26,169 | 28,317 | ||||||
R&D expenses, net (net of grants received from the IIA, and consideration from research collaborations) were $16.7 million for the year ended December 31, 2023, compared to $16.2 million for the year ended December 31, 2022. The increase of $0.5 million, or 3%, in the year ended December 31, 2023 compared to the prior year, is primarily due to the following:
| ● | an increase of $5.3 million primarily due to increased expenses related to conducting the clinical trial of our CF product candidate, BX004; |
| ● | a decrease of $3.1 million in salaries and related expenses and stock-based compensation expenses mainly due to the workforce reduction resulting from the Corporate Restructuring ,as well as, the appreciation of the U.S. dollar against the NIS, which led to reduced salaries and related expenses in our Israeli subsidiary; |
| ● | a decrease of $0.9 million due to pausing in the development of BX005, the product candidate for the treatment of AD; and |
| ● | a decrease of $1.0 million due to increased consideration from research collaborations, which resulted in reduced expenses; |
We recorded grants from the IIA totaling $1.1 million for each of the years ended December 31, 2023 and December 31, 2022.
Amortization of intangible assets ended on December 31, 2022, as the intangible asset was fully amortized.
General and administrative expenses were $8.7 million for the year ended December 31, 2023, compared to $9.5 million for the year ended December 31, 2022. The decrease of $0.8 million, or 8%, is primarily due to a decrease of $0.9 million in the Company’s directors’ and officers’ insurance premium.
Interest expenses were $2.4 million for the year ended December 31, 2023, compared to $2.1 million for the year ended December 31, 2022. The increase of $0.3 million, or 14%, is due to the increase of the U.S. prime rate, which led to increased interest payments under the Hercules Loan Agreement.
Finance income, net was $1.2 million for the year ended December 31, 2023, compared to $0.9 million for the year ended December 31, 2022. The increase of $0.3 million, or 33% is primarily due to rising interest rates, leading to an increase in interest income on our bank deposits. Such increase was partly offset by a decrease due to the appreciation of the U.S. dollar against the NIS, which resulted in higher exchange rate expenses.
Other income was $0.4 million for the year ended December 31, 2023, compared to $0.1 million for the year ended December 31, 2022. The increase of $0.3 million, or 300%, is due to receipt of proceeds from a sublease agreement for a portion of our office space in Ness Ziona, Israel entered into in August 2022 following our Corporate Restructuring.
75
Liquidity and Capital Resources
Sources of Liquidity
We have never generated any revenue from sales of our products and have incurred significant operating losses and negative cash flows from our operations. We have funded our operations to date primarily with proceeds from the sale of our Common Stock, preferred shares and warrants, venture debt, IIA grants and funds from collaboration agreements and through the business combination between Chardan Healthcare Acquisition Corp., a special purpose acquisition company, and BiomX Ltd., pursuant to which Chardan Healthcare Acquisition Corp. changed its name to BiomX Inc. Through December 31, 2023, we had received gross cash proceeds of approximately $154 million from sales of our Common Stock and preferred shares. In August 2021, we borrowed $15.0 million under the Hercules Loan Agreement. In addition, we received approximately $1.9 million from our collaboration agreements and grants from the IIA for each of the years ended December 31, 2023 and December 31, 2022.
Cash in excess of immediate requirements is invested primarily with a view to liquidity and capital preservation.
On December 4, 2020, we filed a shelf registration statement on Form S-3, which was declared effective by the SEC on December 11, 2020. In addition, on December 4, 2020, we entered into an Open Market Sale AgreementSM, or the Sale Agreement, with Jefferies LLC Jefferies, pursuant to which we could issue and sell shares of our Common Stock having an aggregate offering price of up to $50 million from time to time through Jefferies. We were not obligated to make any sales of Common Stock under the Sale Agreement. Through December 31, 2023, we sold an aggregate of 983,384 shares of Common Stock pursuant to the Sale Agreement for aggregate gross proceeds of $5.8 million. We terminated the Sale Agreement on December 7, 2023.
On August 16, 2021 we entered into the Hercules Loan Agreement with Hercules, with respect to a venture debt facility. Under the Hercules Loan Agreement, Hercules provided the us with access to a term loan with an aggregate principal amount of up to $30 million, available in three tranches, subject to certain terms and conditions. The first tranche of $15 million was advanced to us on the date the Hercules Loan Agreement was executed. The milestones for the second and third tranches were not reached and have expired. and accordingly we never received additional amounts under the Hercules Loan Agreement. We were required to make interest-only payments through March 1, 2023, and we were required to repay the principal balance and interest in monthly installments through September 1, 2025. On March 19, 2024, we voluntarily prepaid the outstanding amount under the Hercules Loan Agreement and such agreement expired.
On February 22, 2023, we entered into a securities purchase agreement to issue and sell an aggregate of 15,997,448 shares of our Common Stock and 14,610,714 pre-funded warrants, or the Pre-Funded Warrants, and collectively, the Securities, at a price of $0.245 per share and $0.244 per Pre-Funded Warrant, through a private placement pursuant to an exemption from registration requirements under the Securities Act, or the February 2023 PIPE. The gross proceeds from the February 2023 PIPE were approximately $7.5 million, before deducting issuance costs. The offering closed in two parts. The first closing, which resulted in the issuance of 3,199,491 shares of Common Stock and 2,776,428 Pre-Funded Warrants for gross proceeds of $1.5 million, occurred on February 27, 2023. Such Pre-Funded Warrants became exercisable on February 27, 2023, at an exercise price of $0.001 per share of Common Stock and have no expiration date. At the first closing, we raised net proceeds of $1.3 million, after deducting issuance costs of $0.2 million. On April 24, 2023, our stockholders approved the issuance of up to 24,632,243 shares of Common Stock, including shares underlying Pre-Funded Warrants, in accordance with NYSE American rules. On May 4, 2023, we completed the second closing of the February 2023 PIPE and issued an aggregate of 12,797,957 shares of Common Stock and 11,834,286 Pre-Funded Warrants. Such Pre-Funded Warrants became exercisable on May 4, 2023, at an exercise price of $0.001 per share of Common Stock and have no expiration date. At the second closing, we raised net proceeds of $5.9 million, after deducting issuance costs of $0.1 million. As of December 31, 2023, no Pre-Funded Warrants had been exercised.
On December 7, 2023, we filed a shelf registration statement on Form S-3, which was declared effective by the SEC on January 2, 2024. In addition, on December 7, 2023, we entered into the ATM Agreement, with Wainwright, as manager, pursuant to which we may issue and sell shares of our Common Stock having an aggregate offering price of up to $7.5 million from time to time through Wainwright. We are not obligated to make any sales of Common Stock under the ATM Agreement. From January 1, 2024 through March 26, 2024, we issued 75,179 shares of Common Stock pursuant to the ATM Agreement for aggregate gross proceeds of $19 thousand.
76
On March 15, 2024, in connection with the Acquisition, we consummated the March 2024 PIPE, pursuant to which we sold an aggregate of 216,417 shares of Convertible Preferred Stock and Private Placement Warrants to purchase up to an aggregate of 108,208,500 shares of Common Stock for aggregate gross proceeds of approximately $50 million.
Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern for at least one year until April 3, 2025. In the future, we will likely require or desire additional funds to support our operating expenses and capital requirements or for other purposes, such as acquisitions, and may seek to raise such additional funds through public or private equity or debt financings or collaborative agreements or from other sources, as we are doing now with the ATM Agreement and as we did with the Hercules Loan Agreement. If certain disruptions due to, for instance, the Israel-Hamas War, or Israeli political instability persists and deepens, we could experience an inability to access additional capital, which could in the future negatively affect our capacity to support our operating expenses and capital requirements or to make investments for other purposes, such as acquisitions.
We have no other commitments to obtain additional financing and cannot assure you that additional financing will be available at all or, if available, that such financing would be obtainable on terms favorable to us and would not be dilutive. Our future liquidity and cash requirements will depend on numerous factors, including the introduction of new products as well as the ability to continue to maintain controls over our operating expenditures.
Cash Flows
The following table summarizes our cash flows for each of the periods presented:
| Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| USD In thousands | ||||||||
| Net cash used in operating activities | (21,286 | ) | (29,092 | ) | ||||
| Net cash provided by (used in) investing activities | 1,951 | (2,107 | ) | |||||
| Net cash provided by financing activities | 2,899 | 292 | ||||||
| Effect of exchange rate changes on cash and cash equivalents and restricted cash | 6 | 106 | ||||||
| Net increase (decrease) in cash and cash equivalents | (16,430 | ) | (30,801 | ) | ||||
77
Operating Activities
During the year ended December 31, 2023, operating activities used $21.3 million of net cash, primarily due to a net loss of $26.2 million and by net cash used by changes in our operating assets and liabilities of $2.5 million and non-cash charges of $2.4 million. Non-cash charges for the year ended December 31, 2023, mainly consisted of stock-based compensation expenses of $1.0 million, depreciation and amortization of $0.9 million and amortization of debt issuance costs of $0.6 million. Net changes in our operating assets and liabilities for the year ended December 31, 2023, consisted primarily of an increase in trade account payables of $0.6 million and an increase in other account payables of $1.2 million, partially offset by a decrease in other current assets of $0.8 million.
During the year ended December 31, 2022, operating activities used $29.1 million of net cash, primarily due to a net loss of $28.3 million and by net cash used by changes in our operating assets and liabilities of $4.4 million and non-cash charges of $3.7 million. Non-cash charges for the year ended December 31, 2022, mainly consisted of stock-based compensation expenses of $1.5 million and depreciation and amortization of $2.5 million. Net changes in our operating assets and liabilities for the year ended December 31, 2022, consisted primarily of a decrease in trade account payables of $2.0 million and a decrease in other account payables of $3.3 million, partially offset by a decrease in other current assets of $1.0 million.
Investing Activities
During the year ended December 31, 2023, investment activities used in net cash of $2.0 million, proceeds from withdrawal of short-term deposits of $2.0 million.
During the year ended December 31, 2022, investing activities used in net cash of $2.1 million, mainly consisting of investment in short-term deposits of $13.5 million, partially offset by proceeds from withdrawal of short-term deposits of $11.5 million.
We have invested, and plan to continue to invest, our existing cash in short-term investments in accordance with our investment policy. These investments may include money market funds and investment securities consisting of U.S. Treasury notes, and high quality, marketable debt instruments of corporations and government sponsored enterprises. We use foreign exchange contracts (mainly option and forward contracts) to hedge balance sheet items from currency exposure. These foreign exchange contracts are not designated as hedging instruments for accounting purposes. In connection with these foreign exchange contracts, we recognize gains or losses that offset the revaluation of the balance sheet items also recorded under financial expenses, net. As of December 31, 2023, we had outstanding foreign exchange contracts in the amount of approximately $4.1 million with a fair value asset of $0.3 million. As of December 31, 2022, we had outstanding foreign exchange contracts in the amount of approximately $4.5 million, with a fair value liability of $55,000.
Financing Activities
During the year ended December 31, 2023, financing activities provided net cash of $3.0 million, mainly consisting of $7.2 million due to issuances of Common Stock under the February 2023 PIPE, net of issuance costs, partially offset by the repayment of long-term debt of $4.3 million under the Hercules Loan Agreement.
During the year ended December 31, 2022, financing activities provided net cash of $0.3 million, mainly consisting of $0.3 million due to issuances of Common Stock under the Sale Agreement.
Contractual Obligations, Commitments and Contingencies
Our contractual obligations and commitments relate primarily to our Hercules Loan Agreement, operating leases and non-cancelable purchase obligations under agreements with various research and development organizations and suppliers in the ordinary course of business. In September 2020, we entered into a lease agreement for new office and laboratory space in Ness Ziona, Israel.
In the normal course of business, we enter into contracts and agreements that contain a variety of representations and warranties and provide for general indemnifications. Our exposure under these agreements is unknown because it involves claims that may be made against us in the future but have not yet been made. To date, we have not paid any claims or been required to defend any action related to our indemnification obligations. However, we may record charges in the future as a result of these indemnification obligations.
In accordance with our certificate of incorporation and bylaws, as well as contractual indemnification agreements, we have potential indemnification obligations to our officers and directors for specified events or occurrences, subject to some limits, while they are serving at our request in such capacities. There have been no claims to date, and we have director and officer insurance that may enable us to recover a portion of any amounts paid for future potential claims.
78
Government Grants and Related Royalties
The Government of Israel, through the IIA, encourages research and development projects by providing grants. We may receive grants from the IIA at the rates that range from 20% to 50% of the research and development expenses, as prescribed by the research committee of the IIA. Through December 31, 2023, we had received an aggregate of $8.0 million in the form of grants from the IIA. BiomX Ltd was formed as an incubator company as part of the FutuRx incubator, and, until 2017, the majority of its funding was from IIA grants and funding by the incubator, which is supported by the IIA. We continued to apply for and receive IIA grants after we left the incubator. The requirements and restrictions for such grants are found in the Research Law. Under the Research Law, royalties of 3% to 3.5% on the revenue derived from sales of products or services developed in whole or in part using these IIA grants are payable to the Israeli government. We developed both of our platform technologies, at least in part, with funds from these grants, and, accordingly, we would be obligated to pay these royalties on sales of any of our product candidates that achieve regulatory approval.
Below is a description of our obligations in connection with the grants received from the IIA under the Research Law:
Local Manufacturing Obligation
As long as the manufacturing of our product candidates takes place in Israel and no technology funded with IIA grants is sold or out licensed to a non-Israeli entity, the maximum aggregate royalties paid generally would not exceed 100% of the grants made to us, plus annual interest equal to the 12-month SOFR applicable to U.S. dollar deposits, as published on the first business day of each calendar year.
Under the terms of the Research Law, the products may be manufactured outside of Israel by us or by another entity only if prior approval is received from the IIA (such approval is not required for the transfer of up to 10% of the manufacturing capacity in the aggregate, in which case a notice must be provided to the IIA and not be objected to by the IIA within 30 days of such notice).
Know-How Transfer Limitation
The Research Law restricts the ability to transfer know-how funded by the IIA outside of Israel. Transfer of IIA funded know-how outside of Israel requires prior approval of the IIA and may be subject to payments to the IIA, calculated according to formulae provided under the Research Law. The redemption fee is subject to a cap of six times the total amount of the IIA grants, plus interest accrued thereon (i.e. the total liability to the IIA, including accrued interest, multiplied by six). If we wish to transfer IIA funded know-how, the terms for approval will be determined according to the nature of the transaction and the consideration paid to us in connection with such transfer.
Approval of transfer of IIA funded know-how to another Israeli company may be granted only if the recipient abides by the provisions of the Research Law and related regulations, including the restrictions on the transfer of know-how and manufacturing rights outside of Israel.
Change of Control
Any non-Israeli citizen, resident or entity that, among other things, (i) becomes a holder of 5% or more of our share capital or voting rights, (ii) is entitled to appoint our directors or our chief executive officer or (iii) serves as one of our directors or as our chief executive officer (including holders of 25% or more of the voting power, equity or the right to nominate directors in such direct holder, if applicable) is required to notify the IIA and undertake to comply with the rules and regulations applicable to the grant programs of the IIA, including the restrictions on transfer described above.
Approval to manufacture products outside of Israel or consent to the transfer of IIA funded know-how, if requested, is within the discretion of the IIA. Furthermore, the IIA may impose certain conditions on any arrangement under which it permits us to transfer IIA funded know-how or manufacturing out of Israel.
The consideration available to our shareholders in a future transaction involving the transfer outside of Israel of know-how developed with IIA funding (such as a merger or similar transaction) may be reduced by any amounts that we are required to pay to the IIA.
As of December 31, 2023, no sales were generated and the balance of the principal and interest in respect of our commitments for future payments to the IIA totaled approximately $7.9 million, as compared to $6.6 million as of December 31, 2022. As part of funding our current and planned product development activities, we may submit follow-up grant applications for new grants.
79
Outlook
We expect our expenses to remain substantially in the same level in connection with our ongoing activities. Our expenses will remain substantial and may also increase as we:
| ● | continue the development of our product candidates; |
| ● | complete IND-enabling activities and prepare to initiate clinical trials for our product candidates; |
| ● | work to integrate the business of APT; |
| ● | initiate additional clinical trials and preclinical studies for product candidates in our pipeline; |
| ● | seek to identify and develop or in-license or acquire additional product candidates and technologies; |
| ● | seek regulatory approvals for our product candidates that successfully complete clinical trials, if any; |
| ● | establish a sales, marketing and distribution infrastructure to commercialize any product candidates for which we may obtain regulatory approval; | |
| ● | hire and retain additional personnel, such as clinical, quality control, commercial and scientific personnel; and | |
| ● | expand our infrastructure and facilities to accommodate our growing employee base, including adding equipment and physical infrastructure to support our research and development. |
Our financial statements contain an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern for at least one year until April 3, 2025. We have based these estimates on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect. If we receive regulatory approval for our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, sales, marketing and distribution, depending on where we choose to commercialize.
Until such time, if ever, that we can generate product revenue sufficient to achieve profitability, we expect to finance our cash needs through public or private sales of our equity, including under the ATM Agreement, loans, milestone payments, possibly additional grants from the IIA or other government or non-profit institutions and other outside funding sources. Our ability to raise additional capital in the equity and debt markets is dependent on a number of factors including, but not limited to, market volatility resulting from the, Israel-Hamas War, other armed conflicts such as in Ukraine or other disruptions, and market demand for our securities, which itself is subject to a number of development and business risks and uncertainties, as well as the uncertainty that we would be able to raise such additional capital at a price or on terms that are favorable to the Company. To the extent that we raise additional capital through the sale of equity or convertible debt securities, our stockholders’ ownership interests may be materially diluted, and the terms of such securities could include liquidation or other preferences that adversely affect their rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through government and other third-party funding, collaboration agreements, strategic alliances, licensing arrangements or marketing and distribution arrangements, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market products or product candidates that we would otherwise prefer to develop and market by ourselves. For more information regarding the risks related to our outlook, see “Risk Factors — Risks Related to Our Business, Technology and Industry.”
80
Foreign Exchange Contracts
We entered into forward and option contracts to hedge against the risk of overall changes in future cash flow from payments of salaries and related expenses, as well as other expenses denominated in NIS. As of December 31, 2023 and 2022, we had outstanding foreign exchange contracts in the nominal amount of approximately $4.1 million and $4.5 million, respectively.
Critical Accounting Estimates
Our consolidated financial statements are prepared in accordance with US GAAP. The preparation of our consolidated financial statements and related disclosures requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenue, costs and expenses, and the disclosure of contingent assets and liabilities in our financial statements. We base our estimates on historical experience, known trends and events and various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in note 2 to our consolidated financial statements, we believe that the following accounting policies are those most critical to the judgments and estimates used in the preparation of our consolidated financial statements.
Accrued research and development expenses
As part of the process of preparing our consolidated financial statements, we are required to estimate our accrued research and development expenses. This process involves reviewing open contracts and purchase orders, communicating with our applicable personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of actual costs. The majority of our service providers invoice us in arrears for services performed, on a pre-determined schedule or when contractual milestones are met; however, some require advance payments. We make estimates of our accrued expenses as of each balance sheet date in the consolidated financial statements based on facts and circumstances known to us at that time. We periodically confirm the accuracy of these estimates with the service providers and make adjustments, if necessary. Examples of estimated accrued research and development expenses include fees paid to:
| ● | vendors in connection with preclinical development activities; |
| ● | CROs and investigative sites in connection with preclinical and clinical trials; and |
| ● | subcontractors in connection with the manufacturing of materials for preclinical and clinical trials. |
We measure the expense recognized based on our estimates of the services received and efforts expended pursuant to quotes and contracts with multiple CROs and subcontractors that supply, conduct and manage preclinical studies, human clinical studies and clinical trials on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. There may be instances in which payments made to our vendors will exceed the level of services provided and result in a prepayment of the expense. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of certain milestones. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, we adjust the accrual or the amount of prepaid expenses accordingly. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and may result in changes in estimates that increase or decrease amounts recognized in any particular period. To date, there have not been any material adjustments to our prior estimates of accrued research and development expenses.
Stock-Based Compensation
We apply ASC 718-10, “Stock-Based Payment,” which requires the measurement and recognition of compensation expenses for all stock-based payment awards made to employees and directors, including employee stock options under our stock plans based on estimated fair values.
ASC 718-10 requires that we estimate the fair value of equity-based payment awards on the date of grant using an option-pricing model. The fair value of the award is recognized as an expense over the requisite service periods in our Consolidated Statements of Operations. We recognize stock-based award forfeitures as they occur, rather than estimate by applying a forfeiture rate.
81
We recognize compensation expenses for the fair value of non-employee awards over the requisite service period of each award.
We estimate the fair value of stock options granted as equity awards using a Black-Scholes options pricing model. The option-pricing model requires a number of assumptions, of which the most significant are share price, expected volatility and the expected option term (the time from the grant date until the options are exercised or expire). We determine the fair value per share of the underlying stock by taking into consideration our most recent sales of stock. BiomX Ltd. has historically been a private company and lacks company-specific historical and implied volatility information of its stock. We used an average historical stock price volatility based on a combined weighted average of our historical average volatility and that of a selected peer group of comparable public companies within the biotechnology and pharmaceutical industry that were deemed to be representative of future stock price trends as we do not have a sufficient historical trading history of our Common Stock. We will continue to apply this process until a sufficient amount of historical information regarding the volatility of our stock price becomes available. We have historically not paid dividends and has no foreseeable plans to issue dividends. The risk-free interest rate is based on the yield from governmental zero-coupon bonds with an equivalent term. The expected option term is calculated for all stock option grants using the “simplified” method. Changes in the determination of each of the inputs can affect the fair value of the options granted and the results of our operations.
Intangible assets
In-process research and development acquired in a business combination were recognized at fair value as of the acquisition date and subsequently accounted for as indefinite-lived intangible assets until completion or abandonment of the associated research and development efforts.
We accounted for the acquisition of RondinX Ltd. using the acquisition method of accounting, which required us to estimate the fair values of the assets acquired and liabilities assumed. This included acquired in-process research and development and contingent consideration. Adjustments to the fair value of contingent consideration are recorded in earnings. On January 1, 2020, the in-process R&D efforts were completed. The Company had determined the useful life of the R&D assets for three years and began amortizing these assets accordingly in the financial statements. During the year ended on December 31, 2022 we recorded amortization expenses of $1.5 million. As of December 31, 2022, the intangible asset was fully amortized.
82
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
As a smaller reporting company, we are not required to make disclosures under this Item.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
Our financial statements and the notes thereto begin on page F-1 of this Annual Report.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and our Interim Chief Financial Officer (our principal executive officer and principal financial officer, respectively), performed an evaluation of the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2023. Based on the aforementioned evaluation, our management has concluded that our disclosure controls and procedures were effective at a reasonable assurance level as of December 31, 2023.
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Our internal control over financial reporting has been designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles in the United States of America.
Our internal control over financial reporting includes policies and procedures that pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect transactions and dispositions of our assets; provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles in the United States of America, and that receipts and expenditures are being made only in accordance with authorization of our management and directors; and provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on our financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
83
Management assessed the effectiveness of our internal control over financial reporting on December 31, 2023. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission 2013 framework, in Internal Control—Integrated Framework. Based on that assessment under those criteria, management has determined that, as of December 31, 2023, our internal control over financial reporting was effective.
We are exempt from this requirement to provide an attestation report of our independent registered public accounting firm regarding internal control over financial reporting due to our status under the Exchange Act as a non-accelerated filer as of the current time Changes in Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting (as such term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the fourth quarter of fiscal year 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
Trading Arrangements
During the
three months ended December 31, 2023, none of our directors or officers
Ratification of Stock Issuance
On April 2, 2024, our Board of Directors adopted resolutions, or the Resolutions, approving the ratification of the issuance of one share of Common Stock issued in connection with the consummation of the Acquisition pursuant to Section 204 of the Delaware General Corporation Law, or the Ratification. A copy of the Resolutions adopted by our Board of Directors setting forth the information with respect to the Ratification required under Section 204 of the Delaware General Corporation Law is set forth in Exhibit 99.1 to this Annual Report. Any claim that any defective corporate act or putative stock ratified pursuant to the Ratification is void or voidable due to the failure of authorization specified in the Resolutions, or that the Delaware Court of Chancery should declare in its discretion that the Ratification in accordance with Section 204 of the Delaware General Corporation Law not be effective, or be effective only on certain conditions, must be brought within 120 days from the giving of this notice (which is deemed to be given on the date that this Annual Report is filed with the SEC).
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICITIONS THAT PREVENT INSPECTIONS
Not applicable.
84
part III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Set forth below are the names, ages and positions of each of the individuals who serve as our executive officers and member of the Board of Directors, or Board, as of April 3, 2024.
| Name | Age | Position | ||
| Executive Officers | ||||
| Jonathan Solomon | 47 | Chief Executive Officer and Director | ||
| Assaf Oron | 49 | Chief Business Officer | ||
| Marina Wolfson | 39 | Chief Financial Officer | ||
| Avraham Gabay | 39 | Interim Chief Financial Officer | ||
| Dr. Merav Bassan | 58 | Chief Development Officer | ||
| Non-Employee Directors | ||||
| Dr. Russell Greig(1)(2)(3) | 71 | Director and Chairman of the Board of Directors | ||
| Jonathan Leff(2) | 55 | Director | ||
| Dr. Alan Moses(2) | 74 | Director | ||
| Gregory Merril (3) | 58 | Director | ||
| Edward Williams(1) | 67 | Director | ||
| Dr. Jesse Goodman(3) | 72 | Director |
| (1) | Member of the audit committee |
| (2) | Member of the compensation committee |
| (3) | Member of the nominating and corporate governance committee |
Executive Officers
Jonathan Solomon has served as the Chief Executive Officer and as a director of the Company since October 2019. Mr. Solomon served as Board member of BiomX Ltd., or BiomX Israel, from February 2016 and also as Chief Executive Officer from February 2017 to October 2019. From July 2007 to December 2015, Mr. Solomon was a co-founder, President, and Chief Executive Officer of ProClara Biosciences Inc. (formerly NeuroPhage Pharmaceuticals Inc.), a biotechnology company pioneering an approach to treating neurodegenerative diseases. Prior to joining ProClara, he served for ten years in a classified military unit of the Israeli Defense Forces. Mr. Solomon holds B.Sc. magna cum laude in Physics and Mathematics from the Hebrew University, an M.Sc. summa cum laude in Electrical Engineering from Tel Aviv University, and an MBA with honors from the Harvard Business School.
We believe that Mr. Solomon’s qualifications to sit on our Board include his extensive board and management experience in the biotech industry.
Assaf Oron has served as the Chief Business Officer of the Company since October 2019. Mr. Oron served as Chief Business Officer of BiomX Ltd. from January 2017 to October 2019. Prior to this position, he served in various roles at Evogene Ltd. (Nasdaq:EVGN), an agriculture biotechnology company, which utilizes a proprietary integrated technology infrastructure to enhance seed traits underlying crop productivity, from March 2006 to December 2016, including Executive Vice President of Strategy and Business Development and Executive Vice President of Corporate Development. Prior to joining Evogene, Mr. Oron served as Chief Executive Officer of ChondroSite Ltd., a biotechnology company that develops engineered tissue products in the field of orthopedics and as a senior project manager and strategic consultant at Israeli management consulting company POC Ltd. Mr. Oron holds an M.Sc. in Biology (bioinformatics) and a B.Sc. in Chemistry and Economics, both from Tel Aviv University.
Marina Wolfson has served as the Chief Financial Officer of the Company since April 2022 and is currently on a maternity leave. Ms. Wolfson served in several finance and operations roles in the Company from December 2019 to March 2022. Ms. Wolfson’s experience includes working with large pharmaceutical and hi-tech companies, as well as venture capital funds. Prior to joining the Company, Ms. Wolfson worked as Vice President of Finance at BioView Ltd. (TASE:BIOV) from 2010 to 2019 and a senior auditor at Ernst & Young, from 2007 to 2010. Ms. Wolfson is a certified public accountant in Israel and holds a B.A in Economics and Accounting (with honors) and an MBA (with honors, specializing in finance) from Ben-Gurion University.
85
Avraham Gabay has served as the Company’s interim Chief Financial Officer, since the commencement of the maternity leave of Ms. Wolfson, the Company’s Chief Financial Officer, in November 2023, and will serve in that role for as long as Ms. Wolfson is on such leave. Prior to his appointment, from 2021 until 2023, Mr. Gabay served as the chief financial officer at Oravax Inc., a biotechnology company focusing on research and development of an oral vaccine. Prior to that, from 2019 until 2021, Mr. Gabay was the chief financial officer at Oramed Pharmaceuticals Inc. (Nasdaq: ORMP), which is developing an oral delivery platform for proteins and focusing on oral insulin. From 2015 to 2019, Mr. Gabay served as a corporate controller at Orcam Technologies Ltd., a company which develops, manufactures and sells a wearable assistive technology device for people who are blind, visually impaired or have reading or other disabilities. From 2014 to 2015, Mr. Gabay provided economic services in the advisory department of KPMG Israel, a certified public accounting firm, and from 2013 to 2014, he worked in the tax department of the law firm, Gornitzky & Co. In addition, Mr. Gabay serves as a director on the board of Nala Digital Ltd., a public company whose shares are listed for trading on the Tel Aviv Stock Exchange. Mr. Gabay holds a bachelor’s degree in law and accounting (magna cum-laude) from Tel-Aviv University and is a certified public accountant in Israel and a member of the Israeli Bar Association.
Dr. Merav Bassan has served as the Chief Development Officer of the Company since October 2019. Prior to this position, she served in various development roles at Teva Pharmaceutical Industries Limited between 2005 and 2019, including Vice President, Head of Translational Sciences, Specialty Clinical Development R&D from 2017 to 2019, Vice President, Pain and Global Internal Medicine, Project Leadership, Innovative Product Development, Global IR&D from 2015 to 2017, and Project Champion, Senior Director, Innovative Product Development, Global IR&D from 2009 to 2015. Dr. Bassan holds a B.Sc. in Biology, a M.Sc. in Human Genetics and a Ph.D. in Neurobiology from Tel Aviv University, and she completed a Post-Doctoral Fellowship in Neuroscience at Harvard Medical School at Harvard University.
Directors
The biography of Mr. Solomon is set forth above under the header “Executive Officers.” The biographies of our non-employee directors are set forth below:
Dr. Russell Greig has served as a director and chairman of the Board of the Company since October 2019. Dr. Greig has more than 44 years of experience in the pharmaceutical industry, with knowledge and expertise in research and development, business development and commercial operations. He spent the majority of his career at GlaxoSmithKline, or GSK, where he held a number of positions including GSK’s President of Pharmaceuticals International from 2003 to 2008 and Senior Vice President Worldwide Business Development. From 2008 to 2010, Dr. Greig was also President of SR One, GSK’s corporate venture group. He is currently Chairman of Cardior (Germany), Nucleome Therapeutics (UK) and BiomX (NYSE). In addition, Dr. Greig previously served on the boards of Sanifit (Spain) (acquired by Vifor Pharma AG (SWX: VIFN), Tigenix N.V. (acquired by Takeda Pharmaceutical Company Limited), Ablynx N.V. (acquired by Sanofi, France) and Merus N.V. (Nasdaq: MRUS). He was previously Chairman of Syntaxin Ltd (UK) (acquired by Ipsen), Novagali Pharma S.A. (France) (acquired by Santen Pharmaceutical Co., Ltd.), and Isconova AB (Sweden) (acquired by Novavax, Inc. (Nasdaq: NVAX). He served as acting Chief Executive Officer at Genocea Biosciences (Nasdaq: GNCA) and Isconova AB for an interim period. He was also a member of the Scottish Scientific Advisory Committee, reporting to the First Minister of Scotland.
We believe that Dr. Greig’s qualifications to sit on our Board include his extensive board and leadership experience in business development and in drug research and development in the pharmaceutical industry.
86
Jonathan Leff has served as a director of the Company since March 2024. Mr. Leff is a Partner at Deerfield Management Company, L.P., or Deerfield and Chairman of the Deerfield Institute. He joined Deerfield in 2013 and focuses on venture capital and structured investments in biotechnology and pharmaceuticals. Prior thereto, Mr. Leff served as Managing Director at Warburg Pincus LLC from 2000 to 2012, where he led the firm’s investment efforts in biotechnology and pharmaceuticals. Mr. Leff also previously served as a member of the Executive Committee of the Board of the National Venture Capital Association, or NVCA, and led NVCA’s life sciences industry efforts as Chair of NVCA’s Medical Innovation and Competitiveness Coalition. He also served on the Emerging Companies Section Board of the Biotechnology Industry Organization. Mr. Leff is involved in the governance of several not-for-profit organizations, including serving as a member of the board of directors of the Spinal Muscular Atrophy Foundation and sitting on the Columbia University Medical Center Board of Advisors. He currently serves on the board of directors of Larimar Therapeutics, Inc., a publicly traded biotechnology company. Mr. Leff also previously served on the boards of several other publicly traded biotechnology and pharmaceutical companies, including ARS Pharmaceuticals, Inc., from 2022 to 2023, Proteon Therapeutics, Inc. from 2017 to 2019, AveXis, Inc. from 2014 to 2017 and Nivalis Therapeutics, Inc. from 2014 to 2016. He currently serves on the boards of several private biopharmaceutical companies and has previously served on the boards of other privately held biopharmaceutical companies. Mr. Leff received his A.B. from Harvard University, MBA from the Stanford University Graduate School of Business and M.S. in Biotechnology from Johns Hopkins University.
We believe that Mr. Leff’s qualifications to sit on our Board include his extensive board and leadership experience in capital markets and the pharmaceutical and biotech industries.
Dr. Alan Moses has served as a director of the Company since October 2020. Dr. Moses has been a Board member of Chemomab Therapeutics, Ltd. (Nasdaq: CMMB) since March 2021. Dr. Moses served as the Global Chief Medical Officer of Novo Nordisk A/S from 2013 until his retirement in 2018. Prior to that he served in various roles at Novo Nordisk A/S since 2004, beginning as Associate Vice President of Medical Affairs in the United States. Throughout his career, Dr. Moses has specialized in developing novel therapeutics and diagnostics for diabetes mellitus. He co-founded and directed the Clinical Investigator Training Program at Beth Israel Deaconess-Harvard Medical School-MIT. From 1998 to 2004, Dr. Moses served as Senior Vice President and Chief Medical Officer of the Joslin Diabetes Center with specific responsibility for the Joslin Clinic. He now serves as a member of the Board of Joslin Diabetes Center since December 2021. He also serves as Chairman of the Board of the nonprofit diaTribe Foundation and is a member of the Board of the Greater New England Chapter of the Juvenile Diabetes Research Foundation. Dr. Moses earned his MD from the Washington University School of Medicine in St. Louis, worked for three years at the National Institutes of Health, completed his clinical endocrine/diabetes training at Tufts New England Medical Center, and studied Health Care Strategy at Harvard Business School.
We believe that Dr. Moses’s qualifications to sit on our Board include his extensive leadership experience in clinical development in the pharmaceutical industry.
Gregory Merril has served as a director of the Company since March 2024. Mr. Merril founded APT in October 2016, and served as its Chief Executive Officer until October 2023 and served on its board of directors until March 2024. Currently, he lends his expertise to various startups, serving in capacities ranging from advisor to executive director. Mr. Merril served as Chief Executive Officer of Yost Labs, a developer of inertial motion sensors used in fields such as physical rehabilitation and drone navigation, from August 2015 to December 2017. Between 2011 and August 2015, he founded and led Brain Sentry, a company dedicated to developing wearable sensors to detect head impacts risking traumatic brain injury in sports including football, hockey, and lacrosse. From October 2009 to February 2011, he served as chief operating officer of Decision Technologies, which supported the U.S. Navy and the Missile Defense Agency with technology acquisitions and deployments. Earlier, as the founding chief executive officer and chair of Interaction Laboratories from March 2002 to October 2009, Merril worked on patents and products that enhanced physical activity in video games and military simulations. Before this, he was the founding Chief Executive Officer of HT Medical Systems, a company focusing on surgical training simulators, which merged with Immersion Corp (NASDAQ: IMMR) in July 2000. Mr. Merril is credited as inventor with 22 issued patents and holds a B.A. in psychobiology from McDaniel College.
We believe that Mr. Merril’s qualifications to sit on our Board include his experience in drug research and development in the pharmaceutical industry.
87
Edward “Eddie” Williams has served as a director of the Company since October 2023. Mr. Williams has served as a member of the board of directors of BioAtla, Inc. (Nasdaq: BCAB), a publicly traded biotechnology company focusing on oncology, since December 2021. From January 2018 to December 2022, he served as a member of the board of directors of Catalyst Biosciences Inc. (Nasdaq: CBIO, now GYRE), a publicly traded biopharmaceutical company. He also currently serves as director on the non-profit healthcare boards of Boone Memorial Health, and Innovative Hematology, Inc.
From March 2020 to September 2022, Mr. Williams held the positions of Special Advisor to the Chief Executive Officer and Interim Chief Commercial Officer of Ascendis Pharma, Inc. (Nasdaq: ASND). Prior to Ascendis, from 2006 to January 2017, Mr. Williams served as Senior Vice President and General Manager of US BioPharmaceuticals at Novo Nordisk, Inc. (NYSE: NVO), a multinational pharmaceutical and biotech company. Prior to Novo, from 2003 to 2006, Mr. Williams served as Vice President of Sales at the Respiratory and Dermatology Business Unit at Novartis Pharmaceuticals Corporation. Mr. Williams started his career in 1981 at The Upjohn Company (Pharmacia & Upjohn), where he later served as Vice President of Sales until July 2001 and then as Regional Vice President of Sales of Northeast Region post-merger with Searle, from July 2001 until May 2003. Mr. Williams holds a B.S. in Biology and Chemistry from the Marshall University, Huntington, WV, and the Grambling State University, Grambling, LA.
We believe that Mr. Williams’s qualifications to sit on our Board include his extensive board and leadership experience, coupled with his successful experiences pre-launch and commercialization of novel compounds in the pharmaceutical industry.
Dr. Jesse Goodman has served as a director of the Company since March 2024. Dr. Goodman has been the director of the Center on Medical Product Access, Safety and Stewardship, and professor of medicine and attending physician in infectious diseases, at Georgetown University since March 2014. Dr. Goodman also is an infectious disease physician at the Washington DC Veterans Affairs and Walter Reed Medical Centers. He serves on the board of directors of GlaxoSmithKline plc, a multinational pharmaceutical company, which he joined in 2016, and chaired that board’s science committee until early 2023, and he has served on the board of directors of Intellia Therapeutics, Inc., a publicly traded biotechnology company, since October 2018. Prior to the Merger Agreement, Dr. Goodman served on the board of directors of APT. He also has served as a president (2015 to 2020) and member (2015 to present) of the board of trustees of the United States Pharmacopeia Convention, Inc. From 2009 until February 2014, Dr. Goodman served as the chief scientist of the FDA. Dr. Goodman also served as deputy commissioner for science and public health at the FDA from 2009 through 2012. Prior to that, Dr. Goodman was the director of the FDA’s Center for Biologics Evaluation and Research from 2003 to 2009 and a senior advisor to the FDA commissioner from 1998 through 2000. Prior to his government service, Dr. Goodman was professor of medicine and chief of infectious diseases at the University of Minnesota. Dr. Goodman has served on numerous advisory boards and committees for national and international health care organizations, including the CDC, the National Institute of Health, the World Health Organization and the Coalition on Epidemic Preparedness Innovations. Dr. Goodman received a B.S. in biology from Harvard College, a master’s in public health from the University of Minnesota and an M.D. from the Albert Einstein College of Medicine, and did his residency and fellowship training in medicine, infectious diseases and oncology at the Hospital of the University of Pennsylvania and at the University of California in Los Angeles, where he was also chief medical resident. He has been elected to the Institute of Medicine of the National Academy of Sciences.
We believe that Dr. Goodman’s qualifications to sit on our Board include his extensive board and leadership experience in clinical development in the pharmaceutical industry and regulation.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that applies to all directors, officers and employees. The Code of Business Conduct and Ethics is available on our website at www.biomx.com. If we make any substantive amendments to the Code of Business Conduct and Ethics or grants any waiver from a provision of the Code to any director or executive officer, we will promptly disclose the nature of the amendment or waiver on our website.
88
Board Committees and Corporate Governance
Board Composition and Leadership Structure
As of April 3, 2024, the Board is comprised of seven members. The Board has a flexible policy with respect to the combination or separation of the offices of Chairman of the Board and Chief Executive Officer. Currently, Dr. Russell Greig serves as our independent Chairman, and Mr. Jonathan Solomon serves as our Chief Executive Officer. The Board believes that by having separate roles, the Chief Executive Officer is able to focus on the day-to-day business and affairs of the Company and the Chairman is able to focus on key strategic issues, board leadership and communication. While the Board believes this leadership structure is currently in the best interests of the Company and its stockholders, the Board also recognizes that future circumstances could lead it to combine these roles.
Board Committees
The Board has established three standing committees: the Audit Committee, the Compensation Committee and the Nominating and Corporate Governance Committee, each of which is composed solely of independent directors, and is described more fully below. Each of the Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee operates pursuant to a written charter and each committee reviews and assesses the adequacy of its charter and submits its charter to the Board for approval. The charters for the Audit Committee, Compensation Committee and Nominating and Corporate Governance Committee are all available on our website, www.biomx.com.
Audit Committee
Our Audit Committee engages the Company’s independent accountants: reviews their independence and performance; reviews the Company’s accounting and financial reporting processes and the integrity of its financial statements; reviews the audits of the Company’s financial statements and the appointment, compensation, qualifications, independence and performance of the Company’s independent auditors; reviews the Company’s compliance with legal and reviews regulatory requirements; and reviews the performance of the Company’s internal audit function and internal control over financial reporting.
The members of the Audit Committee are Dr. Russell Greig and Edward Williams, each of whom is an independent director under NYSE American’s listing standards and satisfies the additional independence requirements of Rule 10A-3 of the Exchange Act. Dr. Russell Greig is the Chairperson of the Audit Committee. The Audit Committee does not currently have as a member an “audit committee financial expert,” as defined under the rules and regulations of the SEC.
Compensation Committee
Our Compensation Committee reviews annually the Company’s corporate performance goals and objectives relevant to the Chief Executive Officer’s compensation, evaluates the Chief Executive Officer’s performance in light of such goals and objectives, determines and approves the Chief Executive Office’s compensation level based on this evaluation; makes recommendations to the Board regarding approval, disapproval, modification, or termination of existing or proposed employee benefit plans; makes recommendations to the Board with respect to the compensation of our executive officers, other than the Chief Executive Officer, and directors; and administers the Company’s incentive-compensation plans and equity-based plans, as well as the Company’s clawback policy. The Compensation Committee has the authority to delegate any of its responsibilities to subcommittees as it may deem appropriate in its sole discretion. The Chief Executive Officer of the Company may not be present during voting or deliberations of the Compensation Committee with respect to his compensation. The Company’s executive officers do not play a role in suggesting their own salaries.
The members of the Compensation Committee are Dr. Alan Moses, Mr. Jonathan Leff and Dr. Russell Greig, each of whom is an independent director under NYSE American’s listing standards. Dr. Alan Moses is the Chairperson of the Compensation Committee.
89
The Compensation Committee retained Aon Solutions UK Limited or Aon, an independent compensation consultant, to provide advice with respect to option exchange and repricing of options under the Chardan Healthcare Acquisition Corp. 2019 Equity Incentive Plan, or the 2019 Plan, and the Company’s 2015 Employee Stock Option Plan , or the 2015 Plan, respectively. Aon’s primary responsibilities for the fiscal year ended December 31, 2023 included identifying the methodology of the repricing and option exchange and providing recommendations to the Compensation Committee, which the Compensation Committee considered among the factors it reviewed when determining such repricing and exchange of options.
Nominating and Governance Committee
Our Nominating and Corporate Governance Committee is responsible for overseeing the selection of persons to be nominated to serve on the Board. Specifically, the Nominating and Corporate Governance Committee makes recommendations to the Board regarding the size and composition of the Board, establishes procedures for the director nomination process and screens and recommends candidates for election to the Board. On an annual basis, the Nominating and Corporate Governance Committee recommends for approval by the Board certain desired qualifications and characteristics for Board membership. Additionally, the Nominating and Corporate Governance Committee establishes and oversees the annual assessment of the performance of the Board as a whole and its individual members. The Nominating and Corporate Governance Committee will consider a number of qualifications relating to management and leadership experience, background and integrity and professionalism in evaluating a person’s candidacy for membership on the Board. Although the Nominating and Corporate Governance Committee does not have a formal policy with regard to the consideration of diversity identifying nominees, the Nominating and Corporate Governance Committee may require certain skills or attributes, such as financial or accounting experience, to meet specific needs of the Board that arise from time to time and will also consider the overall experience and makeup of its members to obtain a broad and diverse mix of Board members. The Nominating and Corporate Governance Committee does not distinguish among nominees recommended by stockholders and other persons.
The members of the Nominating and Corporate Governance Committee are Dr. Russell Greig, Dr. Jesse Goodman and Mr. Gregory Merril, each of whom is an independent director under NYSE American’s listing standards. Dr. Russell Greig is the Chairperson of the Nominating and Corporate Governance Committee.
ITEM 11. EXECUTIVE COMPENSATION
Summary Compensation Table
The following table sets forth the total compensation paid or accrued during the last two fiscal years with respect to (i) our Chief Executive Officer, (ii) our two other most highly compensated executive officers, who each earned more than $100,000 during the fiscal year ended December 31, 2023, and were serving as executive officers as of such date.
| Name and Principal Position | Year | Salary ($)(1) | Bonus ($)(1) | Option Awards(2) ($)(2) | All Other Compensation ($)(1)(3) | Total ($)(1) | ||||||||||||||||
| Jonathan Solomon | 2023 | 412,135 | 201,234 | 404,174 | 100,998 | 1,118,541 | ||||||||||||||||
| Chief Executive Officer | 2022 | 424,581 | - | 512,974 | 103,987 | 1,041,542 | ||||||||||||||||
| Marina Wolfson | 2023 | 214,727 | 76,209 | 90,742 | 46,578 | 428,256 | ||||||||||||||||
| Chief Financial Officer | 2022 | 231,414 | - | 116,827 | 48,002 | 396,243 | ||||||||||||||||
| Dr. Merav Bassan | 2023 | 264,105 | 101,145 | 153,218 | 72,463 | 590,931 | ||||||||||||||||
| Chief Development Officer | 2022 | 280,213 | - | 206,332 | 76,373 | 562,948 | ||||||||||||||||
| (1) | All payments were originally made in NIS and were translated into USD using the annual average USD/NIS exchange rate for each fiscal year. |
| (2) | Amounts in this column represent the grant date fair value of the option awards as computed in accordance with ASC 718, not including any estimates of forfeitures related to service-based vesting conditions. See note 12.B. to our Consolidated Financial Statements for the year ended December 31, 2023 for a discussion of assumptions made by the Company in determining the grant date fair value of our option awards for the fiscal years ended December 31, 2023 and 2022. Note that the amounts reported in this column reflect the accounting cost for these stock options and do not reflect the actual economic value that may be realized by the non-employee directors upon the vesting of the stock options, the exercise of the stock options, or the sale of the Common Stock underlying such stock options. |
| (3) | Amounts in this column represent additional payments for welfare benefits, disability insurance and other customary or mandatory social benefits to employees in Israel. |
90
Narrative Disclosure to the Summary Compensation Table
Option Awards
Prior to the Business Combination, option awards were granted to our named executive officers under the 2015 Plan. Option awards granted to our named executive officers after the closing of the Business Combination are granted pursuant to the 2019 Plan. In each case, one fourth of the options vest and become exercisable on the first anniversary of the grant date, and the remainder of the options vest and become exercisable in 12 equal quarterly instalments, subject to the named executive officer’s continued employment; provided that the options will vest and become exercisable in the event the named executive officer is terminated within the twelve (12) month period following the occurrence of a Change in Control (as defined in the applicable grant agreement) as a result of an involuntary termination without Cause (as defined in the applicable grant agreement) or a voluntary termination with Good Reason (as defined in the applicable grant agreement). Subject to the terms of any employment agreement, the unexercised portion of these awards is generally forfeited by a participant on the date his or her employment is terminated other than due to death or disability. In the event of death or disability, the options become fully exercisable and remain exercisable for a period specified in the applicable award agreement.
Bonus Awards
We have an annual corporate and individual goal-setting and review process for our named executive officers that is the basis for the determination of potential annual bonuses. Each of our named executive officers is eligible for annual performance-based bonuses of up to a specific percentage of their salary, ranging from 40% to 50% subject to approval by the Board or the Compensation Committee. The performance-based bonus is tied to a set of specified corporate and/or individual goals and objectives reviewed and approved by the Board, such as clinical and development milestones, meeting budget and strategic goals, and we conduct an annual performance review to determine the attainment of such goals and objectives. Our management may propose bonus awards to the Board primarily based on such review process. The Compensation Committee makes the final determination of the achievement of both the specified corporate and strategic objectives and the eligibility requirements for and the amount of such bonus awards and recommends a bonus award payout to the Board for approval. For fiscal year 2023, bonuses were accrued based on advancing or development plans, the satisfaction of certain product candidate development milestones and strategic objectives.
Employment Agreements
Below are descriptions of our employment agreements with our named executive officers.
Jonathan Solomon
Pursuant to an employment agreement dated February 1, 2016, by and between BiomX Israel and Mr. Solomon, as the Chief Executive Officer of BiomX Israel, Mr. Solomon is entitled to a base salary of NIS 64,000, or approximately $19,500, per month, and an additional gross payment of NIS 16,000, or approximately $4,900, per month for up to 40 hours per month worked outside of normal business hours and normal business days (together with the base salary, Mr. Solomon’s Salary). Starting April 1, 2023, Mr. Solomon is entitled to a base salary of NIS 100,000, or approximately $27,778, per month, and overtime payment of NIS 25,000 or approximately $6,944, per month.
BiomX Israel also makes customary contributions on Mr. Solomon’s behalf to a pension fund or a managers insurance company, at Mr. Solomon’s election, in an amount equal to 8.33% of his Salary, allocated to a fund for severance pay, and an additional amount equal to 5.00% of the Salary in case Mr. Solomon is insured through a managers insurance policy, or 6.50% of Mr. Solomon’s Salary in case Mr. Solomon is insured through a pension fund, which shall be allocated to a provident fund or pension plan. In case Mr. Solomon chooses to allocate his pension payments to a managers insurance policy (and not a pension fund), the Company shall also insure him under a work disability insurance policy at the rate required to insure 100% of Mr. Solomon’s Salary and for this purpose will contribute an amount of up to 2.50% of Mr. Solomon’s Salary insured in such insurance policy for disability insurance in a policy and/or insurance company. These payments are intended to be in lieu of statutory severance pay that Mr. Solomon would otherwise be entitled to receive from BiomX Israel in accordance with Severance Pay Law 5723-1963, or the Severance Pay Law. BiomX Israel also contributes 7.50% of Mr. Solomon’s monthly salary to a recognized educational fund. BiomX Israel also reimburses Mr. Solomon for automobile maintenance and transportation expenses of NIS 2,000, or $556 per month. Mr. Solomon is also entitled to non-statutory 12 months severance (including social benefits), upon either (i) resignation with a good reason, or (ii) termination without cause (as the terms good reason and cause would be defined by the parties, consistent with our past practice), provided that Mr. Solomon waives all claims and continues to comply with the other terms of his employment agreement.
91
Marina Wolfson
Pursuant to an employment agreement dated December 1, 2019, by and between BiomX Israel and Ms. Wolfson, she serves as our Chief Financial Officer. Ms. Wolfson is entitled to a base salary of NIS 39,600, or approximately $11,400, per month, and an additional gross payment of NIS 7,400, or approximately $2,130, per month for up to 40 hours per month worked outside of normal business hours and normal business days (together with the base salary, Ms. Wolfson’s Salary. Starting May 1, 2020, Ms. Wolfson’s base salary was NIS 40,000 or approximately $11,458, per month, and an additional gross payment of NIS 10,000 or approximately $2,865, per month. Starting April 1, 2023, Ms. Wolfson’s base salary is NIS 54,080 or approximately $15,022, per month, and an additional gross payment of NIS 13,520 or approximately $3,756, per month.
BiomX Israel also makes customary contributions on Ms. Wolfson’s behalf to a pension fund or a managers insurance company, at Ms. Wolfson’s election, in an amount equal to 8.33% of Ms. Wolfson’s Salary, allocated to a fund for severance pay, and an additional amount equal to 5.00% of Ms. Wolfson’s Salary in case Ms. Wolfson is insured through a managers insurance policy, or 6.50% of Ms. Wolfson’s Salary in case Ms. Wolfson is insured through a pension fund, which shall be allocated to a provident fund or pension plan. In case Ms. Wolfson chooses to allocate her pension payments to a managers insurance policy (and not a pension fund), the Company shall also insure her under a work disability insurance policy at the rate required to insure 75% of Ms. Wolfson’s Salary and for this purpose will contribute an amount of up to 2.50% of Ms. Wolfson’s Salary insured in such insurance policy for disability insurance in a policy and/or insurance company. These payments are in lieu of statutory severance pay that Ms. Wolfson would otherwise be entitled to receive from BiomX Israel in accordance with the Severance Law. BiomX Israel also contributes 7.50% of Ms. Wolfson’s monthly Salary (not to exceed NIS 15,712, or approximately $4,364) to a recognized educational fund. The Company reimburses Ms. Wolfson for automobile maintenance and transportation expenses of NIS 2,500, or approximately $694, per month. Ms. Wolfson is also entitled to non-statutory 9 months severance (including social benefits), upon either (i) resignation with a good reason, or (ii) termination without cause (as the terms good reason and cause would be defined by the parties, consistent with our past practice), provided that Ms. Wolfson waives all claims and continues to comply with the other terms of his employment agreement.
Dr. Merav Bassan
Pursuant to an employment agreement dated August 26, 2019, by and between BiomX Israel and Dr. Bassan, as the Chief Development Officer of BiomX Israel, Dr. Bassan is entitled to a base salary of NIS 56,000, or approximately $17,230, per month, and an additional gross payment of NIS 14,000, or approximately $4,307, per month for up to 40 hours per month worked outside of normal business hours and normal business days (together with the base salary, Dr. Bassan’s Salary. Starting April 1, 2023, Dr. Bassan is entitled to a base salary of NIS 62,800, or approximately $17,444, per month, and an additional gross payment of NIS 15,700 or approximately $4,361, per month.
BiomX Israel also makes customary contributions on Dr. Bassan’s behalf to a pension fund or a managers insurance company, at Dr. Bassan’s election, in an amount equal to 8.33% of Dr. Bassan’s Salary, allocated to a fund for severance pay, and an additional amount equal to 7.30% of Dr. Bassan’s Salary in case Dr. Bassan is insured through a managers insurance policy, or 6.50% of Dr. Bassan’s Salary in case Dr. Bassan is insured through a pension fund, which shall be allocated to a provident fund or pension plan. In case Dr. Bassan chooses to allocate her pension payments to a managers insurance policy (and not a pension fund), the Company shall also insure her under a work disability insurance policy at the rate required to insure 75% of Dr. Bassan’s Salary and for this purpose will contribute an amount of up to 2.50% of the Salary insured in such insurance policy for disability insurance in a policy and/or insurance company. These payments are in lieu of statutory severance pay that Dr. Bassan would otherwise be entitled to receive from BiomX Israel in accordance with the Severance Law. BiomX Israel also contributes 7.50% of Dr. Bassan’s monthly Salary to a recognized educational fund. The Company reimburses Dr. Bassan for automobile maintenance and transportation expenses of NIS 2,500, or approximately $694, per month. Dr. Bassan is also entitled to non-statutory 9 months severance (including social benefits), upon either (i) resignation with a good reason, or (ii) termination without cause (as the terms good reason and cause would be defined by the parties, consistent with our past practice), provided that Dr. Bassan waives all claims and continues to comply with the other terms of her employment agreement.
92
Outstanding Equity Awards at 2023 Fiscal Year-End
The following table provides information regarding equity awards held by the named executive officers that were outstanding as of December 31, 2023:
| Option Awards | ||||||||||||||||
| Name | Grant Date | Number of Securities Underlying Unexercised Options Exercisable (1) (#) | Number of Securities Underlying Unexercised Options Unexercisable (1) (#) | Option Exercise Price ($) | Option Expiration Date | |||||||||||
| Jonathan Solomon | 11/13/2016 | 167,434 | - | 0.54 | 01/07/2027 | |||||||||||
| 03/26/2017 (2) | 182,133 | - | 0.275 | 03/26/2027 | ||||||||||||
| 05/22/2018 (2) | 201,718 | - | 0.275 | 05/21/2028 | ||||||||||||
| 03/29/2019 (2) | 284,701 | - | 0.275 | 03/29/2029 | ||||||||||||
| 03/25/2020 (3) | 35,527 | 2,368 | 0.275 | 03/25/2030 | ||||||||||||
| 03/30/2021 (3) | 27,500 | 12,500 | 0.275 | 03/30/2031 | ||||||||||||
| 03/29/2022 (3) | 64,063 | 82,366 | 0.275 | 03/29/2032 | ||||||||||||
| 08/22/2022 | 31,250 | 68,750 | 0.66 | 08/22/2032 | ||||||||||||
| 03/01/2023 | 410,000 | 0.4 | 03/01/2033 | |||||||||||||
| Dr. Merav Bassan | 10/10/2019 (2) | 189,997 | - | 0.275 | 10/10/2029 | |||||||||||
| 03/30/2021 (3) | 8,593 | 3,907 | 0.275 | 03/30/2031 | ||||||||||||
| 03/29/2022 (3) | 31,250 | 40,179 | 0.275 | 03/29/2032 | ||||||||||||
| 08/22/2022 | 23,438 | 51,562 | 0.66 | 08/22/2032 | ||||||||||||
| 03/01/2023 | - | 100,000 | 0.4 | 03/01/2033 | ||||||||||||
| Marina Wolfson | 03/25/2020 (3) | 8,882 | 592 | 0.275 | 03/25/230 | |||||||||||
| 03/30/2021 (3) | 6,017 | 2,733 | 0.275 | 03/30/2031 | ||||||||||||
| 03/29/2022 (3) | 15,625 | 20,090 | 0.275 | 03/29/2032 | ||||||||||||
| 08/22/2022 | 23,438 | 51,562 | 0.66 | 08/22/2032 | ||||||||||||
| 03/01/2023 | - | 100,000 | 0.4 | 03/01/2033 | ||||||||||||
| 29/10/2023 | - | 59,800 | 0.275 | 10/29/2033 | ||||||||||||
| (1) | Unless otherwise indicated, options vest and become exercisable as follows: 25% of the options on the first anniversary of the “vesting commencement date” (as defined in the applicable notice of option grant) and, thereafter, in 12 equal quarterly installments of 6.25% each. |
| (2) | On October 29, 2023, the Board of Directors approved a reduction in the exercise price of each outstanding option to purchase shares of the Company’s Common Stock currently held by employees of the Company with an original exercise price above $0.69 per share granted under the 2015 Plan to $0.275 per share. Other than the exercise price, no other terms of grant of the repriced options were changed; however, the options may not be exercised until one year after the repricing date. |
| (3) | On November 9, 2023, the Company filed with the SEC a Tender Offer Statement defining the terms and conditions of a one-time voluntary stock option exchange of certain eligible options for its employees, or the Option Exchange granted under the 2019 Plan. The Company offered to exchange certain out-of-the-money stock options for new stock options at an exchange ratio of between 1.4 and 3.8 surrendered options for one new option exercisable for shares of common stock with a lower exercise price. On December 11, 2023, the completion date of the Option Exchange, the stock options were tendered by eligible employees, and the Company granted new options at an exercise price of $0.275. |
Compensation of Directors
We maintain a non-employee director compensation policy, pursuant to which each non-employee director receives an annual retainer of $35,000. In addition, our non-employee directors receive the following cash compensation for board services, as applicable:
| ● | the chairman of the Board receives an annual retainer of $100,000 (inclusive of annual committee chairmanship and membership); |
93
| ● | each member of our Audit, Compensation and Nominating and Corporate Governance Committees, other than the chairperson, receives an additional annual retainer of $7,500, $5,000 and $4,000, respectively; and |
| ● | each chairperson of our Audit, Compensation and Nominating and Corporate Governance Committees receives an additional annual retainer of $15,000, $10,000 and $8,000, respectively. |
We pay all amounts in quarterly installments. We also reimburse each of our directors for their reasonable travel, lodging and other out-of-pocket expenses incurred relating to their attendance at Board and committee meetings.
Each non-employee director also receives an annual award of options to purchase our Common Stock. One-fourth of each Annual Option Award vests on the first anniversary of the date of grant, and the remainder of the annual option award vests in 12 equal quarterly installments, subject to such director’s continued service on the Board. The Company’s policy is to grant options based, among other things, on the recommendations of a compensation consultant. In 2023, the Company granted 41,000 options to each non-employee director and 82,000 to the Chairman of the Board.
The following table sets forth information concerning compensation accrued or paid to our independent, non-employee directors during the year ended December 31, 2023 for their service on our Board. Mr. Jonathan Solomon, a director who is also our employee, received no additional compensation for his service as a director and is not set forth in the table below:
| Name | Fees earned or paid in cash ($) | Option Awards(2)(3) | All other compensation | Total ($) | ||||||||||||
| Dr. Russell Greig | 100,500 | 61,077 | - | 161,577 | ||||||||||||
| Michael Dambach(1) | 27,205 | 2,449 | - | 29,654 | ||||||||||||
| Jason Marks(1) | 25,605 | 2,449 | - | 28,054 | ||||||||||||
| Dr. Alan Moses | 47,560 | 33,873 | - | 81,433 | ||||||||||||
| Edward L. Williams | 7,704 | 1,085 | - | 8,789 | ||||||||||||
| Lynne Sullivan(1) | 54,000 | 30,538 | - | 84,538 | ||||||||||||
| 262,574 | 131,471 | - | 394,045 | |||||||||||||
| (1) | Effective as of March 15, 2024, the director resigned and no longer serves on the Board |
| (2) | Amounts in this column represent the grant date fair value of the option awards as computed in accordance with ASC 718, not including any estimates of forfeitures related to service-based vesting conditions. See note 12.B. of the notes to Consolidated Financial Statements in our Annual Report on Form 10-K for the year ended December 31, 2023 for a discussion of assumptions made by the Company in determining the grant date fair value of our option awards for the fiscal years ended December 31, 2022 and 2023. Note that the amounts reported in this column reflect the accounting cost for these stock options and do not reflect the actual economic value that may be realized by the non-employee directors upon the vesting of the stock options, the exercise of the stock options, or the sale of the Common Stock underlying such stock options. |
| (3) | As of December 31, 2023, we had outstanding grants to our non-executive directors aggregating 493,800 options of which 134,675 were exercisable or vested, as the case may be, as follows: |
| Name | Total of options granted | Total of options exercisable and vested | ||||||
| Russell Greig | 185,400 | 68,839 | ||||||
| Michael Dambach | 41,000 | - | ||||||
| Jason Marks | 41,000 | - | ||||||
| Dr. Alan Moses | 92,700 | 31,418 | ||||||
| Edward L. Williams | 41,000 | - | ||||||
| Lynne Sullivan | 92,700 | 34,418 | ||||||
| Total | 493,800 | 134,675 | ||||||
94
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
Securities Authorized for Issuance Under Equity Compensation Plans
We have two equity incentive plans, the 2015 Plan, and the 2019 Plan. Although no shares of our Common Stock are available for future issuance under the 2015 Plan, the 2015 Plan will continue to govern outstanding awards granted thereunder. As of December 31, 2023, options to purchase 2,055,836 shares of our Common Stock remained outstanding under the 2015 Plan.
The 2019 Plan was adopted by the Board of Directors and approved by our stockholders in connection with the Business Combination. As of December 31, 2023, there were 1,011,104 shares of our Common Stock available for issuance under the 2019 Plan. The aggregate number of shares of our Common Stock available for issuance pursuant to the 2019 Plan automatically increases on January 1 of each year, for a period of not more than ten years, commencing on January 1, 2020 and ending on (and including) January 1, 2029, in an amount equal to 4% of the total number of shares of Common Stock outstanding on December 31 of the preceding calendar year. Accordingly, on January 1, 2024, 1,839,197 additional shares of our Common Stock were made available for issuance pursuant to the 2019 Plan.
For additional information regarding the 2015 Plan and the 2019 Plan, as of December 31, 2023, please see Part II – Item 8 – Financial Statements and Supplemental Data – Notes to consolidated financial statements – note 12B – Stock-Based Compensation.
| Equity Compensation Plan Information | ||||||||||||
| December 31, 2023 | ||||||||||||
| Plan category | Number of securities to be issued upon exercise of outstanding options and restricted stock (a) | Weighted- average exercise price of outstanding options and restricted stock (b) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) | |||||||||
| Equity compensation plans approved by security holders | 3,224,871 | 0.68 | 1,011,104 | |||||||||
| Equity compensation plans not approved by security holders | 2,055,840 | 0.32 | - | |||||||||
| Total | 5,280,711 | 0.54 | 1,011,104 | |||||||||
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information regarding the beneficial ownership of our Common Stock as of March 28, 2024 (except as otherwise indicated) based on information obtained from the persons named below, with respect to the beneficial ownership of our Common Stock, by (i) each person known by us to be the beneficial owner of more than 5% of our outstanding Common Stock; (ii) each of our named executive officers and directors; and (iii) all our executive officers and directors as a group. Information with respect to beneficial ownership is based on information furnished to us by each director, executive officer or stockholder who holds more than 5% of our outstanding Common Stock, and Schedules 13G or 13D filed with the SEC, as the case may be, and includes shares of our Common Stock which each beneficial owner has the right to acquire within 60 days of March 28, 2024. Unless otherwise indicated, we believe that all persons named in the table have sole voting and investment power with respect to all Common Stock beneficially owned by them. We have based our calculation of beneficial ownership on 55,220,077 shares of our Common Stock outstanding as of March 28, 2024.
95
| Name and Address of Beneficial Owner(1) | Amount and Nature of Beneficial Ownership |
Percent of Class |
||||||
| OrbiMed Israel GP Ltd. (2) 89 Medinat Hayehudim St. Building E Herzliya 4614001 Israel |
12,577,821 | 19.9 | % | |||||
| Cystic Fibrosis Foundation (3) 4550 Montgomery Ave. Suite 1100N Bethesda, MD 20814 |
9,330,580 | 15.6 | % | |||||
|
Nimble Ventures, LLC (4) 1 Letterman Drive, Building A, Suite 4900, San Francisco, CA 94129(2) |
4,598,189 | 8.3 | % | |||||
|
Deerfield Healthcare Innovations Fund II, L.P. (5) 345 Park Avenue South, 12th Floor, New York, New York 10010 |
3,055,049 | 5.5 | % | |||||
|
Deerfield Private Design Fund V, L.P. (6) 345 Park Avenue South, 12th Floor, New York, New York 10010 |
3,055,049 | 5.5 | % | |||||
|
AMR Action Fund, L.P. (7) 225 Franklin Street, Suite 1750, Boston, MA 02110 |
3,054,870 | 5.5 | % | |||||
|
Telmina Limited (8) 34 Rue de l’athenee, PO Box 393, 1211 Geneva 12, Switzerland |
2,839,714 | 5.1 | % | |||||
| Directors and Named Executive Officers | ||||||||
| Jonathan Solomon(9) | 1,167,096 | 2.1 | % | |||||
| Marina Wolfson (10) | 98,272 | * | ||||||
| Dr. Merav Bassan(11) | 292,900 | * | ||||||
| Dr. Russell Greig(12) | 102,365 | * | ||||||
| Dr. Jesse Goodman | - | - | ||||||
| Jonathan Leff | - | - | ||||||
| Gregory Merril | - | - | ||||||
| Dr. Alan Moses(13) | 54,650 | * | ||||||
| Edward L. Williams | - | - | ||||||
| All directors and executive officers as a group (11 persons) | 2,027,633 | 3.7 | % | |||||
| * | Less than 1%. |
| (1) | Unless otherwise indicated, the business address of each of the individuals is c/o BiomX Inc., 22 Einstein St., 4th Floor, Ness Ziona 7414003, Israel. |
| (2) | This stockholder, together with its affiliates and any other persons acting as a group together with the holder or any of the holder’s affiliated, including OrbiMed Israel BioFund GP Limited Partnership, Carl L. Gordon and Erez Chimovits beneficially own 4,517,589 shares of Common Stock and pre-funded warrants to acquire up to 8,060,232 shares of Common Stock. Excludes (x) 4,327 Series X Non-Voting Convertible Preferred Stock, (y) 290,781 Warrants and (y) 1,220,176 Pre-Funded Warrants, and (z) 2,538,500 warrants to purchase Shares. The Warrants and Pre-Funded Warrants each contain an issuance limitation that prohibits the holder from exercising such Warrants or Pre-Funded Warrants to the extent that after giving effect to such issuance after exercise, the holder (together with the holder’s affiliates and any other persons acting as a group together with the holder or any of the holder’s affiliated, including OrbiMed Israel BioFund GP Limited Partnership, Carl L. Gordon and Erez Chimovits) would beneficially own in excess of 19.9% of the Shares outstanding immediately after giving effect to the issuance of the Shares upon exercise of the warrants, or the Beneficial Ownership Limitation. Each share of Series X Preferred Stock is automatically convertible into 1,000 Shares following approval by the Issuer’s stockholders of such conversion, subject to the Beneficial Ownership Limitation. Based on information contained in the Schedule 13D/A filed with the SEC on March 19, 2024 and on the Company’s records. |
| (3) | Consists of (i) 4,552,315 shares of Common Stock and (ii) 4,778,265 shares of Common Stock issuable upon exercise of a warrant exercisable within 60 days. Excludes (i) 21,635 shares of Series X Non-Voting Convertible Preferred Stock, and (ii) 10,817,500 shares of common stock issuable upon exercise of a warrant, as the Series X Preferred Stock and such warrant will only become convertible or exercisable, as applicable, following approval by the Company’s stockholders. Each share of Series X Preferred Stock is convertible into 1,000 shares of common stock following approval by the Company’s stockholders of such conversion, subject to a beneficial ownership limitation. Based solely on information contained in a Schedule 13G filed with the SEC on March 26, 2024 and on the Company’s records. |
| (4) | Consists of (i) 4,550,000 shares of Common Stock and (ii) warrants to acquire up to 552,041 shares of Common Stock, which contain an issuance limitation that prohibits the holder from exercising the Pre-Funded Warrants to the extent that after giving effect to such issuance after exercise, the holder (together with the holder’s affiliates and any other persons acting as a group together with the holder or any of the holder’s affiliated) would beneficially own in excess of 9.99% of the shares of common stock outstanding immediately after giving effect to the issuance of the shares of common stock issuance upon exercise of the warrants. John H. Burbank III is the control person of Nimble Ventures and, in such capacity, may be deemed to indirectly beneficially own the Shares that Nimble Ventures directly beneficially owns. Based on information contained in the Schedule 13G filed with the SEC on June 23, 2023 and on the Company’s records. |
| (5) | Does not include (i) an aggregate of 53,840,000 shares of Common Stock underlying 53,840 shares of Series X Preferred Stock, which will become convertible into Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions, or (ii) an aggregate of 20,897,175 shares of Common Stock underlying warrants that will become exercisable for Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions. Based solely on information contained in a Schedule 13D filed with the SEC on March 22, 2024 and on the Company’s records. |
| (6) | Does not include (i) an aggregate of 53,840,000 shares of Common Stock underlying 53,840 shares of Series X Preferred Stock, which will become convertible into Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions, or (ii) an aggregate of 20,897,175 shares of Common Stock underlying warrants that will become exercisable for Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions. Based solely on information contained in a Schedule 13D filed with the SEC on March 22, 2024 and on the Company’s records. |
96
| (7) | Does not include (i) an aggregate of 42,337,000 shares of Common Stock underlying 42,337 shares of Series X Preferred Stock, which will become convertible into Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions, or (ii) an aggregate of 15,145,647 shares of Common Stock underlying warrants that will become exercisable for Common Stock (subject to a beneficial ownership limitation), if at all, upon the occurrence of certain conditions. Based solely on information contained in a Schedule 13G filed with the SEC on March 25, 2024 and on the Company’s records. |
| (8) | Consists of 2,839,714 shares of Common Stock. Based solely on information contained in a Schedule 13G filed with the SEC on September 29, 2023 and on the Company’s records. |
| (9) | Consists of 25,000 shares of Common Stock, 25,000 warrants (entitling the holder to acquire up to 18,750 shares of Common Stock), 1,105,444 options that are exercisable and 17,902 additional options that will become exercisable within 60 days of March 28, 2024. |
| (10) | Consists of 3,750 shares of Common Stock, 3,750 warrants (entitling the holder to acquire up to 2,813 shares of Common Stock), 84,242 options that are exercisable and 7,467 additional options that will become exercisable within 60 days of March 28, 2024. |
| (11) | Consists of 282,966 options that are exercisable and 9,934 additional options that will become exercisable within 60 days of March 28, 2024. |
| (12) | Consists of 3,750 shares of Common Stock, 3,750 warrants (entitling the holder to acquire up to 2,813 shares of Common Stock), 91,339 options that are exercisable and 4,463 additional options that will become exercisable within 60 days of March 28, 2024. |
| (13) | Consists of 5,000 shares of Common Stock, 5,000 warrants (entitling the holder to acquire up to 3,750 shares of Common Stock), 42,668 options that are exercisable and 3,232 additional options that will become exercisable within 60 days of March 28, 2024. |
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Director Independence
The NYSE American requires that a majority of the Board be composed of “independent directors,” which is defined generally as a person other than an officer or employee of the Company or its subsidiaries or any other individual having a relationship that, as determined by the Board, would interfere with the exercise of his or her objective judgment and will meet the required standards for independence, as established by the applicable rules and regulations of the NYSE American and the SEC.
Dr. Russell Greig, Dr. Alan Moses, Mr. Edward L. Williams, Mr. Jonathan Leff, Dr. Jesse Goodman and Mr. Gregory Merril are our independent directors.
At least annually, the Board evaluates all relationships between us and each director considering relevant facts and circumstances for the purposes of determining whether a material relationship exists that might signal a potential conflict of interest or otherwise interfere with such director’s ability to satisfy his or her responsibilities as an independent director. Based on this evaluation, our Board will make an annual determination of whether each director is independent within the meaning of NYSE American and the SEC independence standards.
Policies and Procedures Regarding Transactions with Related Parties
Our Related-Person Transactions Policy requires us to avoid, wherever possible, all related party transactions that could result in actual or potential conflicts of interests, except under guidelines approved by the Board (or the Audit Committee). For as long as the Company qualifies as a “smaller reporting company” as defined under Rule 12b-2 under the Exchange Act, a related-person transaction is defined under our Related-Person Transactions Policy as a transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which we and any Related Person (as defined in the policy) are, were or will be participants in which the amount involved exceeds the lesser of $120,000 or one percent of the average of the Company’s total assets at year-end for the last two completed fiscal years, and in which any Related Person had or will have a direct or indirect material interest. If the Company ceases to be a smaller reporting company, a related-person transaction will be defined as a transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which the Company and any Related Person are, were or will be participants in which the amount involved exceeds $120,000, and in which any Related Person had or will have a direct or indirect material interest. Transactions involving compensation for services provided to us as an employee, consultant or director are not considered related-person transactions under this policy.
97
In the event that the Company proposes to enter into, or materially amend, a related-person transaction, management of the Company shall present such related-person transaction to the Audit Committee for review, consideration and approval or ratification. The presentation must include, to the extent reasonably available, a description of (a) all of the parties thereto, (b) the interests, direct or indirect, of any Related Person(s) in the transaction in sufficient detail so as to enable the Audit Committee to fully assess such interests, (c) the purpose of the transaction, (d) all of the material facts of the proposed related-person transaction, including the proposed aggregate value of such transaction, or, in the case of indebtedness, the amount of principal that would be involved, (e) the benefits to the Company of the proposed related-person transaction, (f) if applicable, the availability of other sources of comparable products or services, (g) an assessment of whether the proposed related-person transaction is on terms that are comparable to the terms available to or from, as the case may be, unrelated third parties that would have been negotiated at arm’s length, and (h) management’s recommendation with respect to the proposed related-person transaction knowing that there is a potential or actual conflict that will arise of the matter proceeds to fruition. In the event the Audit Committee is asked to consider whether to ratify an ongoing related-person transaction, in addition to the information identified above, the presentation must include (i) a description of the extent of work performed and remaining to be performed in connection with the transaction, (ii) an assessment of the potential risks and costs of termination of the transaction, and (iii) where appropriate, the possibility of modification of the transaction.
The Committee, in approving or rejecting the proposed related-person transaction, will consider all the relevant facts and circumstances deemed relevant by and available to the Committee, including but not limited to (a) the risks, costs and benefits to the Company, (b) the impact on a director’s independence in the event the Related Person is a director, immediate family member of a director or an entity with which a director is affiliated, (c) the terms and timing of the transaction, (d) the availability of other sources of comparable services or products, (e) the terms available to or from, as the case may be, unrelated third parties, and (f) how the related-person transaction was realized and communicated to the Audit Committee as required under the Related-Person Transactions Policy. The Audit Committee will approve only those related-person transactions that, in light of known circumstances, are in, or are not inconsistent with, the best interests of the Company and its stockholders, as the Audit Committee determines in the good faith exercise of its discretion.
Other than compensation, termination, change in control and other arrangements, which are described in Item 11 – Executive Compensation and Item 12 – Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters, our only related-person transactions since January 1, 2023 consisted of (i) a Securities Purchase Agreement we entered into on February 22, 2023 with accredited and non-U.S. investors, including the Cystic Fibrosis Foundation, or CFF, OrbiMed Israel GP Ltd., or Orbimed, and Nimble Ventures LLC our stockholders, each of which holding more than 5% of our outstanding Common Stock, relating to a private placement of an aggregate of 15,997,448 shares of our Common Stock and 14,610,714 pre-funded warrants, at a purchase price of $0.245 per Share and $0.244 per pre-funded warrant. The gross proceeds from this offering are approximately $7.4 million, before deducting issuance costs. The pre-funded warrants became exercisable on May 4, 2023, at an exercise price of $0.001 per share of Common Stock and have no expiration date. Of these proceeds, an aggregate of 3,385,000 shares of Common Stock and 4,778,265 pre-funded warrants were sold to CF for gross proceeds of $2 million, an aggregate of 1,740,000 shares of Common Stock and 9,280,408 pre-funded warrants were sold to Orbimed for gross proceeds of $2.7 million and an aggregate of 4,550,000 shares of Common Stock and 552,041 pre-funded warrants were sold to Nimble Venture LLC for gross proceeds of $1.25 million and (ii) a Securities Purchase Agreement we entered into on March 6, 2024 with certain investors, including CFF, Orbimed and Telmina Limited, or Telmina, our stockholders, each of which hold more than 5% of our outstanding Common Stock, pursuant to which we sold an aggregate of 216,417 shares of Convertible Preferred Stock and Private Placement Warrants to purchase up to an aggregate of 108,208,500 shares of Common Stock, at a combined purchase price of $231.10 per share of Series X Preferred Stock and accompanying Private Placement Warrant. The aggregate gross proceeds from this offering were approximately $50 million. The Private Placement Warrants may be exercised at any time following stockholder approval of the conversion of all issued and outstanding Series X Preferred Stock and the exercise of all Private Placement Warrants in accordance with the listing rules of NYSE American, which we are obligated to bring to the stockholders vote by no later than August 12, 2024, will have an exercise price of $0.2311 and expire on the 24-month anniversary of the date on which they are first exercisable. The exercise price of the Private Placement Warrants is subject to customary adjustments for stock dividends, stock splits, reclassifications and the like. Of these proceeds, an aggregate of 21,635 shares of Convertible Preferred Stock and 10,817,500 Private Placement Warrants were sold to CFF for gross proceeds of $5 million, an aggregate of 4,327 shares of Convertible Preferred Stock and 2,163,500 Private Placement Warrants were sold to Orbimed for gross proceeds of $1 million and an aggregate of 2,596 shares of Convertible Preferred Stock and 1,298,000 Private Placement Warrants were sold to Telmina for gross proceeds of $0.6 million.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The following is a summary and description of fees billed by us to Kesselman & Kesselman, Certified Public Accountants (Isr.) for the fiscal years ended December 31, 2023 and December 31, 2022.
| Fiscal year ended December 31, 2023 | Fiscal year ended December 31, 2022 | |||||||
| Audit fees(1) | $ | 126,000 | $ | 126,000 | ||||
| Audit-related fees(2) | $ | 97,000 | $ | 24,969 | ||||
| Tax fees(3) | $ | 3,393 | $ | - | ||||
| All other fees | $ | - | $ | - | ||||
| Total fees | $ | 226,393 | $ | 150,969 | ||||
| (1) | Audit Fees include fees for professional services rendered for the quarterly reviews of the interim consolidated financial statements and the annual audit of our consolidated financial statements included in our Annual Report on Form 10-K. |
| (2) | Audit-Related Fees include fees for services that were reasonably related to the performance of the audit of the annual consolidated financial statements for the fiscal year, other than Audit Fees, such as for services in connection with the Sale Agreement, our February 2023 PIPE and a registration statement filed for the re-sale of certain shares of Common Stock by selling stockholders. |
| (3) | Tax Fees include fees for tax compliance and tax advice. |
98
Pre-Approval Policies and Procedures
The Audit Committee approves all audit and pre-approves all non-audit services provided by our independent registered public accounting firm before it is engaged by us to render non-audit services. These services may include audit-related services, tax services and other services.
The pre-approval requirement set forth above does not apply with respect to non-audit services if:
| ● | all such services do not, in the aggregate, amount to more than 5% of the total fees paid by us to our independent registered public accounting firm during the fiscal year in which the services are provided; |
| ● | such services were not recognized as non-audit services at the time of the relevant engagement; and |
| ● | such services are promptly brought to the attention of and approved by the Audit Committee (or its delegate) prior to the completion of the annual audit. |
Pre-Approval Policies and Procedures
The Audit Committee approves all audit and pre-approves all non-audit services provided by our independent registered public accounting firm before it is engaged by us to render non-audit services. These services may include audit-related services, tax services and other services.
The pre-approval requirement set forth above does not apply with respect to non-audit services if:
| ● | all such services do not, in the aggregate, amount to more than 5% of the total fees paid by us to our independent registered public accounting firm during the fiscal year in which the services are provided; |
| ● | such services were not recognized as non-audit services at the time of the relevant engagement; and |
| ● | such services are promptly brought to the attention of and approved by the Audit Committee (or its delegate) prior to the completion of the annual audit. |
99
part IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
| (a) | The following are filed with this Annual Report: |
| (1) | The financial statements listed on the Financial Statements’ Table of Contents | |
| (2) | Not applicable |
| (b) | Exhibits |
The following exhibits are filed as part of this Annual Report or are incorporated by reference.
EXHIBIT INDEX
100
| * | Portions of this exhibit have been omitted pursuant to Rule 601(b)(10) of Regulation S-K. The omitted information is not material and would likely cause competitive harm to the Company if publicly disclosed. |
| ** | Indicates a management contract or a compensatory plan or agreement. |
| *** |
Filed herewith. |
| **** | Furnished herewith. |
Item 16. Form 10-K Summary
None.
101
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Exchange Act of 1934, the registrant caused this Annual Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| BIOMX INC. | ||
| Dated: April 3, 2024 | By: | /s/ Jonathan Solomon |
| Name: | Jonathan Solomon | |
| Title: | Chief Executive Officer | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Company and in the capacities and on the dates indicated.
| Signature | Title | Date | ||
| /s/ Jonathan Solomon | Chief Executive Officer | April 3, 2024 | ||
| Jonathan Solomon | (Principal Executive Officer) and Director | |||
| /s/ Avraham Gabay | Interim Chief Financial Officer | April 3, 2024 | ||
| Avraham Gabay | (Principal Financial Officer and Principal Accounting Officer) |
|||
| /s/ Russell Greig | Chairman of the Board of Directors | April 3, 2024 | ||
| Dr. Russell Greig | ||||
| /s/ Jesse Goodman | Director | April 3, 2024 | ||
| Dr. Jesse Goodman | ||||
| /s/ Jonathan Leff | Director | April 3, 2024 | ||
| Jonathan Leff | ||||
| /s/ Gregory Merril | Director | April 3, 2024 | ||
| Gregory Merril | ||||
| /s/ Alan Moses | Director | April 3, 2024 | ||
| Dr. Alan Moses | ||||
| /s/ Eddie Williams | Director | April 3, 2024 | ||
| Eddie Williams |
102
BIOMX INC.
CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2023
CONTENTS
| Page | ||
| REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM (PCAOB name: Kesselman & Kesselman C.P.A.s , PCAOB ID: | F-2 | |
| CONSOLIDATED FINANCIAL STATEMENTS: | ||
| Consolidated Balance Sheets | F-3 - F-4 | |
| Consolidated Statements of Operations | F-5 | |
| Consolidated Statements of Changes in Stockholders’ Equity | F-6 | |
| Consolidated Statements of Cash Flows | F-7 - F-8 | |
| Notes to the Consolidated Financial Statements | F-9 - F-34 |
F-1
Report of Independent Registered Public Accounting Firm

To the Board of Directors and stockholders of BiomX Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of BiomX Inc. and its subsidiaries (the “Company”) as of December 31, 2023 and 2022 and the related consolidated statements of operations, changes in stockholders' equity and cash flows for each of the two years in the period ended December 31, 2023, including the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022 and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2023 in conformity with accounting principles generally accepted in the United States of America.
Substantial Doubt about the Company’s Ability to Continue as a Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1C to the consolidated financial statements, the Company has incurred significant losses and negative cash flows from operations, incurred an accumulated deficit, and has stated that these events or conditions raise substantial doubt on the Company’s ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1C. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these consolidated financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that (i) relate to accounts or disclosures that are material to the consolidated financial statements and (ii) involved our especially challenging, subjective, or complex judgments. We determined there are no critical audit matters.
/s/
Certified Public Accountants (Isr.)
A member of PricewaterhouseCoopers International Limited
April 3, 2024
We have served as the Company's auditor since 2021.
F-2
BIOMX INC.
CONSOLIDATED BALANCE SHEETS
(USD in thousands, except share and per share data)
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | ||||||||
| Restricted cash | ||||||||
| Short-term deposits | ||||||||
| Other current assets | ||||||||
| Total current assets | ||||||||
| Non-current assets | ||||||||
| Operating lease right-of-use assets | ||||||||
| Property and equipment, net | ||||||||
| Total non-current assets | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-3
BIOMX INC.
CONSOLIDATED BALANCE SHEETS
(USD in thousands, except share and per share data)
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities | ||||||||
| Trade account payables | ||||||||
| Current portion of lease liabilities | ||||||||
| Other account payables | ||||||||
| Current portion of long-term debt | ||||||||
| Total current liabilities | ||||||||
| Non-current liabilities | ||||||||
| Contract liability | ||||||||
| Long-term debt, net of current portion | ||||||||
| Operating lease liabilities, net of current portion | ||||||||
| Other liabilities | ||||||||
| Total non-current liabilities | ||||||||
| Commitments and Contingencies (Note 10) | ||||||||
| Stockholders’ equity | ||||||||
| Preferred Stock, $ | ||||||||
| Common stock, $ | ||||||||
| Additional paid in capital | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Stockholders’ equity | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-4
BIOMX INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(USD in thousands, except share and per share data)
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Research and development (“R&D”) expenses, net | ||||||||
| Amortization of intangible assets | ||||||||
| General and administrative expenses | ||||||||
| Operating loss | ||||||||
| Other income | ( | ) | ( | ) | ||||
| Interest expenses | ||||||||
| Finance income, net | ( | ) | ( | ) | ||||
| Loss before tax | ||||||||
| Tax expenses | ||||||||
| Net Loss | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-5
BIOMX INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(USD in thousands, except share and per share data)
| Common stock | Additional paid in | Accumulated | Total Stockholder’ | |||||||||||||||||
| Shares | Amount | capital | deficit | equity | ||||||||||||||||
| Balance as of December 31, 2021 | ( | ) | ||||||||||||||||||
| Issuance of Common Stock under Open Market Sales Agreement net of $ | ||||||||||||||||||||
| Stock-based compensation expenses | - | |||||||||||||||||||
| Proceeds on account of shares (***) | ||||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | |||||||||||||||
| Balance as of December 31, 2022 | ( | ) | ||||||||||||||||||
| Issuance of Common Stock and warrants under Private Investment in Public Equity (“PIPE”), net of $ | ||||||||||||||||||||
| Reissuance of treasury stock (***) | ||||||||||||||||||||
| Stock-based compensation expenses | - | |||||||||||||||||||
| Issuance of Common Stock under Open Market Sales Agreement (**) | ||||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | |||||||||||||||
| Balance as of December 31, 2023 | ( | ) | ||||||||||||||||||
| (*) |
| (**) | |
| (***) |
The accompanying notes are an integral part of the consolidated financial statements.
F-6
BIOMX INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(USD in thousands, except share and per share data)
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| CASH FLOWS – OPERATING ACTIVITIES | ||||||||
| Net loss | ( | ) | ( | ) | ||||
| Adjustments required to reconcile net loss to cash flows used in operating activities | ||||||||
| Depreciation and amortization | ||||||||
| Stock-based compensation | ||||||||
| Amortization of debt issuance costs | ||||||||
| Finance income, net | ( | ) | ( | ) | ||||
| Changes in other liabilities | ( | ) | ( | ) | ||||
| Capital loss, net | ||||||||
| Changes in operating assets and liabilities: | ||||||||
| Other current assets | ||||||||
| Trade account payables | ( | ) | ||||||
| Other account payables | ( | ) | ||||||
| Net change in operating leases | ( | ) | ( | ) | ||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| CASH FLOWS – INVESTING ACTIVITIES | ||||||||
| Investment in short-term deposits | ( | ) | ||||||
| Proceeds from short -term deposits | ||||||||
| Purchase of property and equipment | ( | ) | ( | ) | ||||
| Proceeds from sale of property and equipment | ||||||||
| Net cash provided by (used in) investing activities | ( | ) | ||||||
| CASH FLOWS – FINANCING ACTIVITIES | ||||||||
| Issuance of Common Stock under Open Market Sales Agreement, net of issuance costs | ||||||||
| Issuance of Common Stock and warrants under PIPE | ||||||||
| Issuance costs from PIPE | ( | ) | ||||||
| Repayment of long-term debt | ( | ) | ||||||
| Proceeds on account of shares | ||||||||
| Net cash provided by financing activities | ||||||||
| Decrease in cash and cash equivalents and restricted cash | ( | ) | ( | ) | ||||
| Effect of exchange rate changes on cash and cash equivalents and restricted cash | ||||||||
| Cash and cash equivalents and restricted cash at the beginning of the year | ||||||||
| Cash and cash equivalents and restricted cash at the end of the year | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-7
BIOMX INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(USD in thousands, except share and per share data)
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| RECONCILIATION OF AMOUNTS ON CONSOLIDATED BALANCE SHEETS: | ||||||||
| Cash and cash equivalents | ||||||||
| Restricted cash | ||||||||
| Total cash and cash equivalents and restricted cash | ||||||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | ||||||||
| Cash paid for interest | ||||||||
| Taxes paid | ||||||||
| SUPPLEMENTAL DISCLOSURE OF NON-CASH INVESTING ACTIVITIES: | ||||||||
| Property and equipment purchases included in accounts payable | ||||||||
The accompanying notes are an integral part of the consolidated financial statements.
F-8
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 1 - | GENERAL |
| A. | General information: |
BiomX Inc., (individually, and together with its subsidiaries,
BiomX Ltd. and RondinX Ltd., the “Company” or “BiomX”) was incorporated as a blank check company on
On October 29, 2019, the Company merged with BiomX Israel,
who survived the merger as a wholly owned subsidiary of BiomX Inc. The Company acquired all outstanding shares of BiomX Israel. In exchange,
shareholders of BiomX Israel received
On February 6, 2020, the Company’s Common Stock also began trading on the Tel-Aviv Stock Exchange. On July 6, 2022, the Company announced a voluntary delisting of its shares of Common Stock from the Tel-Aviv Stock Exchange which became effective on October 6, 2022.
BiomX is developing both natural and engineered phage cocktails designed to target and destroy harmful bacteria in chronic diseases, focusing its efforts at this point on cystic fibrosis and to a lesser degree on atopic dermatitis. BiomX discovers and validates proprietary bacterial targets and customizes phage compositions against these targets. The Company’s headquarters are located in Ness Ziona, Israel.
On March 6, 2024, the Company entered into an agreement and plan of merger (the “Merger Agreement”) with Adaptive Phage Therapeutics Inc., a Delaware corporation (“APT”), and certain other parties, as a result of which APT became a wholly-owned subsidiary of the Company (the “Acquisition”). See note 1D for further information regarding the Acquisition.
| B. | Israel-Hamas war |
On October 7, 2023, an unprecedented attack was launched against Israel by terrorists from the Hamas terrorist organization that infiltrated Israel’s southern border from the Gaza Strip and in other areas within the state of Israel attacking civilians and military targets while simultaneously launching extensive rocket attacks on the Israeli population. These attacks resulted in extensive deaths, injuries and kidnapping of civilians and soldiers. In response, the Security Cabinet of the State of Israel declared war against Hamas and a military campaign against these terrorist organizations commenced in parallel to their continued rocket and terror attacks. In addition, Hezbollah, an Islamist terrorist group that controls large portions of southern Lebanon, has attacked military and civilian targets in Northern Israel, to which Israel has responded.
To date, the State of Israel continues to be at war with Hamas and on an armed conflicts with Hezbollah.
BiomX headquarters and principal offices and most of its operations are located in the State of Israel. In addition, all of the key employees and officers are residents of Israel. Accordingly, political, economic and military conditions in Israel and the surrounding region may directly affect its business.
While a few employees of the Company were called to reserve duty in the Israel Defense Forces, the ongoing war with Hamas has not, since its inception, materially impacted BiomX business or operations. Furthermore, BiomX does not expect any delays to its programs as a result of the situation. However, at this time, it is not possible to predict the intensity or duration of Israel’s war against Hamas, nor predict how this war will ultimately affect BiomX business and operations or Israel’s economy in general.
F-9
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 1 - | GENERAL (Cont.) |
| C. | Going concern |
The Company has incurred significant losses and negative
cash flows from operations and incurred an accumulated deficit of $
| D. | Merger Agreement |
On March 6, 2024, the Company, entered into the Merger Agreement with BTX Merger Sub I, Inc., a Delaware corporation and a wholly owned subsidiary of the Company (“First Merger Sub”), BTX Merger Sub II, LLC, a Delaware limited liability company and wholly owned subsidiary of the Company (“Second Merger Sub”), and APT. Pursuant to the Merger Agreement, First Merger Sub merged with and into APT, with APT being the surviving corporation and becoming a wholly owned subsidiary of the Company (the “First Merger”). Immediately following the First Merger, APT merged with and into Second Merger Sub, pursuant to which Second Merger Sub was the surviving entity (together with the First Merger, the “Acquisition”). The Acquisition is intended to qualify as a tax-free reorganization for U.S. federal income tax purposes.
On March 15, 2024, the effective time of the Acquisition,
APT’s former stockholders were issued an aggregate of
Concurrently with the consummation of the Acquisition, the
Company entered into a securities purchase agreement with certain investors, pursuant to which such investors purchased an aggregate of
Immediately following the Acquisition, and without taking
into account the PIPE Preferred Shares and the Private Placement Warrants, the Company’s stockholders prior to the Acquisition owned
approximate
F-10
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 1 - | GENERAL (Cont.) |
| D. | Merger Agreement (Cont.) |
The Acquisition will be accounted in accordance with Accounting Standards Codification Topic 805, “Business Combinations,” using the acquisition method of accounting. The Company was identified as the accounting acquirer, based on the evaluation of the following facts and circumstances:
| ● | Pursuant to the Merger Agreement, the post-Acquisition board of directors of the Company consists of seven directors, out of which the Company designated four board seats, with the Company’s chair of the board prior to the Acquisition continuing in his position, i.e. the majority of the post-closing board was designated by the Company. |
| ● | The Chief Executive Officer and the majority of management roles are held by individuals who were affiliated with the Company prior to the Acquisition. |
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES |
The significant accounting policies applied in the preparation of the financial statements on a consistent basis, are as follows, except for the adoption of new accounting standards:
| A. | Basis of presentation and principles of consolidation |
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States (“GAAP”) and include the accounts of the Company and its wholly owned subsidiaries, BiomX Israel and RondinX Ltd. All intercompany accounts and transactions have been eliminated in consolidation.
| B. | Use of estimates in the preparation of financial statements |
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities in the financial statements and the amounts of expenses during the reported years. The most significant estimates in the Company’s financial statements relate to accruals for research and development expenses and valuation of stock-based compensation awards. These estimates and assumptions are based on current facts, future expectations, and various other factors believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities and the recording of expenses that are not readily apparent from other sources. Actual results may differ materially and adversely from these estimates.
The full extent to which the Israel-Hamas war may directly or indirectly impact the Company’s business, results of operations and financial condition will depend on future developments that are uncertain, as well as the economic impact on local, regional, national and international markets.
| C. | Functional currency and foreign currency translation |
The functional currency of the Company is the U.S. dollar (“USD”) since the dollar is the currency of the primary economic environment in which the Company has operated and expects to continue to operate in the foreseeable future. Transactions and balances originally denominated USD are presented at their original amounts. Balances in non-USD currencies are translated into USDs using historical and current exchange rates for non-monetary and monetary balances, respectively. For non-USD transactions and other items in the statements of income (indicated below), the following exchange rates are used: (i) for transactions – exchange rates at transaction dates or average exchange rates; and (ii) for other items (derived from non-monetary balance sheet items such as depreciation and amortization) – historical exchange rates. Currency transaction gains and losses are presented in finance income, net as appropriate.
| D. | Cash and cash equivalents and restricted cash |
The Company considers cash equivalents to be all short-term, highly liquid investments, which include money market funds, that are not restricted as to withdrawal or use, and short-term bank deposits with original maturities of three months or less from the date of purchase that are not restricted as to withdrawal or use and are readily convertible to known amounts of cash. Restricted cash consists of funds that are contractually restricted to a credit line for outstanding short-term foreign exchange contracts and bank guarantee due to rental agreements. The Company has presented restricted cash separately from cash and cash equivalents in the consolidated balance sheets. The Company includes its restricted bank deposits in cash and cash equivalents when reconciling beginning-of-period and end-of-period total amounts shown on the combined statement of cash flows.
F-11
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| E. | Concentrations of credit risk |
Financial instruments which potentially subject us to credit risk consist primarily of cash, cash equivalents, and short-term deposits. These amounts at times may exceed federally insured limits. We have not experienced any credit losses in such accounts and do not believe we are exposed to any significant credit risk on these funds. Most of the Company’s cash and cash equivalents and bank deposits are invested in major banks in the U.S. and Israel. Management believes that the credit risk with respect to the financial institutions that hold the Company’s cash and cash equivalents and bank deposits is low. Refer to note 2J.
| F. | Property and equipment |
Property and equipment are presented at cost less accumulated
depreciation.
| Estimated Useful Lives | ||
| Laboratory equipment | ||
| Computers and software | ||
| Equipment and furniture | ||
| Leasehold improvements |
| G. | Long-lived assets |
In accordance with ASC 360-10, “Impairment and Disposal of Long-Lived Assets”, management reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable based on estimated future undiscounted cash flows. If so indicated, an impairment loss would be recognized for the difference between the carrying amount of the asset and its fair value. For the years ended December 31, 2023 and 2022, no impairment expenses were recorded.
| H. | Income taxes |
The Company accounts for income taxes using the asset and liability approach. Deferred tax assets and liabilities are recorded based on the differences between the financial statement and tax basis of assets and liabilities and the tax rates in effect when these differences are expected to reverse. Deferred tax assets are reduced by a valuation allowance if, based on the weight of available evidence, it is more likely than not that some or all the deferred tax assets will not be realized. As of December 31, 2023 and 2022, the Company had a full valuation allowance against deferred tax assets.
The Company is subject to the provisions of ASC 740-10-25, “Income Taxes” (“ASC 740”). ASC 740 prescribes a more likely-than-not threshold for the financial statement recognition of uncertain tax positions. ASC 740 clarifies the accounting for income taxes by prescribing a minimum recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. On a yearly basis, the Company undergoes a process to evaluate whether income tax accruals are in accordance with ASC 740 guidance on uncertain tax positions. The Company has not recorded any liability for uncertain tax positions for the years ended December 31, 2023 and 2022. The Company presents unrecognized tax benefits as a reduction to deferred tax asset where a net operating loss, a similar tax loss, or a tax credit carryforward that are available, under the tax law of the applicable jurisdiction, to offset any additional income taxes that would result from the settlement of a tax position.
| I. | Derivative activity |
The Company uses foreign exchange contracts (option and
forward contracts) to hedge cash flows from currency exposure. These foreign exchange contracts are not designated as hedging instruments
for accounting purposes. In connection with these foreign exchange contracts, the Company recognizes gains or losses that offset the revaluation
of the cash flows also recorded under financial expenses (income), net in the consolidated statements of operations. As of December 31,
2023, the Company had outstanding short-term foreign exchange contracts for the exchange of USD to NIS in the amount of approximately
$
F-12
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| J. | Fair value of financial instruments |
The Company accounts for financial instruments in accordance with ASC 820, “Fair Value Measurements and Disclosures” (“ASC 820”). ASC 820 establishes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurements) and the lowest priority to unobservable inputs (Level 3 measurements). The three levels of the fair value hierarchy under ASC 820 are described below:
Level 1 – Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities.
Level 2 – Quoted prices in non-active markets or in active markets for similar assets or liabilities, observable inputs other than quoted prices, and inputs that are not directly observable but are corroborated by observable market data.
Level 3 – Prices or valuations that require inputs that are both significant to the fair value measurement and unobservable.
There were no changes in the fair value hierarchy levelling during the years ended December 31, 2023 and 2022.
| December 31, 2023 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Fair Value | |||||||||||||
| Assets: | ||||||||||||||||
| Cash equivalents: | ||||||||||||||||
| Money market funds | ||||||||||||||||
| Foreign exchange contracts receivable | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Contingent consideration | ||||||||||||||||
| December 31, 2022 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Fair Value | |||||||||||||
| Assets: | ||||||||||||||||
| Cash equivalents: | ||||||||||||||||
| Money market funds | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Contingent consideration | ||||||||||||||||
| Foreign exchange contracts payable | ||||||||||||||||
F-13
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| J. | Fair value of financial instruments (Cont.) |
Financial instruments with carrying values approximating fair value include cash and cash equivalents, restricted cash, short-term deposits, other current assets, trade accounts payable and other current liabilities, due to their short-term nature.
The Company determined the fair value of the liabilities
for the contingent consideration based on a probability discounted cash flow analysis. This fair value measurement is based on significant
unobservable inputs in the market and thus represents a Level 3 measurement within the fair value hierarchy. The fair value of the contingent
consideration is based on several factors, such as: the attainment of future clinical, developmental, regulatory, commercial and strategic
milestones relating to product candidates for treatment of primary sclerosing cholangitis. The discount rate applied ranged from
| K. | Defined contribution plans |
Under Israeli employment laws, employees of BiomX Israel are included under Section 14 of the Severance Compensation Act, 1963 (“Section 14”) for a portion of their salaries. Pursuant to Section 14, these employees are entitled to monthly deposits made by the Company on their behalf with insurance companies.
Payments in accordance with Section 14 release the Company
from any future severance payments (under the Israeli Severance Compensation Act, 1963) with respect of those employees. The aforementioned
deposits are not recorded as an asset on the Company’s balance sheet, and there is no liability recorded as the Company does not
have a future obligation to make any additional payments. The Company’s contributions to the defined contribution plans are charged
to the consolidated statements of operations as and when the services are received from the Company’s employees. Total expenses
with respect to these contributions were $
For U.S. employees the Company has a defined contribution savings plan under Section 401(k) of the Internal Revenue Code. This plan covers substantially all employees of BiomX Inc in the U.S. who meet minimum age and service requirements and allows participants to defer a portion of their annual compensation on a pre-tax basis.
The Company has not elected to match any of the employee’s deferral. During the years ended December 31, 2023 and 2022 the Company did not record any expenses for 401(k) match contributions.
| L. | Financial instruments |
When the Company issues freestanding instruments, it first analyzes the provisions of ASC 480, “Distinguishing Liabilities From Equity” (“ASC 480”) in order to determine whether the instrument should be classified as a liability, with subsequent changes in fair value recognized in the consolidated statements of operations in each period. If the instrument is not within the scope of ASC 480, the Company further analyzes the provisions of ASC 815-10 in order to determine whether the instrument is considered indexed to the entity’s own stock and qualifies for classification within equity. All warrants issued by the Company are classified within stockholders’ equity as “Additional paid-in capital”. Equity classification is permitted when warrants are indexed to the Company’s own shares and meet the classification requirements for stockholders’ equity classification of ASC 815-40,”Contracts in Entity’s Own Equity” (“ASC 815-40”).
F-14
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| M. | Research and development costs |
Research and development costs are charged to statements of operations as incurred. Royalty-bearing grants from the IIA are recognized at the time the Company is entitled to such grants, on the basis of the costs incurred and applied as a deduction from research and development expenses.
| N. | Basic and diluted loss per share |
Basic loss per share is computed by dividing net loss by
the weighted average number of shares of Common Stock outstanding during the year, fully vested warrants with no exercise price for the
Company’s Common Stock and fully vested Pre-Funded Warrants for the Company’s Common Stock at an exercise price of $
| O. | Stock compensation plans |
The Company applies ASC 718-10, “Stock-Based Payment,” (“ASC 718-10”) which requires the measurement and recognition of compensation expenses for all stock-based payment awards made to employees and directors including employee stock options under the Company’s stock plans based on estimated fair values.
ASC 718-10 requires companies to estimate the fair value of stock-based payment awards granted to employees and non-employees on the date of grant using an option-pricing model. The fair value of the award is recognized as an expense over the requisite service periods in the Company’s statements of operations using the graded vesting method. The Company accounts for share-based payment awards classified as equity awards. The Company recognizes stock-based award forfeitures as they occur rather than estimate by applying a forfeiture rate.
All issuances of stock options or other equity instruments to non-employees as consideration for goods or services received by the Company are accounted for based on the fair value of the equity instruments issued.
F-15
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| O. | Stock compensation plans (Cont.) |
The Company estimates the fair value of stock options granted as equity awards using a Black-Scholes option-pricing model. The option-pricing model requires a number of assumptions, of which the most significant are share price, expected volatility and the expected option term (the time from the grant date until the options are exercised or expire). The Company uses an average historical stock price volatility based on a combined weighted average of the Company’s historical average volatility and that of a selected peer group of comparable public companies within the biotechnology and pharmaceutical industry that were deemed to be representative of future stock price trends as the Company does not have a sufficient historical trading history of its own Common Stock. The Company will continue to apply this process until a sufficient amount of historical information regarding the volatility of its own stock price becomes available. The Company has historically not paid dividends and has no foreseeable plans to issue dividends. The risk-free interest rate is based on the yield from governmental zero-coupon bonds with an equivalent term. The expected option term is calculated for all stock option grants using the “simplified” method. Changes in the determination of each of the inputs can affect the fair value of the options granted and the results of operations of the Company.
| P. | Leases |
Under Accounting Standards Update, “Leases” (“ASC 842”), the Company determines if an arrangement is a lease at inception. Upon initial recognition, the Company recognizes a liability at the present value of the lease payments to be made over the lease term, and concurrently recognizes a right-of-use asset at the same amount of the liability, adjusted for any prepaid or accrued lease payments, plus initial direct costs incurred in respect of the lease. The Company uses its incremental borrowing rate based on the information available at the commencement date to determine the present value of the lease payments. The subsequent measurement depends on whether the lease is classified as a finance lease or an operating lease. During the reporting periods, the Company has only operating leases. Lease terms include options to extend the lease when it is reasonably certain that the Company will exercise that option. Lease expenses for operating leases are recognized on a straight-line basis over the lease term.
The Company has made a policy election not to capitalize leases with a term of 12 months or less.
In accordance with ASC 360-10, management reviews operating lease assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable based on estimated future undiscounted cash flows. If so indicated, an impairment loss would be recognized for the difference between the carrying amount of the asset and its fair value.
| Q. | Treasury stock |
Treasury shares are presented as a reduction of equity, at their cost to the Company.
F-16
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 2 - | SIGNIFICANT ACCOUNTING POLICIES (Cont.) |
| R. | New accounting pronouncements |
Recently adopted accounting pronouncements
In June 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) 2016-13, “Financial Instruments—Credit Losses—Measurement of Credit Losses on Financial Instruments.” This guidance replaces the current incurred loss impairment methodology with a methodology that reflects expected credit losses and requires consideration of a broader range of reasonable and supportable information to inform credit loss estimates. The guidance is effective for smaller reporting companies (as defined by the rules under the Securities Exchange Act of 1934, as amended) for the fiscal year beginning on January 1, 2023, including interim periods within that year. The Company adopted the guidance on January 1, 2023, and has concluded the adoption did not have a material impact on its consolidated financial statements.
In October 2021, the FASB issued ASU 2021-08, “Business Combinations (Topic 805), Accounting for Contract Assets and Contract Liabilities from Contracts with Customers”, which requires contract assets and contract liabilities acquired in a business combination to be recognized and measured by the acquirer on the acquisition date in accordance with ASC 606. The guidance will result in the acquirer recognizing contract assets and contract liabilities at the same amounts recorded by the acquiree. The guidance should be applied prospectively to acquisitions occurring on or after the effective date. The guidance is effective for fiscal years beginning after December 15, 2022, including interim periods within those fiscal years. Effective January 1, 2023, the Company has concluded the adoption has not a material impact on its consolidated financial statements.
In December 2022, the FASB issued ASU 2022-06, Reference Rate Reform (Topic 848): Deferral of the Sunset Date of Topic 848. This ASU extends the temporary optional practical expedients for reference rate reform related activities that impact debt, leases, derivatives and other contracts through December 31, 2024. The Company adopted the guidance immediately and has concluded the adoption did not have a material impact on its consolidated financial statements.
Recently issued accounting pronouncements, not yet adopted
In November 2023, the FASB issued ASU 2023-07 “Segment Reporting: Improvements to Reportable Segment Disclosures” (“ASU 2023-07”). This guidance expands public entities’ segment disclosures primarily by requiring disclosure of significant segment expenses that are regularly provided to the chief operating decision maker and included within each reported measure of segment profit or loss, an amount and description of its composition for other segment items, and interim disclosures of a reportable segment’s profit or loss and assets that are currently required annually. Public entities with a single reportable segment are required to provide the new disclosures and all the disclosures required under ASC 280, Segment Reporting. The guidance is effective for fiscal years beginning after December 15, 2023, and interim periods within fiscal years beginning after December 15, 2024, with early adoption permitted. The amendments are required to be applied retrospectively to all prior periods presented in an entity’s financial statements. The Company is currently evaluating the impact that the adoption of ASU 2023-07 may have on its consolidated financial statements.
In December 2023, the FASB issued ASU 2023-09 “Income Taxes (Topic 740): Improvements to Income Tax Disclosures” (“ASU 2023-09”). This guidance is intended to enhance the transparency and decision-usefulness of income tax disclosures. The amendments in ASU 2023-09 address investor requests for enhanced income tax information primarily through changes to disclosure regarding rate reconciliation and income taxes paid both in the U.S. and in foreign jurisdictions. ASU 2023-09 is effective for fiscal years beginning after December 15, 2024 on a prospective basis, with the option to apply the standard retrospectively. Early adoption is permitted. The Company is currently evaluating this guidance to determine the impact it may have on its consolidated financial statements disclosures.
| NOTE 3 - | SHORT-TERM DEPOSITS |
Short-term deposits represent time deposits placed with banks with original maturities of greater than three months but less than one year. Interest earned is recorded as finance income, net in the consolidated statements of operations during the years for which the Company held short-term deposits.
As of December 31, 2023, the Company had deposits. As
of December 31, 2022, the Company had deposits in USD at
F-17
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 4 - | OTHER CURRENT ASSETS |
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Government institutions | ||||||||
| Prepaid insurance | ||||||||
| Other prepaid expenses | ||||||||
| Grants receivables | ||||||||
| Other | ||||||||
| NOTE 5 - | PROPERTY AND EQUIPMENT, NET |
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Computers and software | ||||||||
| Laboratory equipment | ||||||||
| Equipment and furniture | ||||||||
| Leasehold improvements | ||||||||
| Accumulated depreciation | ( | ) | ( | ) | ||||
Substantially all of the Company’s non-current assets are concentrated in Israel.
Depreciation expenses were $
| NOTE 6 - | ACQUISITION OF SUBSIDIARY |
In November 2017, BiomX Israel signed a share purchase agreement
with the shareholders of RondinX Ltd. In accordance with the share purchase agreement, BiomX Israel acquired
The contingent consideration is accounted for at fair value (level 3). There were no changes in the fair value hierarchy levelling during the years ended December 31, 2023 and December 31, 2022. Refer to note 2J.
The consolidated financial statements as of December 31,
2023 and 2022 include a liability with respect to this agreement in the amount of $
Intangible asset acquired in the RondinX Ltd. Acquisition
was fully amortized as of December 31, 2022. For the year ended December 31, 2022, amortization expense recorded in the consolidated statements
of operations was $
F-18
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 7 - | LEASES |
In September 2020, BiomX Israel entered into a lease agreement for
office space in Ness Ziona, Israel for five years beginning on September 1, 2020, with an option to extend for an additional period until
November 30, 2030. The monthly lease payments under the lease agreement are approximately $
On October 1, 2020, the Company entered into a lease agreement
for office space in Branford, Connecticut, U.S., for 25 months beginning on October 5, 2020. Monthly lease payments under the agreement
are approximately $
In August 2022, BiomX Israel entered into a sublease agreement
for a portion of its office space in Ness Ziona, Israel. The agreement is for a period of two years beginning on August 15, 2022. The
monthly lease payments under the agreement are approximately $
Lease expenses recorded in the consolidated statements of
operations were $
| Year ended December 31, 2023 | Year ended December 31, 2022 | |||||||
| Cash payments for operating leases | ||||||||
| Operating Leases | ||||
| 2024 | ||||
| 2025 | ||||
| 2026 | ||||
| 2027 | ||||
| 2028 | ||||
| 2029 | ||||
| 2030 | ||||
| Total operating lease payments | ||||
| Less imputed interest | ( | ) | ||
| Total operating lease liability balance | ||||
| NOTE 8 - | OTHER ACCOUNT PAYABLES |
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Employees and related institutions | ||||||||
| Accrued expenses | ||||||||
| Government institutions | ||||||||
| Deferred fees from collaboration agreements and prepaid sublease income | ||||||||
| Other | - | |||||||
F-19
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 9 - | TRANSACTION WITH RELATED PARTIES |
| A. | In October 2019, BiomX Israel entered into a loan agreement in the amount of $ |
| B. | Refer to note 12A regarding a Securities Purchase Agreement with institutional investors, all of the Company’s directors and certain executive officers. |
| C. | Refer to note 12B regarding stock options granted to related parties. |
| NOTE 10 - | COMMITMENTS AND CONTINGENCIES |
| A. | In March 2021, the IIA approved
two new applications in relation to the Company’s cystic fibrosis product candidate for an aggregate budget of NIS |
In August 2021, the IIA approved an application that supports
upgrading the Company’s manufacturing capabilities for an aggregate budget of NIS
In March 2022, the IIA approved an application for a total
budget of NIS
In March 2023, the IIA approved an application for a total
budget of NIS
According to the agreements with the IIA, BiomX Israel will
pay royalties of
Through December 31, 2023, total grants approved from the
IIA aggregated to approximately $
| B. |
F-20
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 10 - | COMMITMENTS AND CONTINGENCIES (Cont.) |
In July 2019, the Company and Yeda Research and Development Company Limited (“Yeda”) amended the Research and License Agreement (the “License Agreement”) entered into in 2015. Pursuant to the amendment, following the closing of the Recapitalization Transaction, the provisions of the Yeda license agreements related to the Exit Fee were amended so that the Company is obligated to pay Yeda a one-time payment as described in the amendment which will not exceed 1% of the consideration received in the event of any merger or acquisition involving the Company instead of the Exit Fee, with respect to each license agreement. The Merger Agreement as described in note 1D, does not apply for such merger or acquisition as defined in the amendment.
| C. | As successor in interest to RondinX Ltd., BiomX Israel is a party to a license agreement dated March 20, 2016 with Yeda, pursuant to which the Company has a worldwide exclusive license to Yeda’s know-how, information and patents related to the Company’s meta-genomics target discovery platform. As consideration for the license, the Company is obligated to pay annual license fees of $ |
| D. | In December 2017, BiomX Israel signed a patent license agreement with Keio University and JSR Corporation in Japan. According to the agreement, BiomX Israel received an exclusive patent license to certain patent rights related to inflammatory bowel disease (“IBD”) In return, the Company will pay an annual license fee of between $ |
In April 2019, BiomX Israel signed an additional patent license agreement
with Keio University and JSR Corporation in Japan. According to the agreement, BiomX Israel received an exclusive sublicense by JSR to
certain patent rights related to the treatment of primary sclerosing cholangitis. In return, the Company is required (i) to pay a license
issue fee of $
F-21
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 10 - | COMMITMENTS AND CONTINGENCIES (Cont.) |
| F. | On June 23, 2022 (the “Effective Date”), BiomX Israel entered into a research collaboration
agreement with Boehringer Ingelheim International GmbH (“BI”) for a collaboration to identify biomarkers for IBD. Under the
agreement, BiomX Israel is eligible to receive fees totaling $ |
| G. | Refer to note 7 for information regarding the Company’s lease liabilities. |
| NOTE 11 - | LONG-TERM DEBT |
On August 16, 2021, the Company entered into a Loan and
Security Agreement (the “Loan Agreement”) with Hercules Capital, Inc. (“Hercules”), with respect to a venture
debt facility. Under the Loan Agreement, Hercules provided the Company with access to a term loan with an aggregate principal amount of
up to $
The Company may prepay advances under the Loan Agreement,
in whole or in part, at any time subject to a prepayment charge equal to:
Interest on the term loan accrues at a per annum rate equal
to the greater of
F-22
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 11 - | LONG-TERM DEBT (Cont.) |
As of December 31, 2023, the carrying value of the term
loan consists of $
Interest expense relating to the term loan, which is included
in interest expense in the consolidated statements of operations was $
Under the terms of the Loan Agreement, the Company granted
first priority liens and security interests in substantially all of the Company’s intellectual property as collateral for the obligations
thereunder. The Company also granted Hercules the right, at their discretion, to participate in any closing of any single subsequent broadly
marketed financing as defined up to a maximum aggregate amount of $
| December 31, 2023 | ||||
| 2024 | ||||
| 2025 | ||||
| Total principal payments | ||||
| Unamortized discount, debt issuance costs and accretion of End of Term Charge | ||||
| Total future principal payments | $ | |||
| Current portion of long-term debt | ( | ) | ||
| Long-term debt, net | $ | |||
| NOTE 12 - | STOCKHOLDERS EQUITY |
| A. | Share Capital: |
Common Stock:
On August 24, 2022, the Company’s stockholders approved
increasing the number of authorized shares of Common Stock from
Treasury Stock:
Refer to note 9A.
Initial Public Offering:
On December 18, 2018, the Company consummated its initial
public offering (“IPO”) of
Simultaneous with the consummation of the IPO, the Company
consummated the private placement of an aggregate of
F-23
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| A. | Share Capital: (Cont.) |
Stock Exchange:
As detailed in note 1, as part of the Recapitalization Transaction
on October 28, 2019, the Company issued
In addition, the Company also agreed to issue the following number of additional shares of Common Stock, in the aggregate, to stockholders on a pro rata basis, subject to the Company’s achievement of the conditions specified below following the recapitalization transaction (all with respect to the Company’s Common Stock traded on the NYSE American):
| A. |
| B. |
Private Investment in Public Equity:
On February 22, 2023, the Company entered
into a Securities Purchase Agreement to issue and sell an aggregate of
The exercise of the outstanding Pre-Funded
Warrants is subject to a beneficial ownership limitation between
F-24
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| A. | Share Capital: (Cont.) |
At-the-market Sales Agreement:
In December 2020, pursuant to a registration statement
on Form S-3 declared effective by the Securities and Exchange Commission on December 11, 2020, the Company entered into an Open Market
Issuance Sales Agreement (“ATM Agreement”) with Jefferies LLC. (“Jefferies”), which provided that, upon the terms
and subject to the conditions and limitations in the ATM Agreement, the Company could elect, from time to time, to offer and sell shares
of Common Stock having an aggregate offering price of up to $
In December 2023, pursuant to a registration statement on Form S-3 declared effective by the Securities and Exchange
Commission on January 2, 2024, the Company entered into an Open Market Offering Agreement with H.C. Wainwright & Co., LLC (“Wainwright”),
pursuant to which the Company may issue and sell shares of Common Stock having an aggregate offering price of up to $
Maruho Agreement:
In October 2021, the Company entered into a Stock Purchase
Agreement with a subsidiary of Maruho Co. Ltd., (“Maruho”), a leading dermatology-focused pharmaceutical company in Japan,
pursuant to which the Company issued to Maruho
F-25
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| A. | Share Capital: (Cont.) |
CFF Agreement:
In December 2021, the Company entered
into a Securities Purchase Agreement with the CF Foundation, an organization that historically played a role in supporting the development
of innovative therapies for patients suffering from cystic fibrosis (CF). Under the terms of the agreement, the Company will receive up
to $
Preferred Stock:
The Company is authorized to issue
Warrants:
| The Public Warrants became exercisable upon the closing of the Recapitalization Transaction. No fractional shares will be issued upon exercise of the Public Warrants. Therefore, the Public Warrants must be exercised in multiples of two warrants. The Public Warrants will expire
The Company may redeem the Public Warrants: |
| ● | in whole and not in part; | |
| ● | at a price of $ | |
| ● | at any time during the exercise period; | |
| ● | upon a minimum of 30 days prior written notice of redemption; | |
| ● | if, and only if, the last sale price of the Company’s Common Stock equals or exceeds $ | |
| ● | if, and only if, there is a current registration statement in effect with respect to the shares of Common Stock underlying such warrants at the time of redemption and for the entire |
If the Company calls the Public Warrants for redemption, management will have the option to require all holders that wish to exercise the Public Warrants to do so on a “cashless basis,” as described in the warrant agreement. The exercise price and number of shares of Common Stock issuable upon exercise of the warrants may be adjusted in certain circumstances including in the event of a stock dividend, or recapitalization, reorganization, merger or consolidation. However, the warrants will not be adjusted for issuance of Common Stock at a price below their exercise price. Additionally, in no event will the Company be required to net cash settle the warrants.
F-26
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| A. | Share Capital: (Cont.) | |
| Warrant | Issuance Date | Expiration Date | Exercise Price Per Share | Number of Shares of Common Stock Underlying Warrants | ||||||||
| Public Warrants | ||||||||||||
| 2021 Registered Direct Offering Warrants | ||||||||||||
| Pre-Funded Warrants | ||||||||||||
| Pre-Funded Warrants | ||||||||||||
| B. | Stock-based compensation: |
Equity Incentive Plan:
In 2015, the Board of Directors of BiomX Israel approved a plan for the allocation of options to employees, service providers, and officers (the “2015 Plan”). The options represented a right to purchase one Ordinary Share of the BiomX Israel in consideration of the payment of an exercise price. Also, the options were granted in accordance with the “capital gains route” under section 102 and section 3(i) of the Israeli Income Tax Ordinance and section 409A of the U.S. Internal Revenue Code as technically adjusted following the Recapitalization Transaction on October 28, 2019.
As of December 31, 2023, there are no shares of Common Stock remaining for issuance under the 2015 Plan.
In 2019, the Company adopted a new incentive plan (the “2019
Plan”) to grant
The aggregate number of shares of Common Stock that may
be delivered pursuant to the 2019 Plan will automatically increase on January 1 of each year, commencing on January 1, 2020 and ending
on (and including) January 1, 2029, in an amount equal to four percent (
Notwithstanding the foregoing, the Board may act prior to January 1 of a given year to provide that there will be no January 1 increase for such year or that the increase for such year will be a lesser number of shares of Common Stock than provided herein.
As of December 31, 2023, there were
F-27
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| B. | Stock-based compensation: (Cont.) | |
Stock Options:
On March 29, 2022, the Board of Directors approved the grant
of
On June 21, 2022, the Board of Directors approved the grant
of
On August 22, 2022, the Board of Directors approved the
grant of
On September 30, 2022, the Board of Directors approved the
grant of
On March 1, 2023, the Board of Directors approved the grant
of
On August 21, 2023, the Board of Directors approved the
grant of
On October 19, 2023, the Board of Directors approved the
grant of
On October 29, 2023, the Board of Directors approved the
grant of
On October 29, 2023, the Board of Directors approved a reduction
in the exercise price (“the Repricing”) of each outstanding option to purchase shares of the Company’s Common Stock
currently held by employees of BiomX with an original exercise price above $
On November 9, 2023, the Company filed with the Securities
and Exchange Commission a Tender Offer Statement defining the terms and conditions of a one-time voluntary stock option exchange of certain
eligible options for its employees (the “Option Exchange”). the Company offered to exchange certain out-of-the-money stock
options for new stock options at an exchange ratio of between
| 2023 | 2022 | |||||||
| Underlying value of Common Stock ($) | ||||||||
| Exercise price ($) | ||||||||
| Expected volatility (%) | ||||||||
| Expected terms of the option (years) | ||||||||
| Risk-free interest rate (%) | ||||||||
F-28
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| B. | Stock-based compensation: (Cont.) |
Stock Options: (Cont.)
Total fair value embodied in the options granted in 2023
and 2022 at the grant date, is estimated to be $
As of December 31, 2023, the unrecognized compensation cost
related to all unvested, equity classified stock options of $
| For year ended December 31, 2023 | ||||||||||||
| Number of Options | Weighted average exercise price | Aggregate intrinsic value | ||||||||||
| Outstanding at the beginning of period | $ | $ | ||||||||||
| Granted | ||||||||||||
| Forfeited/canceled | ( | ) | ||||||||||
| Replacement options granted | ||||||||||||
| Expired | ( | ) | ||||||||||
| Exercised | $ | |||||||||||
| Outstanding at the end of period | $ | |||||||||||
| Exercisable at end of period | ||||||||||||
| Weighted average remaining contractual life – years as of December 31, 2023 | ||||||||||||
Warrants:
| Warrant | Issuance Date | Expiration Date | Exercise Price Per Share | Number of Shares of Common Stock Underlying Warrants | ||||||||
| Private Warrants issued to scientific founders (see below) | | |||||||||||
F-29
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 12 - | STOCKHOLDERS EQUITY (Cont.) |
| B. | Stock-based compensation: (Cont.) |
Warrants: (Cont.)
| In November 2017, BiomX Israel issued |
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Research and development expenses, net | ||||||||
| General and administrative | ||||||||
The Company recognized stock-based compensation expenses
in connection with options granted to executive officers of the Company in the amount of $
F-30
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 13 - | RESEARCH AND DEVELOPMENT EXPENSES, NET |
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Professional service and subcontractors | ||||||||
| Salaries and related expenses | ||||||||
| Stock-based compensation | ||||||||
| Depreciation | ||||||||
| Materials and supplies | ||||||||
| Rent and related expenses | ||||||||
| Other | ||||||||
| Less change in contingent liabilities (see Note 10D) | ( | ) | - | |||||
| Less income from collaboration agreements (see Note 10F) | ( | ) | ( | ) | ||||
| Less grants from the IIA (see Note 10A) | ( | ) | ( | ) | ||||
| NOTE 14 - | GENERAL AND ADMINISTRATIVE EXPENSES |
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Salaries and related expenses | ||||||||
| Stock-based compensation | ||||||||
| Professional services | ||||||||
| Travel expenses | ||||||||
| Rent and related expenses | ||||||||
| Insurance expenses | ||||||||
| Other | ||||||||
| NOTE 15 - | FINANCE EXPENSES (INCOME), NET |
| Year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Exchange rate differences | ( | ) | ( | ) | ||||
| Interest income from bank deposits | ( | ) | ( | ) | ||||
| Bank fees and other | ||||||||
| Loss (income) from foreign exchange contracts | ( | ) | ||||||
| ( | ) | ( | ) | |||||
F-31
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 16 - | INCOME TAXES |
| A. | The Company files income tax returns in the U.S. federal jurisdiction and in state and local jurisdictions and is subject to examination by the various taxing authorities. The Company’s income tax returns since 2020 remain open and subject to examination. The statutory U.S. federal income tax rate is | |
| B. | BiomX Ltd. and RondinX Ltd. file income tax returns in Israel. Their tax assessments through 2017 are deemed to be final. The statutory Israeli income tax rate is |
| C. | As of December 31, 2023 and 2022, BiomX Israel had total carryforward losses in Israel of approximately $ |
| D. | Management has considered the Company’s history of cumulative net losses incurred since inception and its lack of commercialization of any products or generation of any revenue from product sales since inception and has concluded that it is more likely than not that the Company will not realize the benefits of the deferred tax assets. Accordingly, a full valuation allowance has been established against the deferred tax assets as of December 31, 2023 and 2022. Management reevaluates the positive and negative evidence at each reporting period. | |
| E. | The Company’s policy is to record estimated interest and penalties related to uncertain tax positions in income tax expense. The Company has no amounts recorded for any unrecognized tax positions, accrued interest or penalties as of December 31, 2023 and 2022. |
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Statutory U.S. federal income tax rate | ( | )% | ( | )% | ||||
| U.S. vs foreign tax rate differential | ( | ) | ( | ) | ||||
| Change in deferred tax asset valuation allowance | ||||||||
| Effective tax rate | % | % | ||||||
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| United States | ||||||||
| Israel | ||||||||
| As of December 31, | ||||||||
| 2023 | 2022 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | ||||||||
| Research and development expenses, net | ||||||||
| Lease liability | ||||||||
| Other | ||||||||
| Total deferred tax assets | ||||||||
| Deferred tax liabilities: | ||||||||
| Right of use assets | ( | ) | ( | ) | ||||
| Fixed assets | ( | ) | ( | ) | ||||
| Total deferred tax liabilities | ( | ) | ( | ) | ||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Net deferred tax assets | ||||||||
F-32
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 17 - | BASIC LOSS PER SHARE |
| For the year ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Net loss | ||||||||
Basic loss per share is computed on the basis of the net
loss for the period divided by the weighted average number of shares of Common Stock outstanding during the period, fully vested warrants
with no exercise price for the Company’s Common Stock and fully vested Pre-Funded Warrants for the Company’s Common Stock
at an exercise price of $
As of December 31, 2023, the basic loss per share calculation included
a weighted average number of
The calculation of diluted loss per share as of December
31, 2023 does not include
The calculation of diluted loss per share as of December
31, 2022 does not include
| NOTE 18 - | CORPORATE RESTRUCTURING |
On May 24, 2022, the Company announced a Corporate Restructuring,
intended to extend the Company’s capital resources, while prioritizing the Company’s ongoing cystic fibrosis program and delaying
the Company’s atopic dermatitis program.
F-33
BIOMX INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(USD in thousands, except share and per share data)
| NOTE 19 - | SUBSEQUENT EVENTS |
| A. | On January 18, 2024, the Company received the last instalment of $ |
| B. | On March 6, 2024, the Company entered into a Merger Agreement with APT and certain other parties, as a result of which APT became a wholly-owned subsidiary of the Company. See note 1D for further information. Under the disclosure requirements of Accounting Standards Codification Topic 805, “Business Combinations”, the Company is required to provide information regarding the effect of the business combination. Due to the following limitations, the initial accounting for the business combination was incomplete at the time of the issuance of the financial statements, therefore, the Company did not include the above mentioned information as permitted by ASC 805-10-50-4 and ASC 805-30-50-3. |
| a. | The Acquisition closed on March 15, 2024, while the filing date of the Company’s annual financial statements in its annual report on Form 10-K is April 3, 2024. |
| b. | Full and final financial data of APT was not available to the Company by the filing date of the Company’s annual financial statements in form 10-K. |
| c. | The Company hasn’t completed the work of the purchase price allocation needed under ASC 805. |
| C. | On March 6, 2024, concurrently with the consummation of the Acquisition,
the Company entered into a securities purchase agreement with certain investors for aggregate gross proceeds of $
| |
| D. | On March 19, 2024, the Company prepaid all of the term loan under the
Term Loan Facility in a total of $
| |
| E. | On March 21, 2024, RondinX signed an agreement with the Israeli tax
authority in respect to an assessment for the years 2018-2022. The agreement concluded that RondinX’s IP and employees were transferred
to BiomX Israel on the acquisition date. As a result, RondinX had a capital gain equal to its carryforward losses of $ |
F-34